Showing Spotlights 1649 - 1656 of 2794 in category All (newest first):
 Conventional metal patterning in the electronics industry is done by a photo-resist patterning with a photo-mask and metal evaporation in a vacuum chamber. This technology require expensive vacuum conditions, high processing temperatures, many steps, and toxic chemicals to fabricate one layer of a metal pattern. Furthermore, it is almost impossible to change the design of the expensive photomask once it is fabricated. Researchers have now developed a high-resolution metal nano-patterning technique by combining solution deposited metal nanoparticles and a femtosecond laser without using conventional vacuum deposition or photo-masks. With this method, the time and cost associated with high-resolution metal patterning can be reduced dramatically without using time-consuming vacuum processes and the pattern design can be easily changed in a digital manner without using expensive photo-masks.
Conventional metal patterning in the electronics industry is done by a photo-resist patterning with a photo-mask and metal evaporation in a vacuum chamber. This technology require expensive vacuum conditions, high processing temperatures, many steps, and toxic chemicals to fabricate one layer of a metal pattern. Furthermore, it is almost impossible to change the design of the expensive photomask once it is fabricated. Researchers have now developed a high-resolution metal nano-patterning technique by combining solution deposited metal nanoparticles and a femtosecond laser without using conventional vacuum deposition or photo-masks. With this method, the time and cost associated with high-resolution metal patterning can be reduced dramatically without using time-consuming vacuum processes and the pattern design can be easily changed in a digital manner without using expensive photo-masks.
Jun 22nd, 2011
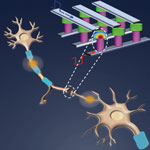 Researchers demonstrate a new single element nanoscale device, based on the successfully commercialized phase change material technology, emulating the functionality and the plasticity of biological synapses. In the nervous system, a synapse is the junction between two neurons, enabling the transmission of electric messages from one neuron to another and the adaptation of the message as a function of the nature of the incoming signal - something that is called plasticity. Synapses dominate the architecture of the brain and are responsible for massive parallelism, structural plasticity, and robustness of the brain. Therefore, a compact nanoelectronic device emulating the functions and plasticity of biological synapses will be the most important building block of brain-inspired computational systems.
Researchers demonstrate a new single element nanoscale device, based on the successfully commercialized phase change material technology, emulating the functionality and the plasticity of biological synapses. In the nervous system, a synapse is the junction between two neurons, enabling the transmission of electric messages from one neuron to another and the adaptation of the message as a function of the nature of the incoming signal - something that is called plasticity. Synapses dominate the architecture of the brain and are responsible for massive parallelism, structural plasticity, and robustness of the brain. Therefore, a compact nanoelectronic device emulating the functions and plasticity of biological synapses will be the most important building block of brain-inspired computational systems.
Jun 21st, 2011
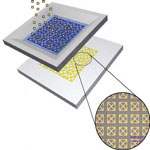 Paper has emerged as a focus area for researchers developing innovative techniques for printed basic electronics components. Another area where paper could lead to low-cost innovative devices and applications is lab-on-a-chip technology. Currently, these microfluidic devices are fairly expensive due to their lithography-based fabrication process with channels patterned in glass or plastic and tiny pumps and valves directing the flow of fluids. Inexpensive paper-based sensing kits already play an important role in ready-to-use diagnostics. Researchers have even managed to create inexpensive microfluidic platforms on hydrophobic paper with laser treatments. In a further advance, scientists have now fabricated a paper-based metamaterial device which can be potentially utilized for quantitative analysis in biochemical sensing applications.
Paper has emerged as a focus area for researchers developing innovative techniques for printed basic electronics components. Another area where paper could lead to low-cost innovative devices and applications is lab-on-a-chip technology. Currently, these microfluidic devices are fairly expensive due to their lithography-based fabrication process with channels patterned in glass or plastic and tiny pumps and valves directing the flow of fluids. Inexpensive paper-based sensing kits already play an important role in ready-to-use diagnostics. Researchers have even managed to create inexpensive microfluidic platforms on hydrophobic paper with laser treatments. In a further advance, scientists have now fabricated a paper-based metamaterial device which can be potentially utilized for quantitative analysis in biochemical sensing applications.
Jun 20th, 2011
 Radiation damage to materials is a major issue for builders of nuclear power plants as well as spacecraft engineers. The former have to worry about material failure due to the destructive radiation created within the reactor; the later are concerned about the exposure to space radiation of both materials and humans during long-term space missions. There is a consensus that interface engineering is the way to go to to improve resistance to extreme conditions in general and to reach radiation tolerance in particular - interfaces are places where defects created by energetic collision in solids can annihilate each other and thus render the material immune to irradiation. All kind of interfaces are being explored, in particular those formed by the contact between two different solid state phases.
Radiation damage to materials is a major issue for builders of nuclear power plants as well as spacecraft engineers. The former have to worry about material failure due to the destructive radiation created within the reactor; the later are concerned about the exposure to space radiation of both materials and humans during long-term space missions. There is a consensus that interface engineering is the way to go to to improve resistance to extreme conditions in general and to reach radiation tolerance in particular - interfaces are places where defects created by energetic collision in solids can annihilate each other and thus render the material immune to irradiation. All kind of interfaces are being explored, in particular those formed by the contact between two different solid state phases.
Jun 15th, 2011
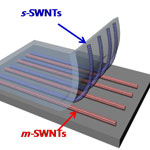 Current production methods for carbon nanotubes (CNTs) result in units with different diameter, length, chirality and electronic properties, all packed together in bundles, and often blended with some amount of amorphous carbon. Often, these mixtures are of little practical use since many advanced applications, especially for nanoelectronics, are sensitively dependent on the structures. Separation of nanotubes according to desired properties is still proving to be a challenging task, especially single-walled carbon nanotube (SWCNT) sorting. The composition of SWCNTs of different types is very similar, but the chemical properties are not. Conventional separation techniques are efficient in separation of carbon nanotubes, but there are many drawbacks. Researchers in Asia have now developed a simple way to realize the separation of single-walled carbon nanotubes via the interaction difference between chemicals and carbon nanotubes.
Current production methods for carbon nanotubes (CNTs) result in units with different diameter, length, chirality and electronic properties, all packed together in bundles, and often blended with some amount of amorphous carbon. Often, these mixtures are of little practical use since many advanced applications, especially for nanoelectronics, are sensitively dependent on the structures. Separation of nanotubes according to desired properties is still proving to be a challenging task, especially single-walled carbon nanotube (SWCNT) sorting. The composition of SWCNTs of different types is very similar, but the chemical properties are not. Conventional separation techniques are efficient in separation of carbon nanotubes, but there are many drawbacks. Researchers in Asia have now developed a simple way to realize the separation of single-walled carbon nanotubes via the interaction difference between chemicals and carbon nanotubes.
Jun 14th, 2011
 Graphene has two distinct types of edges produced when it is cut - armchair type or zigzag type - which correspond to the two crystal axis of graphene. These edge types have distinct electronic, magnetic, and chemical properties, and being able to pattern graphene along particular crystallographic directions to leave edges consisting of a single chirality is crucial for the fabrication of graphene nanoribbon and nanoelectronics devices. A widely discussed method for the patterning of graphene is the channelling of graphite by metal nanoparticles in oxidizing or reducing environments. Researchers now report the live nanoscale observation of this channelling process by silver nanoparticles.
Graphene has two distinct types of edges produced when it is cut - armchair type or zigzag type - which correspond to the two crystal axis of graphene. These edge types have distinct electronic, magnetic, and chemical properties, and being able to pattern graphene along particular crystallographic directions to leave edges consisting of a single chirality is crucial for the fabrication of graphene nanoribbon and nanoelectronics devices. A widely discussed method for the patterning of graphene is the channelling of graphite by metal nanoparticles in oxidizing or reducing environments. Researchers now report the live nanoscale observation of this channelling process by silver nanoparticles.
Jun 13th, 2011
 Apart from making graphene, graphene oxide itself itself is a fascinating material that has many intriguing properties. Researchers have now developed a graphene-based conductive glue that can function as a metal-free solder for creating mechanical and electrical connections in organic optoelectronic devices. As a proof-of-concept, they fabricated polymer tandem solar cells - multi-junction photovoltaic devices, in which two sub-cells are stacked to achieve higher overall solar absorption - by a direct 'gluing' process. The water-based sticky interconnect and the associated adhesive lamination process could transform the serial layer-by-layer fabrication of tandem devices into a parallel mode, in which the subcells can be independently fabricated and adjusted to balance their photocurrents for achieving high efficiency.
Apart from making graphene, graphene oxide itself itself is a fascinating material that has many intriguing properties. Researchers have now developed a graphene-based conductive glue that can function as a metal-free solder for creating mechanical and electrical connections in organic optoelectronic devices. As a proof-of-concept, they fabricated polymer tandem solar cells - multi-junction photovoltaic devices, in which two sub-cells are stacked to achieve higher overall solar absorption - by a direct 'gluing' process. The water-based sticky interconnect and the associated adhesive lamination process could transform the serial layer-by-layer fabrication of tandem devices into a parallel mode, in which the subcells can be independently fabricated and adjusted to balance their photocurrents for achieving high efficiency.
Jun 10th, 2011
 The future of your shirts, socks and gloves will be electronic. In years to come, wearable electronics will look nothing like even your smallest iPod or mobile phone today. Not only will such devices be embedded on textile substrates, but an electronics device or system could become the fabric itself. Here is some recent work that demonstrates the kind of issues scientists are working on today and that will help improve the performance of electronic textile structures. Using atomic layer deposition (ALD), researchers have grown coatings of inorganic materials on the surface of textiles like woven cotton and nonwoven polypropylene. By fabricating an all-fiber capacitor, they show that their coated materials are sufficiently conductive to perform in simple device architectures.
The future of your shirts, socks and gloves will be electronic. In years to come, wearable electronics will look nothing like even your smallest iPod or mobile phone today. Not only will such devices be embedded on textile substrates, but an electronics device or system could become the fabric itself. Here is some recent work that demonstrates the kind of issues scientists are working on today and that will help improve the performance of electronic textile structures. Using atomic layer deposition (ALD), researchers have grown coatings of inorganic materials on the surface of textiles like woven cotton and nonwoven polypropylene. By fabricating an all-fiber capacitor, they show that their coated materials are sufficiently conductive to perform in simple device architectures.
Jun 9th, 2011
 Conventional metal patterning in the electronics industry is done by a photo-resist patterning with a photo-mask and metal evaporation in a vacuum chamber. This technology require expensive vacuum conditions, high processing temperatures, many steps, and toxic chemicals to fabricate one layer of a metal pattern. Furthermore, it is almost impossible to change the design of the expensive photomask once it is fabricated. Researchers have now developed a high-resolution metal nano-patterning technique by combining solution deposited metal nanoparticles and a femtosecond laser without using conventional vacuum deposition or photo-masks. With this method, the time and cost associated with high-resolution metal patterning can be reduced dramatically without using time-consuming vacuum processes and the pattern design can be easily changed in a digital manner without using expensive photo-masks.
Conventional metal patterning in the electronics industry is done by a photo-resist patterning with a photo-mask and metal evaporation in a vacuum chamber. This technology require expensive vacuum conditions, high processing temperatures, many steps, and toxic chemicals to fabricate one layer of a metal pattern. Furthermore, it is almost impossible to change the design of the expensive photomask once it is fabricated. Researchers have now developed a high-resolution metal nano-patterning technique by combining solution deposited metal nanoparticles and a femtosecond laser without using conventional vacuum deposition or photo-masks. With this method, the time and cost associated with high-resolution metal patterning can be reduced dramatically without using time-consuming vacuum processes and the pattern design can be easily changed in a digital manner without using expensive photo-masks.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





