Showing Spotlights 1 - 8 of 24 in category All (newest first):
 Novel biomimetic reinforced graphene architecture overcomes mechanical fragility issues that previously hindered scalable fabrication of graphene membranes for selective transport.
Novel biomimetic reinforced graphene architecture overcomes mechanical fragility issues that previously hindered scalable fabrication of graphene membranes for selective transport.
Nov 14th, 2023
 Enormous free-energy dissipation takes place when fresh river water mixes with salty sea water. This so-called osmotic energy, which normally goes to waste, can be harvested with nanostructured membranes that are put between the two types of water. Porous materials with intrinsic nano or sub-nanochannels, such as metal-organic frameworks (MOFs) and covalent organic frameworks (COFs) are considered good candidates for nanofluidic osmotic energy harvesting. Here, researchers demonstrate an ultrathin, self-standing COF with excellent stability at different salt differential concentrations, pH values, and temperatures.
Enormous free-energy dissipation takes place when fresh river water mixes with salty sea water. This so-called osmotic energy, which normally goes to waste, can be harvested with nanostructured membranes that are put between the two types of water. Porous materials with intrinsic nano or sub-nanochannels, such as metal-organic frameworks (MOFs) and covalent organic frameworks (COFs) are considered good candidates for nanofluidic osmotic energy harvesting. Here, researchers demonstrate an ultrathin, self-standing COF with excellent stability at different salt differential concentrations, pH values, and temperatures.
Aug 2nd, 2022
 Graphene-based membranes have attracted considerable attention as promising candidates for new filtration technologies for filtering out nanoparticles, organic molecules, and even small inorganic salt ions. Putting a nature-inspired spin on the fabrication of high-performance graphene membranes for tricky oil/water separations - even in stable emulsions - researchers have demonstrated a graphene nanomesh membrane that possessed high hydrophilicity, super-oleophobicity and low oil adhesion underwater.
Graphene-based membranes have attracted considerable attention as promising candidates for new filtration technologies for filtering out nanoparticles, organic molecules, and even small inorganic salt ions. Putting a nature-inspired spin on the fabrication of high-performance graphene membranes for tricky oil/water separations - even in stable emulsions - researchers have demonstrated a graphene nanomesh membrane that possessed high hydrophilicity, super-oleophobicity and low oil adhesion underwater.
May 31st, 2022
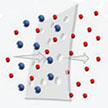 Driven by advanced nanomaterials and nanofabrication technologies, tremendous progress has been made over the last 15 years in making ultrathin membranes based on a variety of advanced nanomaterials and nanotechnologies. Further pushing membrane thickness to sub-50 nm or even atomic level is appealing to researchers, with the ultrafast permeance that may be enabled and ultimate understanding of formation and transport at the extremely low thickness that can be significantly different from their thicker counterparts.
Driven by advanced nanomaterials and nanofabrication technologies, tremendous progress has been made over the last 15 years in making ultrathin membranes based on a variety of advanced nanomaterials and nanotechnologies. Further pushing membrane thickness to sub-50 nm or even atomic level is appealing to researchers, with the ultrafast permeance that may be enabled and ultimate understanding of formation and transport at the extremely low thickness that can be significantly different from their thicker counterparts.
Apr 12th, 2022
 Researchers show that a nanoporous MoS2 membrane allows a higher water flux compared with other 2D materials such as graphene, boron nitride and phosphorene. The team dug deeply into the physical reasons behind why MoS2 performed better than other two-dimensional materials in water desalination processes. Through molecular dynamics simulation, they found out that water density near a MoS2 membrane is lower compared to that near other materials, which indicates that water molecules were more likely to transport through the membrane instead of accumulating around it.
Researchers show that a nanoporous MoS2 membrane allows a higher water flux compared with other 2D materials such as graphene, boron nitride and phosphorene. The team dug deeply into the physical reasons behind why MoS2 performed better than other two-dimensional materials in water desalination processes. Through molecular dynamics simulation, they found out that water density near a MoS2 membrane is lower compared to that near other materials, which indicates that water molecules were more likely to transport through the membrane instead of accumulating around it.
Jul 3rd, 2020
 Nanofluidic membranes based on two-dimensional materials are promising materials for next-generation water desalination and purification. For instance, pristine and chemically modified graphene oxide membranes (GOMs) effectively block organic dyes and nanoparticles as small as 9 �. However, these nanomembranes fail to exclude smaller inorganic salt ions, which would be required to extract pure potable water from unconventional water sources such as, salt water, industrial waste water, and rain water. With a novel approach called planar heterogeneous interface desalination researchers can now achieve a high salt rejection rate close to 97%.
Nanofluidic membranes based on two-dimensional materials are promising materials for next-generation water desalination and purification. For instance, pristine and chemically modified graphene oxide membranes (GOMs) effectively block organic dyes and nanoparticles as small as 9 �. However, these nanomembranes fail to exclude smaller inorganic salt ions, which would be required to extract pure potable water from unconventional water sources such as, salt water, industrial waste water, and rain water. With a novel approach called planar heterogeneous interface desalination researchers can now achieve a high salt rejection rate close to 97%.
Mar 3rd, 2020
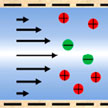 The ability of nanochannels to regulate transported substances in confined spaces is of great research interest in innovative applications, such as high-resolution sensing, filtering, and high-efficiency energy utilization. In the last area, research on nanochannels in energy-related areas continues to face challenges such as low efficiencies, complex preparation processes, and high fabrication costs. Overcoming these challenges is an important and difficult task in the field of energy conversion, energy conservation, and energy recovery.
The ability of nanochannels to regulate transported substances in confined spaces is of great research interest in innovative applications, such as high-resolution sensing, filtering, and high-efficiency energy utilization. In the last area, research on nanochannels in energy-related areas continues to face challenges such as low efficiencies, complex preparation processes, and high fabrication costs. Overcoming these challenges is an important and difficult task in the field of energy conversion, energy conservation, and energy recovery.
Apr 10th, 2018
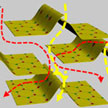 Researchers have developed a new separation membrane with 2D layered transition metal dichalcogenides (tungsten disulfide) for size-selective separation of small molecules of about 3 nm. The as-prepared WS2 membranes exhibit 5 times higher water permeance than graphene oxide membranes with similar rejection. To further improve the water permeance, they team employed ultrathin metal hydroxide nanostrands to create more fluidic channels while keeping the rejection rate of specific molecules unchanged.
Researchers have developed a new separation membrane with 2D layered transition metal dichalcogenides (tungsten disulfide) for size-selective separation of small molecules of about 3 nm. The as-prepared WS2 membranes exhibit 5 times higher water permeance than graphene oxide membranes with similar rejection. To further improve the water permeance, they team employed ultrathin metal hydroxide nanostrands to create more fluidic channels while keeping the rejection rate of specific molecules unchanged.
Jun 17th, 2014
 Novel biomimetic reinforced graphene architecture overcomes mechanical fragility issues that previously hindered scalable fabrication of graphene membranes for selective transport.
Novel biomimetic reinforced graphene architecture overcomes mechanical fragility issues that previously hindered scalable fabrication of graphene membranes for selective transport.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





