Showing Spotlights 1169 - 1176 of 2785 in category All (newest first):
 Over the past few years, researchers have developed numerous methods for synthesizing graphene. The synthesis of high-quality graphene is usually prepared by a complex and costly process - epitaxial growth on transition metal surfaces via chemical vapor deposition using high-purity hydrocarbons as precursors. In new work, researchers demonstrate graphene synthesis by liquid precursor deposition, a process that may give access to a wider range of substrate materials for graphene growth.
Over the past few years, researchers have developed numerous methods for synthesizing graphene. The synthesis of high-quality graphene is usually prepared by a complex and costly process - epitaxial growth on transition metal surfaces via chemical vapor deposition using high-purity hydrocarbons as precursors. In new work, researchers demonstrate graphene synthesis by liquid precursor deposition, a process that may give access to a wider range of substrate materials for graphene growth.
May 28th, 2014
 One of the problems with activated carbon is the disposal of adsorbed contaminants along with the adsorbent. Another concern is that its pores are often blocked during adsorption. By contrast, carbon nanotubes' (CNTs) open structure offers easy, undisrupted access to reactive sites located on nanotubes' outer surface. That's why researchers see CNTs as an attractive potential substitute for activated carbon. Researchers now have demonstrated that individual CNTs can be integrated into micrometer-sized colloidal particles without using a heavy or bulky particulate support.
One of the problems with activated carbon is the disposal of adsorbed contaminants along with the adsorbent. Another concern is that its pores are often blocked during adsorption. By contrast, carbon nanotubes' (CNTs) open structure offers easy, undisrupted access to reactive sites located on nanotubes' outer surface. That's why researchers see CNTs as an attractive potential substitute for activated carbon. Researchers now have demonstrated that individual CNTs can be integrated into micrometer-sized colloidal particles without using a heavy or bulky particulate support.
May 27th, 2014
 A new class of high-density, rechargeable batteries has the potential to address the 'range anxiety' that is inherent to current electric vehicles by drastically increasing their battery capacity: molten air batteries have up to 50 times the storage capacity of lithium-ion batteries. These batteries reversibly use oxygen from the air to store energy via a molten salt and multiple electrons stored per molecule at the counter electrode.
A new class of high-density, rechargeable batteries has the potential to address the 'range anxiety' that is inherent to current electric vehicles by drastically increasing their battery capacity: molten air batteries have up to 50 times the storage capacity of lithium-ion batteries. These batteries reversibly use oxygen from the air to store energy via a molten salt and multiple electrons stored per molecule at the counter electrode.
May 26th, 2014
 Nanobioreactors are emerging as advanced bio-devices, which fuse the advantages of nanomaterials with those of nanobiotechnology. Due to their ultimately small size, high surface area and simulation capacity, they are set to become to be a versatile tool to fabricate ultra-sensitive and selective novel nanobio-devices, which offer us new platform to tackle key energy, medical and environmental issues. Now, a novel two-dimensional bioreactor offers a simple and effective way to overcome many limitations that have been faced by previous designs.
Nanobioreactors are emerging as advanced bio-devices, which fuse the advantages of nanomaterials with those of nanobiotechnology. Due to their ultimately small size, high surface area and simulation capacity, they are set to become to be a versatile tool to fabricate ultra-sensitive and selective novel nanobio-devices, which offer us new platform to tackle key energy, medical and environmental issues. Now, a novel two-dimensional bioreactor offers a simple and effective way to overcome many limitations that have been faced by previous designs.
May 21st, 2014
 Some of the recent and significant developments in the area of nanotechnology applications for food quality control are discussed in this article. In food quality testing methods/devices, nanomaterials have various advantages over conventional materials such as ultra-sensitivity, selectivity, multiple targeting, portability, reproducible data processing, implantable conformability, on-board intelligence, non-invasive testing of packaged items, etc. Analytical chemistry plays a prominent role in food quality control and has already started taking advantages from the versatility and novel merits of nanoscience.
Some of the recent and significant developments in the area of nanotechnology applications for food quality control are discussed in this article. In food quality testing methods/devices, nanomaterials have various advantages over conventional materials such as ultra-sensitivity, selectivity, multiple targeting, portability, reproducible data processing, implantable conformability, on-board intelligence, non-invasive testing of packaged items, etc. Analytical chemistry plays a prominent role in food quality control and has already started taking advantages from the versatility and novel merits of nanoscience.
May 20th, 2014
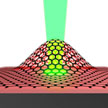 Most nanomotors designs are powered by quantum or, in most cases, catalytic chemical processes, the nanoscale equivalent of conventional internal heat engines that are so prevalent in our daily life has been missing. Researchers have now suggested a new type of ultrathin graphene engine which mimics an internal combustion engine system. This graphene engine consists of only a few parts - functionalized graphene, laser light, and substrate, which would make it simple to work with.
Most nanomotors designs are powered by quantum or, in most cases, catalytic chemical processes, the nanoscale equivalent of conventional internal heat engines that are so prevalent in our daily life has been missing. Researchers have now suggested a new type of ultrathin graphene engine which mimics an internal combustion engine system. This graphene engine consists of only a few parts - functionalized graphene, laser light, and substrate, which would make it simple to work with.
May 16th, 2014
 The nanotechnology-enabled detection of a change in individual cells, for instance cell surface charge, presents a new alternative and complementary method for disease detection and diagnosis. Since diseased cells, such as cancer cells, frequently carry information that distinguishes them from normal cells, accurate probing of these cells is critical for early detection of a disease. For this purpose, researchers have now designed a graphene-based optical refractive index sensor.
The nanotechnology-enabled detection of a change in individual cells, for instance cell surface charge, presents a new alternative and complementary method for disease detection and diagnosis. Since diseased cells, such as cancer cells, frequently carry information that distinguishes them from normal cells, accurate probing of these cells is critical for early detection of a disease. For this purpose, researchers have now designed a graphene-based optical refractive index sensor.
May 15th, 2014
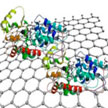 Studies have shown that in a favorable nano environment, enzyme immobilization onto nanosupports could lead to increased enzyme stability and improved specificity, and could allow for prolonged enzyme functionality through chemical and physical treatment. Researchers also have shown that immobilization onto carbon-based nanosupports can increase the enzyme turnover and allow for prolonged enzyme-based conjugates isolation and usage. In new work, researchers have now taken another step towards the detailed characterization and optimization of enzyme-nanosupport interface reactions.
Studies have shown that in a favorable nano environment, enzyme immobilization onto nanosupports could lead to increased enzyme stability and improved specificity, and could allow for prolonged enzyme functionality through chemical and physical treatment. Researchers also have shown that immobilization onto carbon-based nanosupports can increase the enzyme turnover and allow for prolonged enzyme-based conjugates isolation and usage. In new work, researchers have now taken another step towards the detailed characterization and optimization of enzyme-nanosupport interface reactions.
May 14th, 2014
 Over the past few years, researchers have developed numerous methods for synthesizing graphene. The synthesis of high-quality graphene is usually prepared by a complex and costly process - epitaxial growth on transition metal surfaces via chemical vapor deposition using high-purity hydrocarbons as precursors. In new work, researchers demonstrate graphene synthesis by liquid precursor deposition, a process that may give access to a wider range of substrate materials for graphene growth.
Over the past few years, researchers have developed numerous methods for synthesizing graphene. The synthesis of high-quality graphene is usually prepared by a complex and costly process - epitaxial growth on transition metal surfaces via chemical vapor deposition using high-purity hydrocarbons as precursors. In new work, researchers demonstrate graphene synthesis by liquid precursor deposition, a process that may give access to a wider range of substrate materials for graphene growth.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





