Showing Spotlights 161 - 168 of 315 in category All (newest first):
 Researchers in Korea have developed a novel platform for intracellular delivery of genetic material and nanoparticles, based on vertically aligned carbon nanosyringe arrays of controllable height. Stem cell research is being pursued in laboratories all over the world in the hope of achieving major medical breakthroughs. Scientists are striving to create therapies that rebuild or replace damaged cells with tissues grown from stem cells and offer hope to people suffering from cancer, diabetes, cardiovascular disease, spinal-cord injuries, and many other disorders. Nanotechnology is increasingly playing a role in how researchers think about delivering stem cell therapies into cells. Cell plasma membranes are a formidable barrier to the delivery of exogenous macromolecules in cellular engineering and labeling and cell therapy. Attempts have been made to breach this barrier, particularly using mechanical means such as microinjectors that deliver genetic material into the cell. However, there is concern about damage to the cell membrane caused by intrinsic invasiveness of the micro- or submicrosized needle used in these procedures.
Researchers in Korea have developed a novel platform for intracellular delivery of genetic material and nanoparticles, based on vertically aligned carbon nanosyringe arrays of controllable height. Stem cell research is being pursued in laboratories all over the world in the hope of achieving major medical breakthroughs. Scientists are striving to create therapies that rebuild or replace damaged cells with tissues grown from stem cells and offer hope to people suffering from cancer, diabetes, cardiovascular disease, spinal-cord injuries, and many other disorders. Nanotechnology is increasingly playing a role in how researchers think about delivering stem cell therapies into cells. Cell plasma membranes are a formidable barrier to the delivery of exogenous macromolecules in cellular engineering and labeling and cell therapy. Attempts have been made to breach this barrier, particularly using mechanical means such as microinjectors that deliver genetic material into the cell. However, there is concern about damage to the cell membrane caused by intrinsic invasiveness of the micro- or submicrosized needle used in these procedures.
Apr 14th, 2009
 Notwithstanding the tremendous amount of research that has gone into the field of carbon nanotubes, the synthesis of single-walled carbon nanotubes (SWCNTs) with controlled chirality still has not been achieved. Current production methods for carbon nanotubes result in units with different diameter, length, chirality and electronic properties, all packed together in bundles, and often blended with some amount of amorphous carbon. The separation of nanotubes according to desired properties remains a technical challenge. Especially SWCNT sorting is a challenge because the composition and chemical properties of SWCNTs of different types are very similar, making conventional separation techniques inefficient. Using the concept of cloning, scientists in China have discovered an effective method to synthesize any special indices SWCNTs.
Notwithstanding the tremendous amount of research that has gone into the field of carbon nanotubes, the synthesis of single-walled carbon nanotubes (SWCNTs) with controlled chirality still has not been achieved. Current production methods for carbon nanotubes result in units with different diameter, length, chirality and electronic properties, all packed together in bundles, and often blended with some amount of amorphous carbon. The separation of nanotubes according to desired properties remains a technical challenge. Especially SWCNT sorting is a challenge because the composition and chemical properties of SWCNTs of different types are very similar, making conventional separation techniques inefficient. Using the concept of cloning, scientists in China have discovered an effective method to synthesize any special indices SWCNTs.
Apr 3rd, 2009
 Much effort has been invested into finding a non-toxic replacement for semiconductor quantum dots (Q-dots) possessing bright fluorescence. Intrinsic toxicity of Q-dots composed of elements such as selenium, tellurium, cadmium, and lead severely hinders their in vivo applications for fluorescent imaging. Therefore many carbon nanomaterials have been considered as a replacement for Q-dots for in vivo imaging. However, it is still unclear how safe carbon nanomaterials are, and this is an obstacle for their use in medicine. Nanodiamond has been an exception among nanomaterials in many aspects, but what is important for biomedical applications is that it has shown very little or zero toxicity in all tests done so far. In addition, nanodiamond powders are already produced by detonation on a large commercial scale. This is why fluorescent nanodiamonds currently attract so much attention.
Much effort has been invested into finding a non-toxic replacement for semiconductor quantum dots (Q-dots) possessing bright fluorescence. Intrinsic toxicity of Q-dots composed of elements such as selenium, tellurium, cadmium, and lead severely hinders their in vivo applications for fluorescent imaging. Therefore many carbon nanomaterials have been considered as a replacement for Q-dots for in vivo imaging. However, it is still unclear how safe carbon nanomaterials are, and this is an obstacle for their use in medicine. Nanodiamond has been an exception among nanomaterials in many aspects, but what is important for biomedical applications is that it has shown very little or zero toxicity in all tests done so far. In addition, nanodiamond powders are already produced by detonation on a large commercial scale. This is why fluorescent nanodiamonds currently attract so much attention.
Mar 30th, 2009
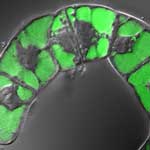 Carbon nanotubes (CNTs) have already been explored as drug carriers into mammalian cells. Compared to nanoparticles, CNTs have a larger inner volume which allows more drug molecules to be encapsulated, and this volume is more easily accessible because the end caps can be easily removed, and they have distinct inner and outer surfaces for functionalization. In addition to nanomedicine applications, plant science research focusing on investigation of plant genomics and gene function as well as improvement of crop species has become a nanotechnology frontier. To what degree nanomaterials can be employed in delivering payloads into plant cells is a subject that has not yet been explored very well although there appears to be demand from plant cell biologists to take advantage of nanomaterials.
Carbon nanotubes (CNTs) have already been explored as drug carriers into mammalian cells. Compared to nanoparticles, CNTs have a larger inner volume which allows more drug molecules to be encapsulated, and this volume is more easily accessible because the end caps can be easily removed, and they have distinct inner and outer surfaces for functionalization. In addition to nanomedicine applications, plant science research focusing on investigation of plant genomics and gene function as well as improvement of crop species has become a nanotechnology frontier. To what degree nanomaterials can be employed in delivering payloads into plant cells is a subject that has not yet been explored very well although there appears to be demand from plant cell biologists to take advantage of nanomaterials.
Mar 27th, 2009
 Carbon nanotubes possess physicochemical properties that make them an attractive possibility for nuclear waste management, especially when compared to the current tools involving activated carbon. In the environmental field, carbon nanotubes application is regarded as extremely promising for the development of novel energy-storage techniques, sensors, and sorbent materials for myriad uses including waste management. A group of European scientists want to stimulate a discussion on how the potential of carbon nanomaterials for nuclear waste mangement could be realized. They argue that the significance of the possible role of carbon nanotubes in treating and sequestering nuclear waste stems from a number of recent research results that specifically investigate the interaction between CNTs and actinides or lanthanides.
Carbon nanotubes possess physicochemical properties that make them an attractive possibility for nuclear waste management, especially when compared to the current tools involving activated carbon. In the environmental field, carbon nanotubes application is regarded as extremely promising for the development of novel energy-storage techniques, sensors, and sorbent materials for myriad uses including waste management. A group of European scientists want to stimulate a discussion on how the potential of carbon nanomaterials for nuclear waste mangement could be realized. They argue that the significance of the possible role of carbon nanotubes in treating and sequestering nuclear waste stems from a number of recent research results that specifically investigate the interaction between CNTs and actinides or lanthanides.
Mar 23rd, 2009
 Buckypaper - in which CNTs collectively behave as a random web - is characterized by its optical transparency, mechanical flexibility, high electric conductivity, uniform dimensions, tunable electronic properties, large specific surface area and smooth surface topology. All of which make this a very promising material as functional element or structural component in a wide range of applications such as optoelectronics, nanocomposites, chemical separations, biocompatible platforms, electronics, and energy conversion and storage. So far it has been difficult to simultaneously retain the intrinsic properties of individual CNTs and to have versatility in creating different shapes, both problems unavoidably resulting from post-growth fabrication processes. A research group in China has now reported a simple approach for the direct and nondestructive assembly of multi-sheeted, single-walled carbon nanotube book-like macrostructures (buckybooks) of several millimeters in thickness with good control of the nanotube diameter, the sheet packing density, and the book thickness.
Buckypaper - in which CNTs collectively behave as a random web - is characterized by its optical transparency, mechanical flexibility, high electric conductivity, uniform dimensions, tunable electronic properties, large specific surface area and smooth surface topology. All of which make this a very promising material as functional element or structural component in a wide range of applications such as optoelectronics, nanocomposites, chemical separations, biocompatible platforms, electronics, and energy conversion and storage. So far it has been difficult to simultaneously retain the intrinsic properties of individual CNTs and to have versatility in creating different shapes, both problems unavoidably resulting from post-growth fabrication processes. A research group in China has now reported a simple approach for the direct and nondestructive assembly of multi-sheeted, single-walled carbon nanotube book-like macrostructures (buckybooks) of several millimeters in thickness with good control of the nanotube diameter, the sheet packing density, and the book thickness.
Mar 9th, 2009
 Currently, all existing methods of fabricating CNT-polymer composites involve quite complicated, expensive, time-demanding processing techniques such as solution casting, melting, molding, extrusion, and in situ polymerization. In all of these techniques, nanotubes must either be incorporated into a polymer solution, molten polymer or mixed with the initial monomer before the formation of the final product. In addition, these methods can not be applied in the case of insoluble or temperature sensitive polymers, which decompose without melting. Kevlar is a well known high-strength polymer with a variety of important applications - think bullet-proof vests and car armor plating. However, Kevlar is not soluble in any common solvent and Kevlar fibers must be produced by wet spinning from sulphuric acid solutions. Researchers in Ireland have now found a way to develop a new effective post-processing technique which would allow to incorporate carbon nanotubes into already formed polymer products, such as for example Kevlar yarns.
Currently, all existing methods of fabricating CNT-polymer composites involve quite complicated, expensive, time-demanding processing techniques such as solution casting, melting, molding, extrusion, and in situ polymerization. In all of these techniques, nanotubes must either be incorporated into a polymer solution, molten polymer or mixed with the initial monomer before the formation of the final product. In addition, these methods can not be applied in the case of insoluble or temperature sensitive polymers, which decompose without melting. Kevlar is a well known high-strength polymer with a variety of important applications - think bullet-proof vests and car armor plating. However, Kevlar is not soluble in any common solvent and Kevlar fibers must be produced by wet spinning from sulphuric acid solutions. Researchers in Ireland have now found a way to develop a new effective post-processing technique which would allow to incorporate carbon nanotubes into already formed polymer products, such as for example Kevlar yarns.
Feb 25th, 2009
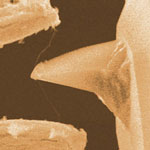 Back in 2006, researchers introduced the concept of a carbon nanotube (CNT) knife that, in theory, would work like a tight-wire cheese slicer. In the meantime, other research groups have developed similar approaches, for instance for cutting and sharpening carbon nanotubes (see for instance: Nanotechnology grinders). Now, the group that introduced the CNT nanoknife in 2006 has refined their design and demonstrated the feasibility of fabricating a nanoknife (compression-cutting tool at the nanoscale) based on an individual CNT. The researchers stretched an individual nanotube between two tungsten needles in a manner that allowed them to test the mechanical strength of assembled device. A force test on the prototype nanoknife indicated that failure was at the weld while the CNT was unaffected by the force we applied. In situ load tests on the nanoknife indicated maximum breaking force to be in micro Newton range.
Back in 2006, researchers introduced the concept of a carbon nanotube (CNT) knife that, in theory, would work like a tight-wire cheese slicer. In the meantime, other research groups have developed similar approaches, for instance for cutting and sharpening carbon nanotubes (see for instance: Nanotechnology grinders). Now, the group that introduced the CNT nanoknife in 2006 has refined their design and demonstrated the feasibility of fabricating a nanoknife (compression-cutting tool at the nanoscale) based on an individual CNT. The researchers stretched an individual nanotube between two tungsten needles in a manner that allowed them to test the mechanical strength of assembled device. A force test on the prototype nanoknife indicated that failure was at the weld while the CNT was unaffected by the force we applied. In situ load tests on the nanoknife indicated maximum breaking force to be in micro Newton range.
Feb 18th, 2009
 Researchers in Korea have developed a novel platform for intracellular delivery of genetic material and nanoparticles, based on vertically aligned carbon nanosyringe arrays of controllable height. Stem cell research is being pursued in laboratories all over the world in the hope of achieving major medical breakthroughs. Scientists are striving to create therapies that rebuild or replace damaged cells with tissues grown from stem cells and offer hope to people suffering from cancer, diabetes, cardiovascular disease, spinal-cord injuries, and many other disorders. Nanotechnology is increasingly playing a role in how researchers think about delivering stem cell therapies into cells. Cell plasma membranes are a formidable barrier to the delivery of exogenous macromolecules in cellular engineering and labeling and cell therapy. Attempts have been made to breach this barrier, particularly using mechanical means such as microinjectors that deliver genetic material into the cell. However, there is concern about damage to the cell membrane caused by intrinsic invasiveness of the micro- or submicrosized needle used in these procedures.
Researchers in Korea have developed a novel platform for intracellular delivery of genetic material and nanoparticles, based on vertically aligned carbon nanosyringe arrays of controllable height. Stem cell research is being pursued in laboratories all over the world in the hope of achieving major medical breakthroughs. Scientists are striving to create therapies that rebuild or replace damaged cells with tissues grown from stem cells and offer hope to people suffering from cancer, diabetes, cardiovascular disease, spinal-cord injuries, and many other disorders. Nanotechnology is increasingly playing a role in how researchers think about delivering stem cell therapies into cells. Cell plasma membranes are a formidable barrier to the delivery of exogenous macromolecules in cellular engineering and labeling and cell therapy. Attempts have been made to breach this barrier, particularly using mechanical means such as microinjectors that deliver genetic material into the cell. However, there is concern about damage to the cell membrane caused by intrinsic invasiveness of the micro- or submicrosized needle used in these procedures.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





