Showing Spotlights 1657 - 1664 of 2790 in category All (newest first):
 The small particles in volcanic ash can damage jet aircraft engines. During jet engine operation, large amounts of air are sucked in. If this air contains ash particles, especially glass-rich silicate ash, they will melt in the jet turbine's operating temperature of 1400-1500C and subsequently fuse to the turbine blades. This will drive the finely tuned blades out-of-balance and could stall the engine. In today's jet engines, a thermal barrier coating (TBC) insulates metallic engine parts from the heat generated by the combustion process. If ingested ash melts onto this coating it will penetrate it and, upon cooling, the molten ash forms a brittle glass that flakes off, taking the coating with it. A team of researchers have now examined a new class of ceramic TBC that could offer jet engines special protection against volcanic ash damage in the future.
The small particles in volcanic ash can damage jet aircraft engines. During jet engine operation, large amounts of air are sucked in. If this air contains ash particles, especially glass-rich silicate ash, they will melt in the jet turbine's operating temperature of 1400-1500C and subsequently fuse to the turbine blades. This will drive the finely tuned blades out-of-balance and could stall the engine. In today's jet engines, a thermal barrier coating (TBC) insulates metallic engine parts from the heat generated by the combustion process. If ingested ash melts onto this coating it will penetrate it and, upon cooling, the molten ash forms a brittle glass that flakes off, taking the coating with it. A team of researchers have now examined a new class of ceramic TBC that could offer jet engines special protection against volcanic ash damage in the future.
May 31st, 2011
 Groundbreaking research has shown a quantum atom has been tracked inside a living human cell and may lead to improvements in the testing and development of new drugs. Researchers conducted studies that confirm that non-invasive quantum measurement is possible on nanodiamonds containing a single nitrogen-vacancy (NV) spin moving within living cells. Studying the quantum properties of a single NV defect within a diamond nanocrystal, the researchers demonstrate a new technique which enables the orientation of a nanoparticle to be determined to an accuracy of less than one degree in an acquisition time of 89 milliseconds. This new technique offers biologists an extra degree of freedom when studying the translational motion of nanoparticles. Monitoring the coherence from a single electron spin paves the ways for nanoscale bio-magnetometry allowing scientists to probe changes in the cell's electromagnetic environment.
Groundbreaking research has shown a quantum atom has been tracked inside a living human cell and may lead to improvements in the testing and development of new drugs. Researchers conducted studies that confirm that non-invasive quantum measurement is possible on nanodiamonds containing a single nitrogen-vacancy (NV) spin moving within living cells. Studying the quantum properties of a single NV defect within a diamond nanocrystal, the researchers demonstrate a new technique which enables the orientation of a nanoparticle to be determined to an accuracy of less than one degree in an acquisition time of 89 milliseconds. This new technique offers biologists an extra degree of freedom when studying the translational motion of nanoparticles. Monitoring the coherence from a single electron spin paves the ways for nanoscale bio-magnetometry allowing scientists to probe changes in the cell's electromagnetic environment.
May 26th, 2011
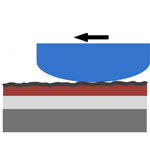 Interaction forces between contact surfaces, such as the adhesion and friction forces, are crucial in many applications at the nanoscale because of the high surface-to-volume ratio of nanomaterials and nanodevices. Over the last decade, various solid lubricant materials, micro/nano patterns, and surface treatment processes have been developed for efficient operation and extended lifetime in MEMS/NEMS applications, and for various fabrication processes such as nanoimprint lithography and transfer printing. From a practical viewpoint, large-area CVD-graphene holds the greatest promise as a solid lubricant because of its excellent scalability and transferability. So far, the tribological characteristics of CVD-graphene at the micro- and nanoscale have not been reported yet. Researchers have now demonstrated the feasibility of using large-area CVD-graphene as a solid lubricant.
Interaction forces between contact surfaces, such as the adhesion and friction forces, are crucial in many applications at the nanoscale because of the high surface-to-volume ratio of nanomaterials and nanodevices. Over the last decade, various solid lubricant materials, micro/nano patterns, and surface treatment processes have been developed for efficient operation and extended lifetime in MEMS/NEMS applications, and for various fabrication processes such as nanoimprint lithography and transfer printing. From a practical viewpoint, large-area CVD-graphene holds the greatest promise as a solid lubricant because of its excellent scalability and transferability. So far, the tribological characteristics of CVD-graphene at the micro- and nanoscale have not been reported yet. Researchers have now demonstrated the feasibility of using large-area CVD-graphene as a solid lubricant.
May 25th, 2011
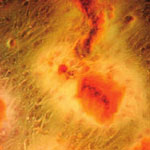 It is widely believed that stem cell therapies have the potential to revolutionize the treatment of human diseases. Key to the success of such therapies are two crucial properties: the ability of stem cells to develop into any specialized cell type depending on the specific need at hand; and the ability to guide the fate of the stem cells by various external factors. Researchers in Asia have now demonstrated that graphene provides a promising biocompatible scaffold that does not hamper the proliferation of human mesenchymal stem cells and accelerates their specific differentiation into bone cells. The differentiation rate is comparable to the one achieved with common growth factors, demonstrating graphene's potential for stem cell research.
It is widely believed that stem cell therapies have the potential to revolutionize the treatment of human diseases. Key to the success of such therapies are two crucial properties: the ability of stem cells to develop into any specialized cell type depending on the specific need at hand; and the ability to guide the fate of the stem cells by various external factors. Researchers in Asia have now demonstrated that graphene provides a promising biocompatible scaffold that does not hamper the proliferation of human mesenchymal stem cells and accelerates their specific differentiation into bone cells. The differentiation rate is comparable to the one achieved with common growth factors, demonstrating graphene's potential for stem cell research.
May 24th, 2011
 Germanium was the basic material of first-generation transistors in the late 1940s and early 1950s before it was replaced by silicon (the first silicon transistor was produced by Texas Instruments in 1954). Using germanium instead of silicon as transistor material would enable faster chips containing smaller transistors because higher switching speeds than in silicon could be achieved using germanium. A novel fabrication route demonstrated by a research team in Australia, using a combination of scanning tunneling microscope (STM) lithography and high-quality crystal growth, opens up an entire new area where quantum behavior of highly confined electrons in germanium can be studied for the first time.
Germanium was the basic material of first-generation transistors in the late 1940s and early 1950s before it was replaced by silicon (the first silicon transistor was produced by Texas Instruments in 1954). Using germanium instead of silicon as transistor material would enable faster chips containing smaller transistors because higher switching speeds than in silicon could be achieved using germanium. A novel fabrication route demonstrated by a research team in Australia, using a combination of scanning tunneling microscope (STM) lithography and high-quality crystal growth, opens up an entire new area where quantum behavior of highly confined electrons in germanium can be studied for the first time.
May 20th, 2011
 Microfluidic chips have proven to be a breakthrough analytical technique that has rendered analysis of proteins a medical routine. The sensitivity limits of immunoassays have been enhanced to picomolar concentrations using monoclonal antibodies, new labeling techniques, and devices for signal transduction and acquisition. It is now possible to routinely determine levels of hormones, cancer markers, response to infection with bacteria and viruses, monitor the evolution of a disease and test for medication levels. In a new review article, researchers argue that the most promising opportunities of microfluidics for diagnostics reside in point-of-care applications because a number of unmet needs can be fulfilled by microfluidic devices due to their portability, short sample processing time, and flexibility.
Microfluidic chips have proven to be a breakthrough analytical technique that has rendered analysis of proteins a medical routine. The sensitivity limits of immunoassays have been enhanced to picomolar concentrations using monoclonal antibodies, new labeling techniques, and devices for signal transduction and acquisition. It is now possible to routinely determine levels of hormones, cancer markers, response to infection with bacteria and viruses, monitor the evolution of a disease and test for medication levels. In a new review article, researchers argue that the most promising opportunities of microfluidics for diagnostics reside in point-of-care applications because a number of unmet needs can be fulfilled by microfluidic devices due to their portability, short sample processing time, and flexibility.
May 17th, 2011
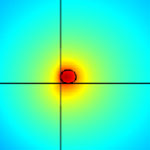 A memristor is a two-terminal electronic device whose conductance can be precisely modulated by charge or flux through it. It has the special property that its resistance can be programmed (resistor) and subsequently remains stored (memory). It is thought memristors - with the ability to 'remember' the total electronic charge that passes through them - will be of greatest benefit when they can act like synapses within electronic circuits, mimicking the complex network of neurons present in the brain, enabling our own ability to perceive, think and remember. Researchers have now analyzed in unprecedented detail the physical and chemical properties of an electronic device that computer engineers hope will transform computing.
A memristor is a two-terminal electronic device whose conductance can be precisely modulated by charge or flux through it. It has the special property that its resistance can be programmed (resistor) and subsequently remains stored (memory). It is thought memristors - with the ability to 'remember' the total electronic charge that passes through them - will be of greatest benefit when they can act like synapses within electronic circuits, mimicking the complex network of neurons present in the brain, enabling our own ability to perceive, think and remember. Researchers have now analyzed in unprecedented detail the physical and chemical properties of an electronic device that computer engineers hope will transform computing.
May 16th, 2011
 Several manufacturers are incorporating nano-sized particles of silver into, among other things, garments like socks and shirts to kill bacteria that cause odor. But does the silver stay in the socks or T-shirts? And what happens to it if it washes out? Also, what is the climate footprint of producing the required nanosilver? To answer these questions, a group of researchers have performed a cradle-to-grave life cycle assessment to compare nanosilver T-shirts with conventional T-shirts with and without biocidal treatment. For their assessment, the team used conventional T-shirts treated with triclosan, a commonly applied biocide to prevent textiles from emitting undesirable odors. The results show significant differences in environmental burdens between nanoparticle production technologies.
Several manufacturers are incorporating nano-sized particles of silver into, among other things, garments like socks and shirts to kill bacteria that cause odor. But does the silver stay in the socks or T-shirts? And what happens to it if it washes out? Also, what is the climate footprint of producing the required nanosilver? To answer these questions, a group of researchers have performed a cradle-to-grave life cycle assessment to compare nanosilver T-shirts with conventional T-shirts with and without biocidal treatment. For their assessment, the team used conventional T-shirts treated with triclosan, a commonly applied biocide to prevent textiles from emitting undesirable odors. The results show significant differences in environmental burdens between nanoparticle production technologies.
May 13th, 2011
 The small particles in volcanic ash can damage jet aircraft engines. During jet engine operation, large amounts of air are sucked in. If this air contains ash particles, especially glass-rich silicate ash, they will melt in the jet turbine's operating temperature of 1400-1500C and subsequently fuse to the turbine blades. This will drive the finely tuned blades out-of-balance and could stall the engine. In today's jet engines, a thermal barrier coating (TBC) insulates metallic engine parts from the heat generated by the combustion process. If ingested ash melts onto this coating it will penetrate it and, upon cooling, the molten ash forms a brittle glass that flakes off, taking the coating with it. A team of researchers have now examined a new class of ceramic TBC that could offer jet engines special protection against volcanic ash damage in the future.
The small particles in volcanic ash can damage jet aircraft engines. During jet engine operation, large amounts of air are sucked in. If this air contains ash particles, especially glass-rich silicate ash, they will melt in the jet turbine's operating temperature of 1400-1500C and subsequently fuse to the turbine blades. This will drive the finely tuned blades out-of-balance and could stall the engine. In today's jet engines, a thermal barrier coating (TBC) insulates metallic engine parts from the heat generated by the combustion process. If ingested ash melts onto this coating it will penetrate it and, upon cooling, the molten ash forms a brittle glass that flakes off, taking the coating with it. A team of researchers have now examined a new class of ceramic TBC that could offer jet engines special protection against volcanic ash damage in the future.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





