| Sep 08, 2014 |
'Pick 'n' mix' chemistry to grow cultures of bioactive molecules
|
|
(Nanowerk News) Professor Jeffrey Bode of ETH-Zürich and the Institute of Transformative Bio-Molecules (ITbM) of Nagoya University, and his co-worker have established a new strategy called "synthetic fermentation" to rapidly synthesize a large number of bioactive molecules, which can be directly screened in biological assays simply by mixing a few building blocks in aqueous media.
|
|
Using a highly selective amide-forming ligation, the reaction proceeds in high efficiency in the absence of organisms, enzymes or reagents. The fermentation products can be screened directly for biological activity without any purification. Synthetic fermentation has enabled the generation of about 6,000 unnatural peptide-like molecules from only 23 building blocks. The practicality of this approach is shown by identifying a bioactive compound for inhibiting an enzyme responsible for the replication of the hepatitis C virus.
|
|
The study, published online on September 7, 2014 in Nature Chemistry ("Synthetic fermentation of bioactive non-ribosomal peptides without organisms, enzymes or reagents") as an Advanced Online Publication, is expected to be a powerful and practical method to allow rapid generation and screening of active molecules useful for drug discovery and other industrial applications as well as for simple biological assays on site.
|
 |
| 'Pick 'n' Mix' chemistry synthesizes cultures of potent bioactive peptides, which can be directly screened in biological assays. No additional reagents are required.
|
|
Microbial fermentation is a metabolic process by which microbes synthesize complex organic substances like antibiotics from simple organic building blocks. This process is known to be one of the most successful methods to discover biologically active compounds.
|
|
"We were inspired by Nature's approach to rapidly generate complex molecules by microbial fermentation, so we considered the possibility of using synthetic organic chemistry to prepare and screen libraries of bioactive peptides, a method that we call synthetic fermentation", says Prof. Bode who led the research.
|
|
Organic synthesis often involves many time-consuming steps, the use of toxic reagents to initiate the reaction, and requires difficult purification prior to biological screening.
|
|
"Instead of using reagents or enzymes to combine simple building blocks into complex molecules, we decided to synthesize such molecules by synthetic fermentation with our chemical reactions using carefully designed precursors", says Prof. Bode.
|
|
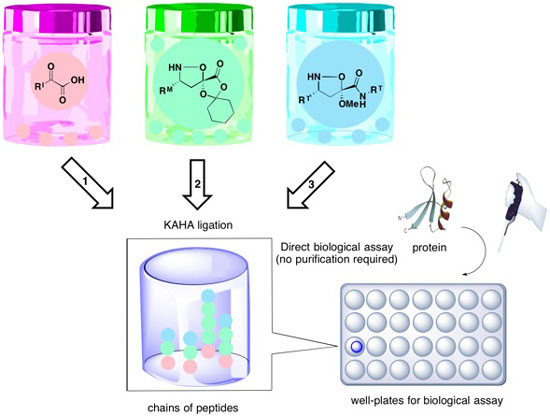
In order to accomplish this, Prof. Bode applied his unique KAHA (α-ketoacid-hydroxylamine) ligation developed by his group in 2006. KAHA ligation enables highly efficient coupling between an α-ketoacid molecule and a hydroxylamine molecule by forming an amide (N(H)C=O) bond in aqueous media without any additional reagents. Synthetic fermentation, which uses KAHA ligation chemistry, enables the construction of libraries of compounds that would be challenging to prepare by either traditional organic synthesis or by microbial fermentation.
|
|
In synthetic fermentation, an α-ketoacid initiator (I) molecule is mixed with isooxazolidine elongation monomers (M) that builds chains of peptides, which is finally stopped by adding a terminator (T) building block. A new α-ketoacid is generated each time a monomer (M) is added to the growing peptide chain, which reacts with another monomer (M) leading to peptides of varying length and sequence. After adding a terminator molecule, the peptide chain cannot grow any further. "We initially chose to work with 23 building blocks, including 6 initiators, 8 monomers, and 9 terminators, which can lead to around 30,000 compounds from all possible combinations of building blocks. In this study, the researchers have made 6,000 compounds by selected combinations.
|
|
By increasing the number of building blocks, we can dramatically expand the number of potential compounds that can be formed", explains Prof. Bode. The resulting 'culture', containing peptide chains, cyclohexanone by-products and small amounts of the terminator molecule, which is added in slight excess, can be diluted with buffer and directly subjected to biological screening.
|
|
"To the best of our knowledge, there is currently no synthetic method to prepare such large numbers of compounds from relatively few starting materials by simply mixing building blocks followed by direct biological assays", says Prof. Bode.
Bode's group demonstrates the applicability of synthetic fermentation by preparing synthetic cultures and screening them to find biologically active lead molecules for the inhibition of HCV (Hepatitis C virus) protease. HCV protease is an enzyme having a key role in the replication of the hepatitis C virus. As this enzyme lacks a well-defined pocket in its active site, this is a challenging target for finding bioactive hit molecules. "We have 'grown' our cultures for screening using various combinations of initiators (I), monomers (M) and terminators (T) and have performed assays in well plates. From the assay results, we eliminated the poorly performing moieties and rapidly deconvoluted the mixtures found in active wells to identify the combinations of components that lead to the active products", describes Prof. Bode.
|
|
|
|
Prof. Bode says, "The hardest part of this work was looking beyond the entrenched idea that we needed single, pure compounds to do the biological screening. It was only when we looked at the way Nature makes new medicines – by producing dozens of similar compounds together – that we realized we could use our chemistry to do something very similar. By taking this inspiration, we found that we could make thousands of compounds from a few building blocks in a few hours, rather than the months it would normally take."
|
|
Synthetic fermentation enables the rapid formation of a vast number of compounds from readily available building blocks that can be screened to find active hit molecules. "At ITbM, we hope to apply this approach for phenotypic assays, in order to identify small molecules that alter the characteristics of cells and organisms with biologists", says Prof. Bode. The synthetic fermentation approach may also be useful in the fields of materials science and catalysis to identify potent peptides.
|
|
"These studies show that simply by mixing compounds, anyone can create new molecules and test them in simple biological assays like anti-fungal or anti-bacterial screens. This is possible because of our very unique chemistry of the KAHA ligation developed in our group. Our idea is to provide a quick way to make bioactive molecules just by mixing the components in water. We would like to use this as a platform for chemistry that anyone can do, including scientists in other fields, high school students and farmers. Anyone could prepare libraries in a few hours with a micropippete, explore different combinations of building blocks and culture conditions along with simple assays to find novel molecules", says Prof. Bode.
|

