| Aug 17, 2011 |
Noble metals and semiconductor nanocrystals combine to make nanocomposites with superior catalytic activity
|
|
(Nanowerk News) When metals are shrunk to nanoscale proportions, their properties often change in useful ways. For example, gold—normally an inert metal—can become an active catalyst when made into particles a few tens of atoms in size. However, such benefits are not restricted to metals alone. By combining metals and semiconductor crystals together, it is possible to produce novel composites with properties not attainable from pure metals.
|
|
Jackie Y. Ying and her former group member Jun Yang from the A*STAR Institute of Bioengineering and Nanotechnology have now developed a metal–semiconductor nanocomposite that can improve the performance of fuel cells ("Nanocomposites of Ag2S and Noble Metals").
|
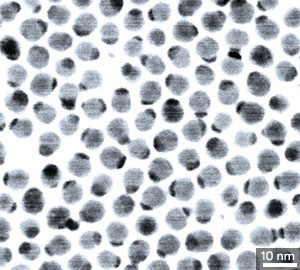 |
| A transmission electron microscopy image of silver sulfide nanocrystals with gold grown on one face.
|
|
The nanocomposite was made by depositing various metals, including gold, platinum and osmium, onto silver sulfide nanocrystals (pictured). Multiple metals could be sequentially deposited onto the nanocrystal surface, which makes it possible to control the catalytic properties of the final composite.
|
|
The researchers tested the catalytic performance of their platinum-containing nanocomposites in a methanol fuel cell—an electrochemical device that breaks down methanol to produce electricity. At the anode side of the fuel cell, methanol is catalytically converted by a methanol oxidation reaction (MOR) into carbon dioxide, releasing hydrogen and electrons. The electrons flow around an electrical circuit before recombining with the hydrogen and oxygen in an oxygen reduction reaction (ORR) at the cathode, producing water as a by-product.
|
|
In commercial fuel cells, the anode is made of platinum and carbon. Ying found that the nanocomposites outperformed conventional platinum–carbon anodes. The composite of silver sulfide, gold and platinum, with and without osmium, performed particularly well.
|
|
The researchers explain that the remarkable catalytic performance of the anodes is due to the high surface area of the platinum deposits on the nanocrystals, which provides a larger electrochemically active surface for the MOR, and also the enhanced electronic properties of the nanocomposite itself. "Electron donation from the silver sulfide and gold to platinum leads to a substantial increase in the electron density around the platinum sites," says Ying. This prevents carbon monoxide, an intermediate in the MOR, from sticking to the platinum and 'poisoning' the catalyst.
|
|
The next step in the research is to develop an efficient electrocatalyst for the cathode side of the fuel cell. This reaction is normally hampered by hydroxyl intermediates, which stick to the catalyst. "Our laboratory is developing core–shell alloy–semiconductor nanocrystals in which preferential crystalline planes are exposed to minimize hydroxyl adsorption to facilitate the ORR," says Ying.
|

