Showing Spotlights 65 - 72 of 204 in category All (newest first):
 Neural interfaces establish direct communication between the central nervous system (CNS) and a sovereign, man-made digital system. This technology is perhaps the most important advance in the study and treatment of the brain is the development of the neural interface. Nanotechnology fabrication methods can overcome the limitations of existing interface devices by producing electrodes with an extremely high surface to volume ratio, i.e., more probe units within the same volume, resulting in unprecedented specificity.
Neural interfaces establish direct communication between the central nervous system (CNS) and a sovereign, man-made digital system. This technology is perhaps the most important advance in the study and treatment of the brain is the development of the neural interface. Nanotechnology fabrication methods can overcome the limitations of existing interface devices by producing electrodes with an extremely high surface to volume ratio, i.e., more probe units within the same volume, resulting in unprecedented specificity.
Jul 26th, 2017
 In addition to economic, social and political measures, new technologies can provide tools for poverty reduction. Many people in developing countries don't just lack money. Especially in remote regions, many also lack access to electricity, clean drinking water and basic sanitation, cooking facilities, healthcare, adequate housing, etc. The innovative solutions for developing countries supported by the use of nanotechnologies mainly target five areas: water; medicine; agriculture; food; and energy.
In addition to economic, social and political measures, new technologies can provide tools for poverty reduction. Many people in developing countries don't just lack money. Especially in remote regions, many also lack access to electricity, clean drinking water and basic sanitation, cooking facilities, healthcare, adequate housing, etc. The innovative solutions for developing countries supported by the use of nanotechnologies mainly target five areas: water; medicine; agriculture; food; and energy.
Jul 13th, 2017
 New work demonstrates that one of the most important security primitives, i.e. a true random number generator (TRNG), can be realized within the rigorous constraints required for future Internet-of-Things electronics. The solution-processability of semiconducting single-walled carbon nanotubes allows to meet these strict constraints by simultaneously enabling small-scale, low cost fabrication of low-power, ultra-thin, printable, and mechanically flexible security devices. This presents a significant milestone in enabling higher level cryptographic solutions using scalable solution processing.
New work demonstrates that one of the most important security primitives, i.e. a true random number generator (TRNG), can be realized within the rigorous constraints required for future Internet-of-Things electronics. The solution-processability of semiconducting single-walled carbon nanotubes allows to meet these strict constraints by simultaneously enabling small-scale, low cost fabrication of low-power, ultra-thin, printable, and mechanically flexible security devices. This presents a significant milestone in enabling higher level cryptographic solutions using scalable solution processing.
Jul 12th, 2017
 The robustness, accuracy, and validity of an atomistic simulations hinge on the appropriate choice of force fields. Force fields are key for modeling the interaction between atoms of a matter under study, and the challenge is to have an accurate force field working for any specific material at any desired temperature. To serve this objective and make a benchmark as well as a shortcut for users to find their best force fields, scientists have examined a number of force fields for materials that are popular in micro- and nanotechnologies.
The robustness, accuracy, and validity of an atomistic simulations hinge on the appropriate choice of force fields. Force fields are key for modeling the interaction between atoms of a matter under study, and the challenge is to have an accurate force field working for any specific material at any desired temperature. To serve this objective and make a benchmark as well as a shortcut for users to find their best force fields, scientists have examined a number of force fields for materials that are popular in micro- and nanotechnologies.
Jun 12th, 2017
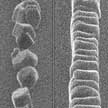 In recent years, researchers working on de-icing and anti-icing strategies have been inspired by biology and nanotechnology to develop nanocoatings and other nanostructured surfaces. Researchers now have demonstrated the ability to spatially control frost nucleation (ice formation from water vapor) and to manipulate ice crystal growth kinetics. This ice nucleation control and the confinement of ice crystal growth direction through manipulating roughness scale have not been reported before.
In recent years, researchers working on de-icing and anti-icing strategies have been inspired by biology and nanotechnology to develop nanocoatings and other nanostructured surfaces. Researchers now have demonstrated the ability to spatially control frost nucleation (ice formation from water vapor) and to manipulate ice crystal growth kinetics. This ice nucleation control and the confinement of ice crystal growth direction through manipulating roughness scale have not been reported before.
Mar 17th, 2017
 Carbon nanotubes (CNTs), by possessing a uniquely large disparity among its intertube and intratube interaction strengths, have been established as ultralow friction nanostructures and are serving as testbeds for tuning frictional response. In new woirk, researchers now have revealed the phononic origins of friction in CNT oscillators. This work, for the first time, provides a precise connection between individual phonon mode scattering and friction force.
Carbon nanotubes (CNTs), by possessing a uniquely large disparity among its intertube and intratube interaction strengths, have been established as ultralow friction nanostructures and are serving as testbeds for tuning frictional response. In new woirk, researchers now have revealed the phononic origins of friction in CNT oscillators. This work, for the first time, provides a precise connection between individual phonon mode scattering and friction force.
Mar 6th, 2017
 Here are the 10 most popular Nanowerk Nanotechnology Spotlight articles of 2016. This year, the list includes nanotechnology in textiles; nanotechnology for next-generation inkjet color printing; graphene-based smart contact lens works as self-powered biosensor; nanotechnology's tiny steps toward atomic-scale 3D fabrication; stick-on epidermal electronics tattoo to measure UV exposure; a nanotechnology approach to scavenging wind and solar energy in cities; 3D printing highly conductive nanocomposites; using household items to make a multi-sensory 'Paper Skin'; an analogue smart skin that is self-powered; and writing nanotubes with a nano fountain pen.
Here are the 10 most popular Nanowerk Nanotechnology Spotlight articles of 2016. This year, the list includes nanotechnology in textiles; nanotechnology for next-generation inkjet color printing; graphene-based smart contact lens works as self-powered biosensor; nanotechnology's tiny steps toward atomic-scale 3D fabrication; stick-on epidermal electronics tattoo to measure UV exposure; a nanotechnology approach to scavenging wind and solar energy in cities; 3D printing highly conductive nanocomposites; using household items to make a multi-sensory 'Paper Skin'; an analogue smart skin that is self-powered; and writing nanotubes with a nano fountain pen.
Dec 30th, 2016
 Written by Nanowerk's Michael Berger, this just published book is a collection of essays about researchers involved in all facets of nanotechnologies. Nanoscience and nanotechnology research are truly multidisciplinary and international efforts, covering a wide range of scientific disciplines such as medicine, materials sciences, chemistry, biology and biotechnology, physics and electronics. Each of the book's chapters is based on a scientific paper that has been published in a peer-reviewed journal. Although each story revolves around one or two scientists who were interviewed for this book, many, if not most, of the scientific accomplishments covered here are the result of collaborative efforts by several scientists and research groups, often from different organizations and from different countries.
Written by Nanowerk's Michael Berger, this just published book is a collection of essays about researchers involved in all facets of nanotechnologies. Nanoscience and nanotechnology research are truly multidisciplinary and international efforts, covering a wide range of scientific disciplines such as medicine, materials sciences, chemistry, biology and biotechnology, physics and electronics. Each of the book's chapters is based on a scientific paper that has been published in a peer-reviewed journal. Although each story revolves around one or two scientists who were interviewed for this book, many, if not most, of the scientific accomplishments covered here are the result of collaborative efforts by several scientists and research groups, often from different organizations and from different countries.
Aug 31st, 2016
 Neural interfaces establish direct communication between the central nervous system (CNS) and a sovereign, man-made digital system. This technology is perhaps the most important advance in the study and treatment of the brain is the development of the neural interface. Nanotechnology fabrication methods can overcome the limitations of existing interface devices by producing electrodes with an extremely high surface to volume ratio, i.e., more probe units within the same volume, resulting in unprecedented specificity.
Neural interfaces establish direct communication between the central nervous system (CNS) and a sovereign, man-made digital system. This technology is perhaps the most important advance in the study and treatment of the brain is the development of the neural interface. Nanotechnology fabrication methods can overcome the limitations of existing interface devices by producing electrodes with an extremely high surface to volume ratio, i.e., more probe units within the same volume, resulting in unprecedented specificity.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





