| Posted: April 2, 2007 |
Scientists track remarkable 'breathing' in nanoporous materials |
|
(Nanowerk News) Scientists all over the world are participating in the quest of new materials with properties suitable for the environmentally friendly and economically feasible separation, recovery, and reuse of vapours and greenhouse gases. A team of scientists from France, UK and the European Synchrotron Radiation Facility (ESRF) have recently discovered an unprecedented giant and reversible swelling of nanoporous materials with exceptional properties: huge flexibility and profound selectivity. Their results were recently published in Science.
|
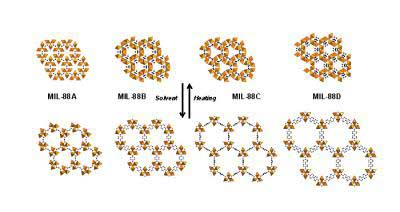 |
| Structures (along the c axis) of the MIL-88A, B, C, D series in their dry forms (top) and open forms (bottom). (Image: Institut Lavorsier)
|
|
Porous hybrid solids are the new materials that could make the world more environmentally friendly. The team from Institut Lavoisier at University of Versailles have developed metal-organic three-dimensional structures with cages and channels (known as MIL, for Material Institut Lavoisier). These compounds contain metal ions (in this case chromium and iron), with organic linkers and are very flexible, and hence, can change shape very easily. They can open up or close down to external factors such as pressure, temperature, light or influence of gases and solvents.
|
|
The French researchers, in collaboration with the staff of the Swiss-Norwegian experimental station (called beamline) at the ESRF, have tracked, for the first time, a reversible giant increase in volume of these solids. It ranges from 85% of their size to up the unprecedented 230%. Such a large expansion in crystalline materials has not been observed before. This reversible “breathing” action is similar to the lungs’ function in humans: they grow in size when inhaling and go back to their original size when exhaling. The lungs expand, however, by only around 40%.
|
|
The huge swelling effect has been achieved in a simple way: MIL materials were immersed into solvents, and their cavities were filled and thus opened by entering solvent molecules. This made the structures grow, without breaking bonds and retaining the crystallinity of the materials. This process was monitored at the ESRF, using high-quality synchrotron radiation and the experimental results were combined with computer chemistry simulations.
|
|
This process can reversed by heating the solvated form the dry form is recovered. In this form, the material exhibited closed pores with almost no accessible porosity. Surprisingly, the same team published a paper last autumn where they showed that some gas molecules can close, but not open, the pores upon absorption. Moreover, the closed hydrated form demonstrates a remarkable selectivity in absorption of polar and nonpolar gases.
|
|
The next step for the team now is to investigate how hydrogen or green-house gases can be stored in these kinds of materials. This may open a door to ecological applications such as hydrogen cars or the capture of carbon dioxide in the near future.
|

