Grow and eat your own vaccines?
 Grant enables study of plants as mRNA factories.
Grant enables study of plants as mRNA factories.
Sep 16th, 2021
Read more
 Subscribe to our Biotechnology News feed
Subscribe to our Biotechnology News feed
 Grant enables study of plants as mRNA factories.
Grant enables study of plants as mRNA factories.
Sep 16th, 2021
Read more Biologists developed comprehensive software that will help scientists in a variety of industries create engineered chemicals more quickly and easily.
Biologists developed comprehensive software that will help scientists in a variety of industries create engineered chemicals more quickly and easily.
Sep 15th, 2021
Read more Studying these organoids could help researchers develop and test new treatments for pancreatic cancer, one of the deadliest types of cancer.
Studying these organoids could help researchers develop and test new treatments for pancreatic cancer, one of the deadliest types of cancer.
Sep 13th, 2021
Read more Nanoengineers have developed COVID-19 vaccine candidates that can take the heat. Their key ingredients? Viruses from plants or bacteria.
Nanoengineers have developed COVID-19 vaccine candidates that can take the heat. Their key ingredients? Viruses from plants or bacteria.
Sep 7th, 2021
Read more A hair-like protein hidden inside bacteria serves as a sort of on-off switch for nature's 'electric grid', a global web of bacteria-generated nanowires that permeates all oxygen-less soil and deep ocean beds.
A hair-like protein hidden inside bacteria serves as a sort of on-off switch for nature's 'electric grid', a global web of bacteria-generated nanowires that permeates all oxygen-less soil and deep ocean beds.
Sep 1st, 2021
Read more Researchers developed a novel artificial intelligence algorithm for clinical applications called 'scArches'. It efficiently compares patients' cells with a reference atlas of cells of healthy individuals. This enables physicians to pinpoint cells in disease and prioritize them for personalized treatment in each patient.
Researchers developed a novel artificial intelligence algorithm for clinical applications called 'scArches'. It efficiently compares patients' cells with a reference atlas of cells of healthy individuals. This enables physicians to pinpoint cells in disease and prioritize them for personalized treatment in each patient.
Aug 30th, 2021
Read more Researchers have developed a synthetic chemistry approach to polymerize proteins inside of engineered microbes. This enabled the microbes to produce the high molecular weight muscle protein, titin, which was then spun into fibers.
Researchers have developed a synthetic chemistry approach to polymerize proteins inside of engineered microbes. This enabled the microbes to produce the high molecular weight muscle protein, titin, which was then spun into fibers.
Aug 30th, 2021
Read more This novel method not only allows scientists to engineer proteins with unique functions, but also helps them to better understand how eukaryotic cell functions evolve.
This novel method not only allows scientists to engineer proteins with unique functions, but also helps them to better understand how eukaryotic cell functions evolve.
Aug 25th, 2021
Read more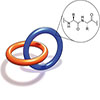 Researchers have built the world's smallest mechanically interlocked biological structure, a deceptively simple two-ring chain made from tiny strands of amino acids called peptides.
Researchers have built the world's smallest mechanically interlocked biological structure, a deceptively simple two-ring chain made from tiny strands of amino acids called peptides.
Aug 23rd, 2021
Read more Closing wounds in the digestive tract is a challenge. Researchers have now developed a polymer patch for the intestine that can be used to stably bond and seal internal injuries.
Closing wounds in the digestive tract is a challenge. Researchers have now developed a polymer patch for the intestine that can be used to stably bond and seal internal injuries.
Aug 19th, 2021
Read more The sensor reveals where in the cell hydrogen peroxide is produced and how the signaling pathways run.
The sensor reveals where in the cell hydrogen peroxide is produced and how the signaling pathways run.
Aug 19th, 2021
Read more Scientists have developed a bioprocess using engineered yeast that completely and efficiently converted plant matter consisting of acetate and xylose into high-value bioproducts.
Scientists have developed a bioprocess using engineered yeast that completely and efficiently converted plant matter consisting of acetate and xylose into high-value bioproducts.
Aug 17th, 2021
Read more Scientists are developing gene activity sensors and activators that hold unprecedented opportunities for treating diabetes, cancer, infectious diseases and genetic disorders.
Scientists are developing gene activity sensors and activators that hold unprecedented opportunities for treating diabetes, cancer, infectious diseases and genetic disorders.
Aug 16th, 2021
Read more Researchers have identified a new way to measure DNA torsional stiffness - how much resistance the helix offers when twisted - information that can potentially shed light on how cells work.
Researchers have identified a new way to measure DNA torsional stiffness - how much resistance the helix offers when twisted - information that can potentially shed light on how cells work.
Aug 12th, 2021
Read more Imagine if you could attach something to your skin without needing glue. A biosensor, a watch, a communications device, a fashion accessory - the possibilities are endless.
Imagine if you could attach something to your skin without needing glue. A biosensor, a watch, a communications device, a fashion accessory - the possibilities are endless.
Aug 10th, 2021
Read more Scientists have developed a rapid-sealing paste that can stop bleeding organs independent of clotting.
Scientists have developed a rapid-sealing paste that can stop bleeding organs independent of clotting.
Aug 10th, 2021
Read more Scientists have developed spider silk that can be produced in larger volumes without strong chemicals, making it possible to spin continuously for 75 hours, resulting in a 125 km long fibre.
Scientists have developed spider silk that can be produced in larger volumes without strong chemicals, making it possible to spin continuously for 75 hours, resulting in a 125 km long fibre.
Aug 9th, 2021
Read more An injection made of a hydrogel containing stem cells could treat damaged tissue following a heart attack.
An injection made of a hydrogel containing stem cells could treat damaged tissue following a heart attack.
Aug 6th, 2021
Read more