 Researchers report that bovine serum albumin (BSA), a standard-issue protein in nano-bio lab experimentation, is prone to pushing gold nanorods into right-handed chiral assemblies - while producing chiral plasmonic signals to match.
Researchers report that bovine serum albumin (BSA), a standard-issue protein in nano-bio lab experimentation, is prone to pushing gold nanorods into right-handed chiral assemblies - while producing chiral plasmonic signals to match.
Sep 26th, 2019
Read more
 Single atoms work great as catalysts, but they usually don't stay single for long. Scientists use high-temperature shock waves to keep them in their place.
Single atoms work great as catalysts, but they usually don't stay single for long. Scientists use high-temperature shock waves to keep them in their place.
Sep 26th, 2019
Read more
 An international research consortium has developed guidelines that should enable the safe development of nanoparticles for medical use.
An international research consortium has developed guidelines that should enable the safe development of nanoparticles for medical use.
Sep 26th, 2019
Read more
 A new way to achieve integrated photonics could have applications in imaging, sensing and quantum information processing, such as on-chip transformation optics, mathematical operations and spectrometers.
A new way to achieve integrated photonics could have applications in imaging, sensing and quantum information processing, such as on-chip transformation optics, mathematical operations and spectrometers.
Sep 26th, 2019
Read more
 A study commissioned by the EU nanomaterials observatory has found that the current EU regulatory framework for characterising and identifying 'next generation' nanomaterials is able to address the majority of them and that no significant changes will be needed in the near future.
A study commissioned by the EU nanomaterials observatory has found that the current EU regulatory framework for characterising and identifying 'next generation' nanomaterials is able to address the majority of them and that no significant changes will be needed in the near future.
Sep 26th, 2019
Read more
 Scientists have invented a new way to monitor chemotherapy concentrations, which is more effective in keeping patients' treatments within the crucial therapeutic window.
Scientists have invented a new way to monitor chemotherapy concentrations, which is more effective in keeping patients' treatments within the crucial therapeutic window.
Sep 25th, 2019
Read more
 Researchers integrate diamond-based sensing components onto a chip to enable low-cost, high-performance quantum hardware.
Researchers integrate diamond-based sensing components onto a chip to enable low-cost, high-performance quantum hardware.
Sep 25th, 2019
Read more
 Design would enable thermophotovoltaic devices that convert waste heat to electricity.
Design would enable thermophotovoltaic devices that convert waste heat to electricity.
Sep 25th, 2019
Read more
 New method fills a need in emerging applications for electronic devices.
New method fills a need in emerging applications for electronic devices.
Sep 25th, 2019
Read more
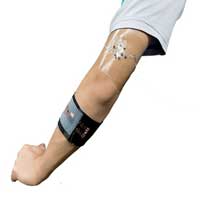 Researchers have developed a unique new flexible and stretchable device, worn against the skin and capable of producing electrical energy by transforming the compounds present in sweat.
Researchers have developed a unique new flexible and stretchable device, worn against the skin and capable of producing electrical energy by transforming the compounds present in sweat.
Sep 25th, 2019
Read more
 The 2D polymers consist of only a few single atomic layers and, due to their very special properties, are a promising material for use in electronic components and systems of a new generation.
The 2D polymers consist of only a few single atomic layers and, due to their very special properties, are a promising material for use in electronic components and systems of a new generation.
Sep 25th, 2019
Read more
 Researchers have engineered genetically encoded protein crystals that can generate magnetic forces many times stronger than those already reported.
Researchers have engineered genetically encoded protein crystals that can generate magnetic forces many times stronger than those already reported.
Sep 25th, 2019
Read more
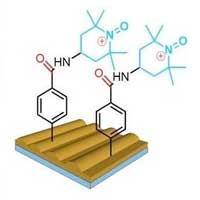 Scientists have designed extremely sensitive sensors for free oxygen-containing radicals that are able to disrupt cell function. These sensors are an alternative to traditional analytical chemical methods of analysis.
Scientists have designed extremely sensitive sensors for free oxygen-containing radicals that are able to disrupt cell function. These sensors are an alternative to traditional analytical chemical methods of analysis.
Sep 25th, 2019
Read more
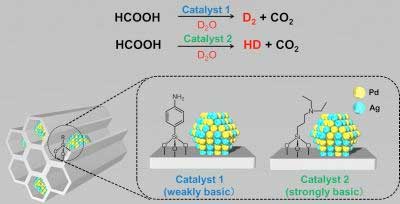 Researchers report an inexpensive and scalable route to hydrogen isotope compounds.
Researchers report an inexpensive and scalable route to hydrogen isotope compounds.
Sep 25th, 2019
Read more
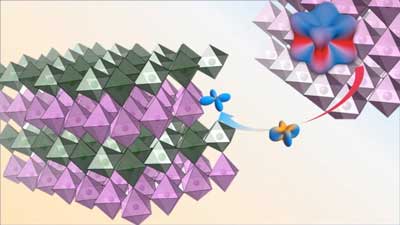 Study could lead to greater manipulation of quantum materials and deeper understanding of the quantum state for novel electronics.
Study could lead to greater manipulation of quantum materials and deeper understanding of the quantum state for novel electronics.
Sep 24th, 2019
Read more
 When a thin layer of water is squeezed between two hydrophobic surfaces, the laws of classical physics break down.
When a thin layer of water is squeezed between two hydrophobic surfaces, the laws of classical physics break down.
Sep 24th, 2019
Read more
 Researchers report that bovine serum albumin (BSA), a standard-issue protein in nano-bio lab experimentation, is prone to pushing gold nanorods into right-handed chiral assemblies - while producing chiral plasmonic signals to match.
Researchers report that bovine serum albumin (BSA), a standard-issue protein in nano-bio lab experimentation, is prone to pushing gold nanorods into right-handed chiral assemblies - while producing chiral plasmonic signals to match.















 Subscribe to our Nanotechnology News feed
Subscribe to our Nanotechnology News feed