| Posted: December 6, 2007 |
Intracellular transport: New portals of entry into cells for pathogenic agents and for medicinal products |
|
(Nanowerk News) How does the cell membrane capture pathogenic agents bound to its surface? Surprisingly, the membrane invaginates through a spontaneous and autonomous movement and swallows pathogens. This mechanism has been demonstrated in cells, and also in a minimal artificial membrane system. An international collaboration between physicists, including Patricia Bassereau and her CNRS team, chemists, and cell biologists at the Institut Curie, has observed this process at work with a particular pathogen, Shiga toxin. The work has been done by Ludger Johannes, Research Director at Inserm, and his CNRS Trafficking, Signaling, and Delivery team, using the Institut Curie's imaging equipment. The results shed new light on unexpected aspects of a fundamental process in biology—endocytosis(1). They also point to new leads in the search for the portal of entry of certain pathogenic agents, or to expedite the entry of drugs, therapeutic vaccines, or diagnostic agents in cancer cells. This work was published online in Nature of 29 November 2007.
|
|
Like in cartoons depicting how the inhospitable jungle magically opens before an intruder only to close behind him as he passes on his way, the cell membrane acts similarly when it spontaneously invaginates on contact with certain pathogenic agents. The membrane has no need of magic or trickery, just the laws of physics, as shown at the Institut Curie by a group of researchers working on Shiga toxin, a pathogenic agent produced by intestinal bacteria (see references and box). Shiga toxin clutches several receptors along its way, drawing an “islet in the ocean of the membrane”, according to Winfried Römer, a young post-doc in the team of Ludger Johannes(2). This islet forms a depression to produce a sort of “finger”, or tubule, pushing into the cell. The tubule is then cut off by the cellular machinery and internalized.
|
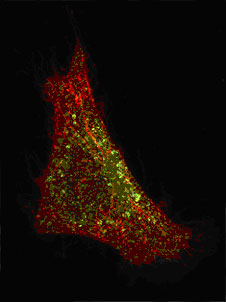 |
| In this cell, the energy has been suppressed. The presence of Shiga toxin (labeled red) is enough to invaginate the cell membrane, without the help of clathrin (indicated by the green labeling of the clathrin-dependent receptor). Shiga toxin and clathrin are not co-localized. (Image: W. Rοmer-V. Fraisier/CNRS/Institut Curie)
|
|
This discovery may seem trivial to the uninitiated, but in fact represents an important conceptual advance, which casts new light on endocytosis. This mechanism allows large molecules and particles, generically called “cargos” (extracellular ligands, nutrients, viruses, bacteria, toxins) to enter the cell in endocytotic vesicles, as if in a Trojan horse. Current data suggest that the cargo reaches a predetermined platform before being taken over by the cell, using energy in the process and with the help of proteins, one of which, clathrin, constructs a coat that presses into the membrane the shape of an empty cage. This cage closes around the membrane vesicle containing the cargo, which is internalized in the cell. The endocytotic vesicles then fuse with the endosomes, which are intracellular compartments enabling the sorting of internalized molecules. These molecules then meet various fates: return to the plasma membrane (recycling), degradation, transfer to other intracellular compartments (Golgi apparatus, endoplasmic reticulum, etc). It has been recognized, however, that this clathrin-dependent pathway of endocytosis is not unique. Indeed, researchers have observed before that some cargos, like Shiga toxin, are captured by other, as yet unspecified mechanisms.
|
|
After more than three years of research at the Institut Curie, the scientists have discovered a new method of access to the cell that does not involve cell proteins like clathrin. The mechanical forces imposed on the membrane, in this case by Shiga toxin, are enough to trigger its invagination.
|
|
Using various techniques to suppress at will the cell's energy or certain molecules (actin, dynamin...), the researchers were able to identify how the membrane invaginates and forms tubules, in response to the binding of the B-subunit of Shiga toxin: this part of the molecule has specific receptors in the cell membrane, the Gb3 glycolipid. The invagination is spontaneous, but highly selective: the tubules were not formed when the researchers replaced the B-subunit by a decoy.
|
|
The CNRS team of physicists headed by Patricia Bassereau at the Institut Curie has reproduced this mechanism in an original system: a made-to-measure artificial lipid bilayer modeled on the plasma membrane. When the B-subunit of Shiga toxin is put in contact with this giant vesicle of 10 to 30 micrometers in diameter, they form intense spots, from which the tubules form. As expected, neither spots nor tubules are formed when the membrane is devoid of the right receptors. Using receptor variants synthesized by chemists in Jean-Claude Florent's CNRS group, the researchers tested and verified another much more groundbreaking hypothesis: that the Gb3 receptor is itself implicated in the invagination. Each B-subunit grips up to 15 receptors, whose bulky lipid tails form the wide base of a cone. “Our artificial membrane system is the key manipulation showing, and we have images to back this up, that the invagination needs no energy or cellular components to occur. Only the concentration of toxins closely bound to their receptors creates microdomains with depressions in the membrane that accumulate and end up forming a tubule”, summarizes Winfried Römer.
|
|
This discovery points to new concepts about endocytosis. This purely physical mechanism may apply to cargos other than Shiga toxin, notably certain viruses. Ludger Johannes has said that “the membrane is more active than we thought, and even when clathrin intervenes, it perhaps organizes microdomains rather than scaffolding the membrane into cage-like structures.” This could lead on to new strategies in the fight against infections, poisonings, and cancers, by, for instance, targeting the membrane lipids, which are still poorly understood. This is how Institut Curie researchers, headed by Ludger Johannes, use the B-subunit of Shiga toxin to transport antigens inside certain immune cells to trigger an anti-tumor or antiviral immune response. Similarly, the same transporter can target and selectively destroy cancer cells.
|

