| Jan 19, 2022 | |
Large-scale synthesis of graphene and other 2D materials |
|
| (Nanowerk Spotlight) Since its first demonstration in 2004, the large-scale commercial production of graphene has proven difficult and costly ('large scale' usually defined as weights more than 200 mg or films larger than 200 cm2). For instance, at an estimated cost of $50 000 to $200 000 per ton for graphene powders and $45 000 to $100 000 per m2 of graphene film, industrial production methods and costs are restraining graphene utility. | |
| The quality of the graphene produced in the world today is rather poor and not optimally suited for most applications. This is possibly the main reason for the slow development of wider-scale commercial graphene applications, which usually require a customized solution in terms of graphene properties (read more on this issue in our previous Nanowerk Spotlight "Beware the fake graphene"). | |
| A review paper in Advanced Materials ("Large-Scale Syntheses of 2D Materials: Flash Joule Heating and Other Methods") summarizes the current industrial graphene synthetic and analytical methods, as well as recent academic advancements in larger-scale or sustainable synthesis of graphene. | |
| The authors, led by Prof. James M. Tour from Rice University, place specific emphasis on recent research in the use of flash Joule heating as a rapid, efficient, and scalable method to produce graphene and other 2D nanomaterials. They present reactor design, synthetic strategies, safety considerations, feedstock selection, Raman spectroscopy, and future outlooks for flash Joule heating syntheses. | |
| Concluding their review, they discuss the remaining challenges and opportunities in the larger-scale synthesis of graphene and a perspective on the broader use of flash Joule heating for larger-scale 2D materials synthesis. | |
| As illustrated in the figure below, a multitude of demonstrated and hypothesized applications of graphene exist: composites, energy storage, lubricants, coatings, gas storage and separations, flexible electronics, displays, sensors, catalysts, and water filtration are among the most published upon. | |
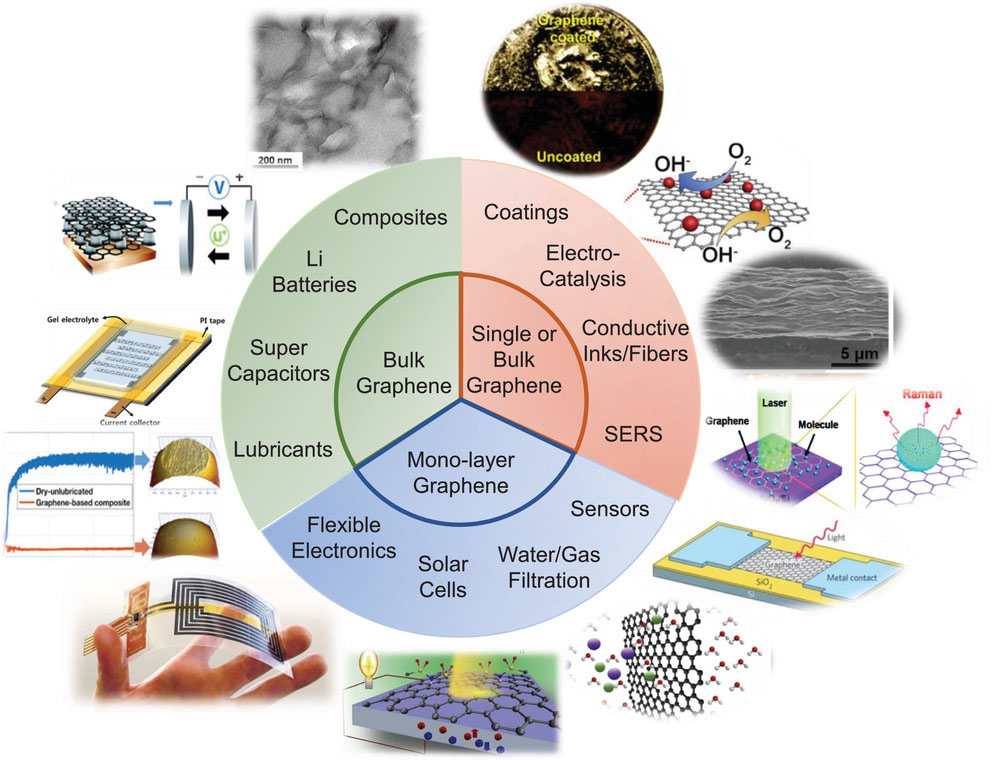 |
|
| A diagram displaying various graphene products and some of their eventual applications. (Reprinted with permission by Wiley-VCH Verlag) | |
Top-down and bottom-up production of graphene |
|
| Graphene production can be broadly classified into two different methodologies: the top-down and bottom-up strategies. The name of each method acknowledges whether the carbon atoms had their hexagonal molecular arrangement before the synthesis began. | |
| The bottom-up method (which is used almost exclusively for industrial larger-scale synthesis of graphene) employs for instance chemical vapor deposition (CVD), in which CH4, C2H4, or other simple hydrocarbons are catalytically decomposed to form graphene films on Ni, Cu, or other metal surfaces. Thus, the films are grown with individual carbon atoms from the bottom up to graphene sheets, adding one or a few atoms at a time to the graphene crystal. | |
| Conversely, the top-down method uses the exfoliation of graphite to form graphene sheets. Here, the graphene sheets are already fully formed and are merely separated physically or chemically, from the top down to the individual or a few graphene layers. | |
| Market research (Nature Nanotechnology, "The lab-to-fab journey of 2D materials") suggests that there are currently more than 800 companies producing various graphene products including graphene-enhanced composite materials, and some 300 producing graphene powders. | |
| Approximately 75% of the world's graphite supply is controlled by China, and most worldwide graphene-based patents also reside in China (Resources, Conservation and Recycling, "Dynamic material flow analysis of natural graphite in China for 2001-2018"). Total production of graphene and graphene nanoplatelets is currently estimated to reach 3800 ton per year, a minuscule amount when compared to other materials. | |
| As the authors point out, significant progress in graphene production capability has been achieved over the past decade. However, the highly variable properties of commercially available graphene would likely benefit from stronger characterization standards and closer interaction between end-users and producers, to specifically tailor the material for the desired application. | |
| The review dedicates comprehensive chapters to discuss current academic advancements in bulk top-down graphene production, larger-scale bottom-up graphene production, and sustainable larger-scale graphene production. | |
Flash Joule Heating |
|
| Flash Joule Heating (FJH) – using an electrical charge to dramatically raise a material's temperature to more than 3000 K – has been known in the literature for many decades but was more recently applied to the synthesis of graphene and other 2D materials. | |
| Making pristine graphene in a flash. | |
| In 2020, FJH was used to synthesize gram-scale quantities of graphene from a variety of feedstocks including carbon black, coal, petroleum coke, waste foods, and plastics ("Turning trash into valuable graphene in a flash"). | |
| The authors discuss the mechanism and process of FJH graphene synthesis in detail and in particular address the synthesis of flash graphene from waste materials such as plastics. | |
| For instance, FJH results in carbonization of mixed waste plastic and allows for direct conversion of waste plastic into graphene, without any intermediate pyrolysis processing. Since the heat necessary is generated in situ, there is no need for furnaces or inefficient heat transfer. | |
 |
|
| Photographs showing the process workflow to convert post-consumer high-density polyethylene (HDPE) into graphene (the conductive additive, carbon black is abbreviated CB). (Reprinted with permission by Wiley-VCH Verlag) | |
| Researchers extended this technique to produce graphene in a flash to tailor the properties of 2D dichalcogenides molybdenum disulfide and tungsten disulfide, quickly turning them into metastable metallics for electronic and optical applications (read more: "A technique to produce graphene in a flash can be used to tailor the properties of other 2D materials"). | |
Concluding considerations |
|
| The authors conclude their review with several helpful considerations for industrial or academic synthesis and use of larger-scale graphene include: | |
| "1. Stricter industrial quality control and certification of industrial graphene products are suggested. The overall poor quality of graphene on the market today delays successful application and depreciates the field of graphene research. Standardized characterization of graphene, such as presenting average Raman spectra, elemental purity information, standardized dispersibility data, and AFM or TEM derived sheet size and thickness, will increase the quality and utility of the 'black powder' on the market today. | |
| "2. Efforts to expand the use of green chemical methods are advantageous. The use of large amounts of solvent or water that is not easily recovered can lessen the attractiveness. The hope to recycle or upcycle waste materials, minimize waste streams, and minimize process mass intensity or process energy have all been demonstrated academically. Translation of these methods to industry should be investigated and implemented where possible. A wider utilization of strenuous life cycle assessments to better understand the impacts of graphene synthesis and applications can be used to identify problems and mitigate waste. | |
| "3. A better understanding of the environmental and biological impact of larger-scale graphene is needed. Fortunately, graphene is naturally occurring as crystalline aggregates of graphite and is present in coal in large amounts. Graphene may even shear from graphite in flowing river water. Further dedicated study on the retention and degradation of larger-scale graphene should be carried out at academic and industrial levels as graphene application increases. | |
| "4. Research collaborations between graphene manufacturers and commercial product users should be sought. Communication of desired characteristics and end goals can help tailor graphene products, resulting in the higher-quality implementation of graphene. Cross-industry or academic-industrial collaborations can also foster new graphene family materials applications, rather than following a one-size-fits-all use. | |
| "5. Sustained grant and commercial funding focused specifically on larger-scale graphene targets and applications should remain. Funding for improved graphene production research will in turn spawn further research due to increased accessibility of the materials needed." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
