| Posted: February 28, 2007 |
Next generation, sequentially assembled ultrathin films: beyond electrostatics |
|
(Nanowerk News) Frank Caruso and co-workers at the University of Melbourne, Australia, discuss how non-covalent interactions can be used for layer-by-layer surface modification.
|
|
The surface properties of a material are extremely important in many different areas, from biomedicine to optics. For instance, the life of a contact lens will be greatly affected by its surface, mediating the extent to which proteins, lipids and other biomolecules are adsorbed. Likewise, a very thin layer at the surface of a window or mirror can greatly affect its wettability, and therefore its usefulness in wet conditions. In these cases, modifying only the top few nanometres of a surface can make a profound difference to the way the material behaves macroscopically. Surface modification is therefore an area of intense scientific activity.
|
|
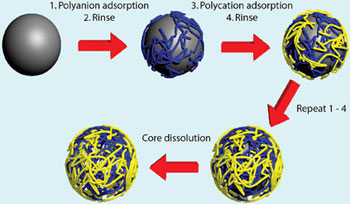
The layer-by-layer (LbL) assembly technique is one of the most versatile approaches for modifying surfaces. This method exploits the sequential adsorption of complementary species onto a surface, allowing stepwise buildup of a film of desired thickness and composition. The power of this approach is found in its simplicity and versatility: it is almost trivial experimentally and the number of materials that can be used is astounding. Of particular note is that the approach is readily transferable to three-dimensional surfaces, which has allowed the preparation of a wide variety of functional core-shell particles and hollow capsules, in addition to modification of planar surfaces. The genesis of LbL assembly occurred in 1991, when Decher and co-workers used electrostatic forces to construct a coating by alternately exposing positive and negatively charged polymers.
|
 |
|
Recent work has demonstrated that the driving forces that can be used to assemble LbL films extend beyond electrostatic interactions ("Next generation, sequentially assembled ultrathin films: beyond electrostatics"). For example, hydrogen bonding, sequential covalent reactions and specific molecular recognition processes (such as DNA hybridization) are being used in LbL surface modification. This has stimulated interest in the approach as a key methodology in thin film construction, whereby almost any chemical functionality can be imparted to a surface.
|
|
In our full review article, recent approaches that are likely to have a significant impact on the preparation of multilayer thin films on surfaces are examined. The use of secondary non-electrostatic interactions is important because it opens up LbL assembly to a host of materials that are not inherently charged, but are of interest from an applications standpoint. Therefore, exploiting non-electrostatic interactions becomes an essential tool for using these materials in stepwise, controlled surface modification.
|
|
The most prominent secondary interaction used so far in LbL assembly is hydrogen bonding. Materials that present either a hydrogen-bonding donor or acceptor are adsorbed sequentially to build up a film. Some of these films have the advantage of being susceptible to disassembly via pH variation, which is a useful attribute in controlled release applications such as drug delivery. The transfer of hydrogen-bonded LbL films from planar to colloidal surfaces is of considerable research interest as a means of preparing particulate-based drug delivery systems.
|
|
Within hydrogen-bonded LbL films, one powerful subset is prepared via DNA hybridization. This approach is especially useful as the interaction between each individual base pair can be prescribed by using oligonucleotides with defined sequence composition. This can be used to control the final film properties, such as thickness and density. DNA is being used more as a building block in nanoscale material science, and its potential in surface modification is considerable.
|
|
While hydrogen bonding and DNA hybridization are useful for incorporating stimuli-responsive behaviour and orientational control to LbL films, they are limited to materials that contain the appropriate chemical moieties. However, another approach to LbL modified surfaces is the use of covalent interactions. The advent of approaches like the recently introduced 'click' chemistry has made covalent surface modification under very mild conditions possible. By incorporating reactive moieties into a polymer that is otherwise unsuitable for LbL, the technique can be made applicable to almost any material. This will see LbL, used in tandem with synthetic polymer chemistry, become an increasingly common method for modifying surfaces.
|
|
|

