| May 01, 2013 |
Don't cry over spilled oil - use nanosheets
|
|
(Nanowerk News) We’re all too familiar with footage of ruptured tankers or busted rigs dumping millions of litres of oil into the sea, coating shorelines and animals in crude sludge.
|
|
Many traditional methods of cleaning oil spills, such as breaking up the oil with dispersants or skimming it off the surface, are expensive, slow, and unsafe – and often don’t really work all that well anyway.
|
|
But imagine being able to quickly and easily slurp up the floating oil – and do it over and over again using the same material?
|
|
This scenario may not be far off.
|
|
In Nature Communications today ("Porous boron nitride nanosheets for effective water cleaning"), we showed how we produced, probably for the first time, nanosheets that could revolutionise oil spill clean ups and water purification.
|
|
Not only do our nanosheets absorb 33 times their weight in oil, they’re also recyclable.
|
|
It sounds like a huge win for the environment – but what makes these nanosheets so effective?
|
|
A hole lotta pores
|
|
We found that porous boron nitride nanosheets have a couple of properties that make them particularly suitable for absorbing organic (carbon-based) contaminants, such as oil or dyes.
|
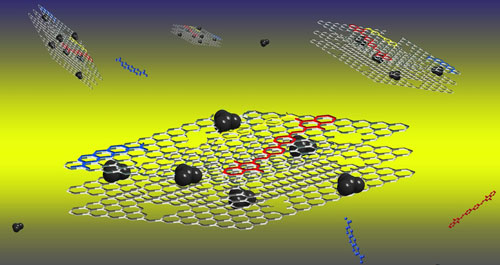 |
| A schematic of porous boron nitride nanosheet structure: layers of boron nitride, with oil molecules (black) and dyes bound to the surfaces. (Image: Weiwei Lei) (click image to enlarge)
|
|
The nanosheets are made of a few layers of boron nitride atomic planes, and these sheets have a large number of holes.
|
|
It’s these holes that increase the surface area of the nanosheets to a huge 1,425m2 a gram.
|
|
This means one gram of porous boron nitride nanosheets has the same surface area as nearly 5.5 tennis courts – so plenty of surface for absorption.
|
|
Another advantage is that the saturated boron nitride nanosheets can be cleaned for reuse by simply heating in air for two hours.
|
|
The absorbed oil is burned off, leaving the nanosheets clean and free to absorb again.
|
|
To make our porous nanosheets, boron oxide powder and guanidine hydrochloride are mixed in methane and heated at 1,100C for several hours in nitrogen gas.
|
|
The guanidine hydrochloride decomposes to release several gasses that tunnel out, resulting in the formation of holes.
|
 |
| Oil absorption, oil removal and re-use of porous BN nanosheets. (© Nature Communcations)
|
|
These pores in the nanosheets provide the surface area to absorb oils and organic solvents up to 33 times its own weight.
|
|
Cleaning capabilities
|
|
Besides high absorbent capabilities, the porous boron nitride nanosheets are lightweight and hydrophobic – so they can float on as well as repel water – and will easily separate from the water surface.
|
|
In other words, the nanosheets do not absorb water. If you sprinkle them on the surface of water, they’re easily lifted off again.
|
|
Reusing the nanosheets doesn’t have to involve burning off the oil.
|
|
Solvent extraction can be used as an alternative cleaning method to avoid generating potentially harmful gases from burning oil; washing with organic solvents such as ethanol (also known as drinking alcohol) enables recovery of the initial absorption capacity of porous boron nitride nanosheets.
|
|
Good for you, me, and the environment
|
|
This new material and related technology will have important impacts on environmental protection.
|
|
In particular, removal of contaminants from water is of significant importance for environmental and water source protection on a global scale.
|
|
Oil spillages, organic solvents and dyes discharged from textile, paper and tannery industries are primary pollutants of water sources.
|
|
Conventional methods used to solve spillages today include combustion, mechanical collection, chemical dispersants, bioremediation, and sorbent materials.
|
|
But common absorbents tend to suffer from environmental incompatibility, low separation selectivity and low absorption capacity.
|
|
Our porous boron nitride nanosheets are a simple, economic and efficient way to clean up potentially-disastrous spills from water.
|
|
And the sight of an oil-coated pelican, like the one pictured above, could well become a thing of the past.
|


