| Posted: September 15, 2006 |
Materials scientists tame tricky carbon nanotubes |
|
(Nanowerk News) Based on a new theory, MIT scientists may be able to manipulate carbon nanotubes -- one of the strongest known materials and one of the trickiest to work with -- without destroying their extraordinary electrical properties.
|
|
The work is reported in the Sept. 15 issue of Physical Review Letters, the journal of the American Physical Society.
|
|
Carbon nanotubes -- cylindrical carbon molecules 50,000 times thinner than a human hair -- have properties that make them potentially useful in nanotechnology, electronics, optics and reinforcing composite materials. With an internal bonding structure rivaling that of another well-known form of carbon, diamonds, carbon nanotubes are extraordinarily strong and can be highly efficient electrical conductors.
|
|
The problem is working with them. There is no reliable way to arrange the tubes into a circuit, partly because growing them can result in a randomly oriented mess resembling a bowl of spaghetti.
|
|
Researchers have attached to the side walls of the tiny tubes chemical molecules that work as "handles" that allow the tubes to be assembled and manipulated. But these molecular bonds also change the tubes' structure and destroy their conductivity.
|
|
Now Young-Su Lee, an MIT graduate student in materials science and engineering, and Nicola Marzari, an associate professor in the same department, have identified a class of chemical molecules that preserve the metallic properties of carbon nanotubes and their near-perfect ability to conduct electricity with little resistance.
|
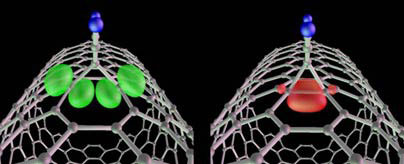 |
MIT researchers have discovered that certain molecules can attach themselves to metallic carbon nanotubes without interfering with the nanotubes' exceptional ability to conduct electricity. At left, the high conductance state has two molecular orbitals, shown in green. Some molecules even let the nanotube switch between highly conductive, left, and poorly conductive (right, with one red molecular orbital), creating the potential for new applications. (Image courtesy Marzari Lab)
|
|
Using these molecules as handles, Marzari and Lee said, could overcome fabrication problems and lend the nanotubes new properties for a host of potential applications as detectors, sensors or components in novel optoelectronics.
|
|
Playing with atoms
|
|
Marzari and Lee use the fundamental laws of quantum mechanics to simulate material properties that are difficult or impossible to measure, such as molten lava in the Earth's core or atomic motion in a fast chemical reaction. Then they run these simulations on interconnected PCs and use the results to optimize and engineer novel materials such as electrodes for fuel cells and polymers that contract and expand like human muscles.
|
|
With the help of a powerful algorithm created by Lee and published last year in Physical Review Letters, the theorists focused on solving some of the problems of working with carbon nanotubes.
|
|
Like fuzzy balls and Velcro, the hexagon of carbon that makes up a nanotube has a predilection for clinging to other hexagons. One of the many challenges of working with the infinitesimally small tubes is that they tend to stick to each other.
|
|
Attaching a molecule to the sidewall of the tube serves a double purpose: It stops nanotubes from sticking so they can be processed and manipulated more easily, and it allows researchers to control and change the tubes' electronic properties. Still, most such molecules also destroy the tubes' conductance because they make the tube structurally more similar to a diamond, which is an insulator, rather than to graphite, a semi-metal.
|
|
Lee and Marzari used Lee's algorithm to identify a class of "molecular handles" (carbenes and nitrenes) that stop this from happening and preserve the tubes' original conductivity. "We now have a way to attach molecules that allows us to manipulate the nanotubes without losing their conductance," Marzari said.
|
|
Carbenes and nitrenes work by breaking a molecular bond on the nanotube's wall while creating their own new bond to the tube. This process -- one bond formed, one bond lost -- restores the perfect number of bonds each carbon atom had in the original tube and "conductance is recovered," Marzari said.
|
|
Some molecular handles can even transform between a bond-broken and a bond-intact state, allowing the nanotubes to act like switches that can be turned on or off in the presence of certain substances or with a laser beam. "This direct control of conductance may lead to novel strategies for the manipulation and assembly of nanotubes in metallic interconnects, or to sensing or imaging devices that respond in real-time to optical or chemical stimuli," Marzari said.
|
|
The next step is for experiments to confirm that the approach works.
|
|
This work is supported by the MIT Institute for Soldier Nanotechnologies and the National Science Foundation.
|

