Showing Spotlights 921 - 928 of 2785 in category All (newest first):
 Although developed only recently, inorganic halide perovskite quantum dot systems have exhibited comparable and even better performances than traditional quantum dots in many fields. They are expected to be applied in display and lighting technologies. Researchers now have reported an interesting cyclable surface dissolution and recrystallization phenomenon of inorganic perovskite crystals. This allows them to freely change size between nanometer and micrometer scales, and can be used to healing the defects inside perovskite films and hence improve the performances of optoelectronic devices.
Although developed only recently, inorganic halide perovskite quantum dot systems have exhibited comparable and even better performances than traditional quantum dots in many fields. They are expected to be applied in display and lighting technologies. Researchers now have reported an interesting cyclable surface dissolution and recrystallization phenomenon of inorganic perovskite crystals. This allows them to freely change size between nanometer and micrometer scales, and can be used to healing the defects inside perovskite films and hence improve the performances of optoelectronic devices.
Jul 7th, 2016
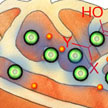 Studies show hat mitochondrial metabolism theoretically is a plausible target for cancer therapy. Mitochondria are the primary controllers of cellular suicide; however, cancer cells trick this normal cellular mechanism and evade this process leading to uncontrolled cellular growth. To trigger the suicide switch back on, new work uses a highly selective nanotechnology-based approach to deliver a widely available small molecule commonly found as a by-product of a water chlorination process.
Studies show hat mitochondrial metabolism theoretically is a plausible target for cancer therapy. Mitochondria are the primary controllers of cellular suicide; however, cancer cells trick this normal cellular mechanism and evade this process leading to uncontrolled cellular growth. To trigger the suicide switch back on, new work uses a highly selective nanotechnology-based approach to deliver a widely available small molecule commonly found as a by-product of a water chlorination process.
Jul 6th, 2016
 There is an often-asked question: 'When are we finally going to start seeing nanotechnology products on the market?' As a matter of fact, the average home is already filled with products enhanced or reliant upon nanotechnology. In fact, there are several online repositories listing the more than 2,000 commercially available products that incorporate nanotechnology. The application of nanotechnology in some areas, such as batteries, microelectronics and sunscreens is relatively well known. Let's take a virtual tour through a home to see what else we can find.
There is an often-asked question: 'When are we finally going to start seeing nanotechnology products on the market?' As a matter of fact, the average home is already filled with products enhanced or reliant upon nanotechnology. In fact, there are several online repositories listing the more than 2,000 commercially available products that incorporate nanotechnology. The application of nanotechnology in some areas, such as batteries, microelectronics and sunscreens is relatively well known. Let's take a virtual tour through a home to see what else we can find.
Jul 5th, 2016
 Researchers have demonstrated that the coupling of pristine graphene sheets on practically any polymer surface can be accomplished in mild reaction conditions and in aqueous medium. The method leaves intact the 2D planar structure of graphene preserving its original features. This novel hybrid construct enables in vivo photoacoustic signal enhancement and is a very promising step forward for an implementation of photoacoustic imaging, a powerful preclinical diagnostic tool.
Researchers have demonstrated that the coupling of pristine graphene sheets on practically any polymer surface can be accomplished in mild reaction conditions and in aqueous medium. The method leaves intact the 2D planar structure of graphene preserving its original features. This novel hybrid construct enables in vivo photoacoustic signal enhancement and is a very promising step forward for an implementation of photoacoustic imaging, a powerful preclinical diagnostic tool.
Jun 30th, 2016
 Currently in the clinic and in clinical development, there is heavy emphasis placed on understanding how drugs can be 'complementary'. That is to say, what combination of drugs will be most effective in killing tumor cells and overcome potential resistance mechanisms. In fact, adding synergistic combinations of drugs is reshaping how cancer is treated, even in the context of immunotherapy. A new study provides critical evidence that complementary drugs must be in spatial proximity to truly exert their synergistic potential.
Currently in the clinic and in clinical development, there is heavy emphasis placed on understanding how drugs can be 'complementary'. That is to say, what combination of drugs will be most effective in killing tumor cells and overcome potential resistance mechanisms. In fact, adding synergistic combinations of drugs is reshaping how cancer is treated, even in the context of immunotherapy. A new study provides critical evidence that complementary drugs must be in spatial proximity to truly exert their synergistic potential.
Jun 28th, 2016
 Education has long been recognized as an important factor for growing the fields of nanoscience and nanotechnology and solidifying and expanding their roles in the global economy. Leading researchers from the field discuss innovative learning models that are being applied at the undergraduate level in order to train future leaders at the interface of engineering and management. They have a set of five recommendations to improve the current situation.
Education has long been recognized as an important factor for growing the fields of nanoscience and nanotechnology and solidifying and expanding their roles in the global economy. Leading researchers from the field discuss innovative learning models that are being applied at the undergraduate level in order to train future leaders at the interface of engineering and management. They have a set of five recommendations to improve the current situation.
Jun 21st, 2016
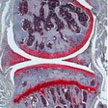 Researchers have shown, using a mouse model of osteoarthritis, that curcumin loaded nanoparticles topically applied to arthritic knees stopped the progression of the disease and eliminated associated pain by locally delivering curcumin to the fat pad associated with the knee cap. The study also demonstrated several osteoarthritis relevant inflammatory pathways were suppressed by curcumin, including those that result in the production of proteins that destroy cartilage.
Researchers have shown, using a mouse model of osteoarthritis, that curcumin loaded nanoparticles topically applied to arthritic knees stopped the progression of the disease and eliminated associated pain by locally delivering curcumin to the fat pad associated with the knee cap. The study also demonstrated several osteoarthritis relevant inflammatory pathways were suppressed by curcumin, including those that result in the production of proteins that destroy cartilage.
Jun 17th, 2016
 First there was graphene, an atomically thin film of carbon atoms with record-crushing properties. Then thousands of other atomically thin materials entered the scene which can be layered to create new hybrid supermaterials - so called van der Waals heterostructures. Until now, only a few leading groups have been able assemble these materials with sufficiently high quality. With the 'hot pickup' stacking method, researchers aim to make atomic scale nano-assembly faster and easier than ever before.
First there was graphene, an atomically thin film of carbon atoms with record-crushing properties. Then thousands of other atomically thin materials entered the scene which can be layered to create new hybrid supermaterials - so called van der Waals heterostructures. Until now, only a few leading groups have been able assemble these materials with sufficiently high quality. With the 'hot pickup' stacking method, researchers aim to make atomic scale nano-assembly faster and easier than ever before.
Jun 16th, 2016
 Although developed only recently, inorganic halide perovskite quantum dot systems have exhibited comparable and even better performances than traditional quantum dots in many fields. They are expected to be applied in display and lighting technologies. Researchers now have reported an interesting cyclable surface dissolution and recrystallization phenomenon of inorganic perovskite crystals. This allows them to freely change size between nanometer and micrometer scales, and can be used to healing the defects inside perovskite films and hence improve the performances of optoelectronic devices.
Although developed only recently, inorganic halide perovskite quantum dot systems have exhibited comparable and even better performances than traditional quantum dots in many fields. They are expected to be applied in display and lighting technologies. Researchers now have reported an interesting cyclable surface dissolution and recrystallization phenomenon of inorganic perovskite crystals. This allows them to freely change size between nanometer and micrometer scales, and can be used to healing the defects inside perovskite films and hence improve the performances of optoelectronic devices.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





