Ten things you should know about nanotechnology
 |
3) What's so special about nanotech and why is it an issue now? |
Where and how did nanotechnology start?
Chemists have dealt with naturally occurring nanoparticles all along. Think molecules or viruses. Toxicologists have dealt with nanoparticles that are the result of modern human life such as carbon particles in combustion engine exhaust. Without being aware of it, tire manufacturers used nanoparticles - carbon black - to improve the performance of tires as early as the 1920s. Medieval artists used gold nanoparticles to achieve the bright red color in church windows (gold particles in nanometer size are red, not golden). You might even say that we are surrounded by, and made of, nanostructures - atoms and molecules are nanoscale objects after all. So what is all the fuss about, all of a sudden?
The ongoing quest for miniaturization has resulted in tools such as the atomic force microscope (AFM) or the scanning tunneling microscope (STM). Combined with refined processes such as electron beam lithography, these instruments allow the deliberate manipulation and manufacture of nanostructures ("High-speed AFM enables real-time nanofabrication "). Something that wasn't possible before.
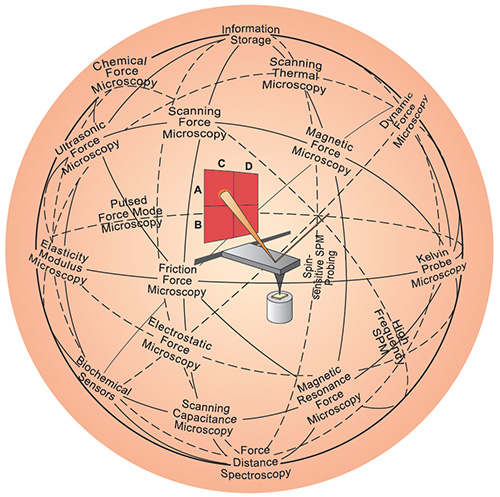
The AFM (center) has inspired a variety of other scanning probe techniques. Originally the AFM was used to image the topography of surfaces, but by modifying the tip it is possible to measure other quantities (for example, electric and magnetic properties, chemical potentials, friction and so on), and also to perform various types of spectroscopy and analysis. (Image: Christoph Gerber; copyright Nature Publishing Group)
With new tools came new concepts and it turned out that the mechanical rules that govern the nanoworld are quite different from our everyday, macroworld experience. Characterizing the extreme small forces that are at play at the nanoscale also led to further development of instruments. Only little more than a decade ago, the possibility that one could directly measure the forces acting between molecules appeared remote. The advent of nanotechnology has dramatically changed this perception. Today there are a number of tools that can be used to characterize the nanomechanics of biomolecular and cellular interactions. Examples are optical tweezers, magnetic pullers, and cantilever-based instruments like the AFM.
Engineered nanomaterials – either by way of a top-down approach (a bulk material is reduced in size to nanoscale pattern or structure) or a bottom-up approach (larger structures are built or grown atom by atom or molecule by molecule) – go beyond just a further step in miniaturization. For a discussion on nanotechnology top-down versus bottom-up see part 5 'Nanomanufacturing"of this section.
The bulk properties of materials often change dramatically when reduced to nanoscale dimensions. Starting roughly at 100 nanometers and below, materials break a size barrier below which quantization of energy for the electrons in solids becomes relevant.
Quantum Effects
The so-called quantum size effect describes the physics of electron properties in solids with great reductions in particle size. This effect does not come into play by going from macro to micro dimensions. However, it becomes dominant when the nanometer size range is reached. Quantum effects can begin to dominate the behavior of matter at the nanoscale - particularly at the lower end (single digit and low tens of nanometers) - affecting the optical, electrical and magnetic behavior of materials. Materials can be produced that are nanoscale in one dimension (for example, very thin surface coatings), in two dimensions (for example, nanowires and nanotubes) or in all three dimensions (for example, nanoparticles and quantum dots).
The causes of these drastic changes stem from the weird world of quantum physics. The bulk properties of any material are merely the average of all the quantum forces affecting all the atoms that make up the material. As you make things smaller and smaller, you eventually reach a point where the averaging no longer works and you have to deal with the specific behavior of individual atoms or molecules - behavior that can be very different to when these atoms are aggregated into a bulk material.
Materials reduced to the nanoscale can suddenly show very different properties compared to what they show on a macroscale. For instance, opaque substances become transparent (copper); inert materials become catalysts (platinum); stable materials turn combustible (aluminum); solids turn into liquids at room temperature (gold); insulators become conductors (silicon).
Surface area
Another important aspect of nanomaterials is surface area. When compared to the same mass of material in bulk form, nanoscale materials have a relatively larger surface area. This can make materials more chemically reactive (in some cases materials that are inert in bulk form are reactive when produced in their nanoscale form), and affect their strength or electrical properties.

To understand the effect of particle size on surface area, consider an American Silver Eagle coin. This silver dollar contains 31 grams of coin silver and has a total surface area of approximately 3000 square millimeters. If the same amount of coin silver were divided into tiny particles – say 10 nanometer in diameter – the total surface area of those particles would be 7000 square meters (which is equal to the size of a soccer field – or larger than the floor space of the White House, which is 5100 square meters). In other words: when the amount of coin silver contained in a silver dollar is rendered into 10 nm particles, the surface area of those particles is over 2 million times greater than the surface area of the silver dollar!
The fascination with nanotechnology stems from these unique quantum and surface phenomena that matter exhibits at the nanoscale, making possible novel applications and interesting materials.
Evolutionary vs. Revolutionary Nanotechnologies
However, there clearly is a need to differentiate between two types of nanotechnologies. One is happening right now and the other is the stuff of science fiction and way-out technology scenarios.
What we are dealing with today is evolutionary nanotechnology. The goal of evolutionary nanotechnology is to improve existing processes, materials and applications by scaling down into the nano realm and ultimately fully exploit the unique quantum and surface phenomena that matter exhibits at the nanoscale. This trend is driven by companies' ongoing quest to improve existing products by creating smaller components and better performance materials, all at a lower cost.
Take the example of the computer/electronics industry – because chip design structures have broken the 100 nanometer range, the semiconductor industry is on its way to become a nanotechnology industry.


Left: the first point contact transistor built in 1947. Right: Intel's 32 nm Nehalem chip architecture incorporates around 1.9 billion transistors in a single chip.
Think about it: The first transistors were over 1 centimeter in size, the smallest transistors today are less than 30 nanometers long – over three hundred thousand times smaller. This feat would be equivalent to shrinking the 509-meter tall Taipei 101 Tower, currently the tallest building in the world, to the size of a 1.6 millimeter tall grain of rice. So today's advanced semiconductor manufacturing is already well into the nano realm; and when we reach single-molecule transistors it will have been the end of a gradual miniaturization of electronic components.
Due to this ever continuing trend of "smaller, better, cheaper," the number of companies that are, by the same definition, "nanotechnology companies" (because they use nanoformulated food ingredients; nanoparticle coatings; nanostructured surface technology; carbon nanotube based electronics; etc.) will grow very fast and soon make up the majority of all companies across many industries – and they will have familiar names such as Kraft, L'Oreal, Toshiba, GE, BMW, Nokia or Bayer, just to name a few.
By contrast, truly revolutionary nanotechnology envisages a bottom-up approach where functional devices and entire fabrication systems are built atom by atom (just to be clear, here we are not just talking self-assembly and chemical synthesis of nanomaterials but functional machinery). Unless you resort to science fiction scenarios it will be impossible to make even educated guesses as to what that future might bring.
