 Researchers have developed three-dimensional component architectures based on novel, printable thermoelectric materials. This might be a milestone on the way towards use of inexpensive thermoelectric generators.
Researchers have developed three-dimensional component architectures based on novel, printable thermoelectric materials. This might be a milestone on the way towards use of inexpensive thermoelectric generators.
Feb 5th, 2021
Read more
 Inductors are set to join other electronic components in being miniaturized thanks to a quantum effect.
Inductors are set to join other electronic components in being miniaturized thanks to a quantum effect.
Feb 5th, 2021
Read more
 A 'FROC' can both transmit and reflect the same color simultaneously, a breakthrough in optical coating.
A 'FROC' can both transmit and reflect the same color simultaneously, a breakthrough in optical coating.
Feb 5th, 2021
Read more
 A new nanoimaging technique that will allow researchers to test and identify novel 2D materials in a comprehensive way at the nanoscale for the first time.
A new nanoimaging technique that will allow researchers to test and identify novel 2D materials in a comprehensive way at the nanoscale for the first time.
Feb 5th, 2021
Read more
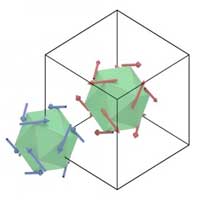 Scientists reveal peculiar surface structure in materials resembling quasicrystals with interesting implications for its magnetic properties.
Scientists reveal peculiar surface structure in materials resembling quasicrystals with interesting implications for its magnetic properties.
Feb 4th, 2021
Read more
 Scientists have succeeded in synthesizing macroscopic carbons that are similar in handling to common technical catalysts.
Scientists have succeeded in synthesizing macroscopic carbons that are similar in handling to common technical catalysts.
Feb 4th, 2021
Read more
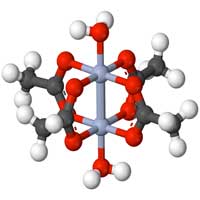 In order to produce tiny electronic memories or sensors in future, it is essential to be able to arrange individual metal atoms on an insulating layer. Scientists have now demonstrated that this is possible at room temperature.
In order to produce tiny electronic memories or sensors in future, it is essential to be able to arrange individual metal atoms on an insulating layer. Scientists have now demonstrated that this is possible at room temperature.
Feb 4th, 2021
Read more
 Two-dimensional materials can be used to create smaller, high-performance transistors traditionally made of silicon.
Two-dimensional materials can be used to create smaller, high-performance transistors traditionally made of silicon.
Feb 4th, 2021
Read more
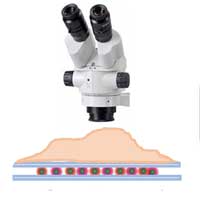 With the help of these quantum dots, researchers can use super-resolution technology to break the optical diffraction limit and fill the gap between the electron microscope (about 1 nm) and the ordinary visible optical microscope (200-250 nm).
With the help of these quantum dots, researchers can use super-resolution technology to break the optical diffraction limit and fill the gap between the electron microscope (about 1 nm) and the ordinary visible optical microscope (200-250 nm).
Feb 4th, 2021
Read more
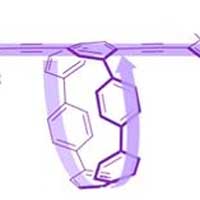 Scientists have created a new type of conducting polymer containing both linear and ring elements. The new polymers have very different electronic properties than scientists would expect if the polymers simply added the contributions from each linear and ring-shaped component.
Scientists have created a new type of conducting polymer containing both linear and ring elements. The new polymers have very different electronic properties than scientists would expect if the polymers simply added the contributions from each linear and ring-shaped component.
Feb 4th, 2021
Read more
 Researchers have shown how the structure of the MOF, and its interactions with the guest molecules, can produce a signal. With this understanding, researchers can design MOFs to be sensor materials for many important applications. These findings could lead to simpler ways to detect chemicals, including explosives.
Researchers have shown how the structure of the MOF, and its interactions with the guest molecules, can produce a signal. With this understanding, researchers can design MOFs to be sensor materials for many important applications. These findings could lead to simpler ways to detect chemicals, including explosives.
Feb 4th, 2021
Read more
 Sending tiny droplets to a tumor and having them vaporized using focused ultrasound: it could be a new way of tracing a tumor or deliver drugs locally.
Sending tiny droplets to a tumor and having them vaporized using focused ultrasound: it could be a new way of tracing a tumor or deliver drugs locally.
Feb 4th, 2021
Read more
 Researchers have come much closer to their goal of converting waste heat into electrical power at small temperature differences. They managed to increase electrical power per footprint of thermomagnetic generators based on Heusler alloy films by a factor of 3.4.
Researchers have come much closer to their goal of converting waste heat into electrical power at small temperature differences. They managed to increase electrical power per footprint of thermomagnetic generators based on Heusler alloy films by a factor of 3.4.
Feb 3rd, 2021
Read more
 The sensitivity of the device is already superior to commercially available analogs based on semiconductors and superconductors, which opens up prospects for applications of the graphene detector in wireless communications, security systems, radio astronomy, and medical diagnostics.
The sensitivity of the device is already superior to commercially available analogs based on semiconductors and superconductors, which opens up prospects for applications of the graphene detector in wireless communications, security systems, radio astronomy, and medical diagnostics.
Feb 3rd, 2021
Read more
 A surprisingly simple method improves 'drop casting' fabrication of tiled nanosheets that could be used in next-generation electronic devices. All you need is a pipette and a hotplate.
A surprisingly simple method improves 'drop casting' fabrication of tiled nanosheets that could be used in next-generation electronic devices. All you need is a pipette and a hotplate.
Feb 3rd, 2021
Read more
 Just as pressing a guitar string produces a higher pitch, sending laser light through a material can shift it to higher energies and higher frequencies. Scientists have discovered how to use this process to explore quantum materials in more detail.
Just as pressing a guitar string produces a higher pitch, sending laser light through a material can shift it to higher energies and higher frequencies. Scientists have discovered how to use this process to explore quantum materials in more detail.
Feb 2nd, 2021
Read more
 Researchers have developed three-dimensional component architectures based on novel, printable thermoelectric materials. This might be a milestone on the way towards use of inexpensive thermoelectric generators.
Researchers have developed three-dimensional component architectures based on novel, printable thermoelectric materials. This might be a milestone on the way towards use of inexpensive thermoelectric generators.















 Subscribe to our Nanotechnology News feed
Subscribe to our Nanotechnology News feed