| Aug 27, 2008 |
Nanoparticles stick a perfect landing
|
|
(Nanowerk News) A silicon nanoparticle flying at 8 times the speed of sound can slam into a surface and stick, but it bounces off if colliding at half that speed. This puzzling observation is now explained by computer simulations reported in the August Physical Review B (" Efficient sticking of surface-passivated Si nanospheres via phase-transition plasticity").
|
|
The pressure of impact causes the higher speed particle to change its crystalline structure, which soaks up so much energy that the particle can't bounce away. These results may help researchers who are developing wear-resistant coatings that are created by many such high-speed impacts.
|
|
Steven Girshick and his team at the University of Minnesota in Minneapolis are designing super-hard, wear-resistant coatings for machine tools. In their patented technique, a spray of particles--each a few nanometers across and containing silicon or some combination of chemicals--strikes a target substrate, such as a silicon surface, with velocities between 1 and 2 kilometers per second. The lumpy coating is built up as these nanoparticles stick to the surface. This technique can create similar rough-textured coatings that are being developed for other purposes, such as for sensors and catalysts.
|
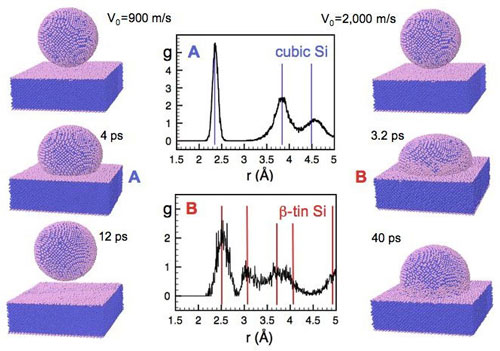 |
| On the rebound. A nanoparticle containing some 30,000 silicon atoms and moving at 900 meters per second will bounce off a surface (left sequence), but at 2,000 meters per second, it sticks (right sequence), according to computer simulations. The higher-speed impact causes two sequential changes in the crystalline structure. (Image: T. Dumitrică/Univ. of Minnesota)
|
|
Girshick's team uses high-speed nanoparticles because they stick without splattering, unlike lower-speed particles, but the team never understood why the higher speed helps. Sticking implies that the kinetic energy is changed into another form, or "dissipated." If these were macroscopic objects--like sticky BBs shot into a wall--the energy would dissipate through buckling or crumpling in the crystalline structure. "But at the nanoscale, there is not enough space or time to create these dislocations," says Traian Dumitrică, also at the University of Minnesota.
|
|
To investigate where the energy goes, Dumitrică and graduate student Mayur Suri ran computer simulations of a hypersonic nanoparticle. They began with a sphere of 30,000 silicon atoms arranged in a diamond-like crystalline structure in which each atom bonds to four neighbors. They then simulated this nanoparticle smashing into a silicon surface.
|
|
They found that for speeds less than 1.2 kilometers per second, the nanoparticle bounces off the surface like a basketball. But at higher speeds, some of the nanoparticle undergoes a phase transition to a compressed state called β-tin, where each atom bonds to six neighbors. This transition is surprising, Dumitrică says, because the collision energy is not high enough to induce a phase transition in a macroscopic object. However, the impact force is applied over a few square nanometers, so the pressure inside the nanoparticle is extremely large--around 200,000 atmospheres, which is more than enough to cause the phase transition.
|
| The β-tin state only lasts a few picoseconds, though. As the nanoparticle begins to bounce back, there is a second phase transition to an amorphous, or disordered, state. The combination of the two phase transitions, plus some heat generation, takes up all of the kinetic energy, and the particle remains on the surface. After all of this action, "the recoil is too weak to beat the adhesion forces between the nanoparticle and the substrate," Dumitrică says.
|
|
Girshick says this work provides new insight into what makes a nanoparticle stick or bounce. This information could help engineers tune the speeds to increase the adhesion of a nanostructured film to a substrate. "Adhesion is a very important property, especially if you are using the film in an aggressive environment like industrial machining," he says.
|
|
|

