| Posted: December 26, 2007 |
Nanotechnology pioneer Ferrari test-launching multi-stage drug delivery system |
|
(Nanowerk News) Nanomedicine pioneer Mauro Ferrari, Ph.D., and colleagues at The University of Texas Health Science Center at Houston have developed a new way to get intravenous agents for cancer and other diseases to the parts of the body where they’re needed most and to reduce the amount that goes to others areas where unintended injury can occur.
|
|
UT-Houston’s Mauro Ferrari, Ph.D., has begun preclinical trials on a new nanoscale drug delivery system that could increase efficacy and reduce toxicity.
|
|
Their innovative nanoscale drug delivery system uses a multi-stage approach to guide nanocarriers that are about 100 times smaller than a strand of hair through the blood system and to the site of diseased cells where they release a payload of diagnostic agents or medications, or both.
|
|
“NanoMedicine is about getting the right amount of medication to the right location at the right time,” said Ferrari, who heads the 40-person NanoMedicine Research Center in the university’s Brown Foundation Institute of Molecular Medicine for the Prevention of Human Diseases (IMM), as well as the Alliance for NanoHealth, a consortium made up of UT-Houston and six other institutions.
|
|
Nanomedicine, an offshoot of nanotechnology, refers to highly specific medical intervention at the molecular scale. A nanometer is a billionth of a meter.
|
|
Currently being tested on human tumors in animal models, the system developed in Ferrari’s IMM lab could allow physicians to get the same results with less medication. Only a tiny fraction of an injected drug now reaches its intended target. Too much medication can injure a patient—too little, and there are no therapeutic effects.
|
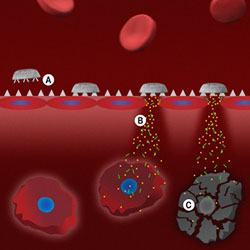 |
| A new multi-stage drug delivery system developed in the laboratory of nanomedicine pioneer Dr. Mauro Ferrari, delivers therapeutic or diagnostic agents directly at the site of a tumor or other problem area. This illustration shows the final stage of the intravenous journey with the arrival of a nanocarrier which is about 100 times smaller than a strand of hair and its payload of anti-cancer medication. (A) shows the injected nanocarrier landing on the inner wall of a tumor-associated blood vessel, (B) the release of nanoparticles that penetrate both the blood vessel wall and the tumor cell membrane and, (C) the delivery to the tumor of doses of a cancer killing medication. Ferrari is testing the system on human tumors in animal models. (Illustration by Matthew Landry)
|
|
“The multi-stage approach to nano drug delivery will increase efficacy and reduce toxicity,” Ferrari said. “We hope to reduce systemic exposure which will allow current drugs to be more effective. Furthermore, newer drugs may have a better chance of coming to market because of reduced toxicity profiles.”
|
|
Getting these nanocarriers and their contents from the tip of a syringe to diseased cells is no easy task. The nanocarriers must avoid being corrupted by enzymes, swallowed up by the body’s immune system and trapped inside blood vessels.
|
|
“This is kind of like opening a door. You need to open all ten doors to get to the diseased cells. It’s not enough to open nine,” Ferrari said.
|
|
Thus, the idea for the multiple stages that is similar to the concept of a solid-fuel NASA rocket.
|
|
“The first stage goes to the inner wall of a blood vessel near the diseased cells. As the nanocarrier degrades, it releases the second stage – tiny nanoparticles that penetrate the walls of the inner blood vessels and enter the diseased cells. The third stage is then released and it consists of either the medication to kill the tumor cells or the contrasting agents to get quality medical images of it, or both” said Ennio Tasciotti, Ph.D., the senior postdoctoral researcher in Ferrari’s IMM lab.
|
|
The nanocarriers are engineered to seek out blood vessels that sprout to support diseased cells, which differ from normal vessels. This is accomplished by coating the nanocarriers with targeting molecules that can recognize and latch onto the cells lining the inner walls of disease-associated blood vessels. Targeting molecules include antibodies, peptides, oligonucleotides and aptamers.
|
|
The nanocarriers have pores that expand as the nanocarrier degrades and allow the nanoparticles inside to escape, Tasciotti said. The nanoparticles then travel through the tiny holes in disease-associated blood vessels and into the targeted cells, where the therapeutic or imaging agents are released.
|
|
Supported with recently-awarded grants totaling $4.3 million from the National Cancer Institute (NCI) and three organizations in the U.S. Department of Defense, Ferrari’s IMM lab is working closely on the multi-stage project with scientists from the Alliance for NanoHealth.
|
|
Department of Defense and NCI officials are particularly interested in applying this emerging technology to breast cancer, which is the focus of the initial preclinical tests. (The American Cancer Society reports that 40,460 women will die from breast cancer in the United States this year.)
|
|
Ferrari’s multi-stage drug delivery system is envisioned to be used one day in treating heart disease, hemorrhaging and other conditions with disease-associated changes in blood vessel arrangement.
|
|
Ferrari’s IMM lab, which develops therapeutic and diagnostic platforms through nano-engineering, mathematical modeling and biomedical sciences, is also working on intelligent implants for controlled time release of therapeutic agents and on silicon microchips for early disease detection of blood proteomic signatures.
|

