| Posted: May 23, 2008 |
Looking beneath the surface |
|
(Nanowerk News) Historically, analysis of the behavior of individual proteins has required the physical destruction of the cells in which they are found. More recently, however, a new generation of microscopy techniques has emerged that make it possible to directly visualize individual fluorescently tagged molecules within the living cell, giving scientists unprecedented capabilities to observe biological processes in their natural context.
|
|
“These techniques enable us to visualize molecular dynamics and interactions, to analyze molecular mechanisms quantitatively, and to detect biomolecules with great sensitivity in living cells,” explains Makio Tokunaga, an imaging specialist at the RIKEN Research Center for Allergy and Immunology in Yokohama.
|
|
One popular technique is total internal reflection fluorescence (TIRF) microscopy, which takes advantage of the physics of refraction to specifically excite fluorescent molecules in the immediate proximity of the microscope objective. Unfortunately, although TIRF is useful for single-molecule visualization, its limited depth of visualization means that it can only observe targets located near the cell surface.
|
|
In order to overcome this limitation, Tokunaga’s team developed a new variant of TIRF, which they term highly-inclined and laminated optical sheet (HILO) microscopy ("Highly inclined thin illumination enables clear single-molecule imaging in cells"). HILO makes use of an alternative refraction strategy, in which the laser beam that illuminates the sample is converted into a thin sheet that passes through the center of the specimen. HILO is capable of imaging targets at depths of tens of microns—well beyond the cell membrane—and can be used to generate three-dimensional reconstructions via the computerized assembly of multiple scans into a single image.
|
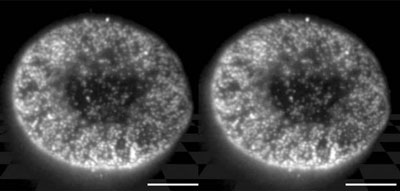 |
| A stereo pair of images demonstrating the three-dimensional reconstruction of the distribution of nuclear pore complexes within the nuclear membrane from a serial set of HILO images. Scale bars = 5.0 µm. (Image: RIKEN)
|
|
In testing out HILO, the researchers started big—relatively—by imaging nuclear pore complexes (NPCs), the massive multiprotein assemblies that act as the gateway between the nucleus and cytoplasm. The researchers obtained remarkably clear images of these complexes, with virtually no background haze. They subsequently used HILO to visualize the movement of fluorescently labeled importin β, a protein that shuttles other molecules into the nucleus via the NPCs, generating videos that show the kinetics of nuclear import at high resolution and in real-time.
|
|
Tokunaga sees HILO as a promising tool for immunology research. “We intend to visualize signaling pathways from the cell membrane to the nucleus after stimulation using single-molecule microscopy,” he says. By combining this real-time imaging data with sophisticated computational modeling strategies, it should be possible to gain unprecedented insight into complex cellular pathways. “We aim to open up new frontiers for understanding immune cells as molecular systems,” concludes Tokunaga.
|

