| Posted: March 27, 2009 |
Selectivity at the double |
|
(Nanowerk News) The discovery of new synthetic methodology is an important goal in synthetic organic chemistry. New methodology may focus on the efficient production of a particular class of small molecules, but in deciding on a target, two important factors must be taken into account. First, the target must be a useful ‘synthon’—that is, it must be easy to make into more complex molecules. Second, the new methodology needs to be able to provide a significant improvement in, for example, selectivity or yield over those already available.
|
|
Writing in the international edition of Angewandte Chemie (Synthesis of (Z)-1-Aza-1,3-enynes by the Cross-Coupling of Terminal Alkynes with Isocyanides Catalyzed by Rare-Earth Metal Complexes), Zhaomin Hou and colleagues from the RIKEN Advanced Science Institute in Wako have used an yttrium catalyst to develop a new method for the synthesis of (Z)-1-aza-1,3-enynes. The methodology fulfills both objectives set out above. The products are densely functionalized—within the central four-atom structure, they contain a basic nitrogen, an imine group and a highly reactive alkyne. However, there are two possible isomers (E and Z) of the products owing to the arrangement of substituents, or stereochemistry, around the carbon-nitrogen (C=N) double bond. Previously reported synthetic methods usually yield a mixture of the two isomers.
|
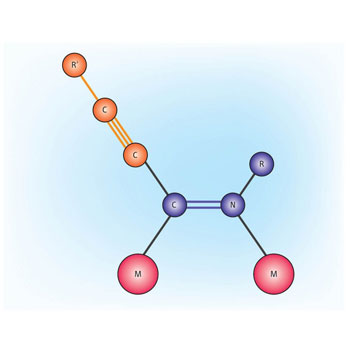 |
| Fig. 1:A schematic of the catalyst having two metal centers (M = metal), at which an alkyne unit (orange) is coupled to an isocyanate unit (blue). The stereochemistry of the product is enforced by the presence of two metal centers. (R and R’ represent a variety of hydrocarbon substituents.)
|
|
The methodology developed by Hou and coworkers results in selective formation of only the Z isomer. The researchers first tested a series of rare-earth metal half-sandwich complexes—so called because the metal is coordinated from one side by a single planar ligand. Yttrium complexes were found to have the highest activity in this reaction. They then showed that the catalyst was effective for the cross-coupling of a wide variety of alkyl and aryl acetylenes (R’ in Fig. 1) with alkyl isocyanates (R in Fig. 1).
|
|
Further investigations showed that the active catalyst species likely contains two metal centers (Fig. 1). “We believe that the two metals work together in a cooperative way and provide products unavailable previously with a catalyst incorporating a single metal center,” says Hou. The use of a half-sandwich complex is important as it is known that the sandwich complexes tend to form monomeric active catalysts—and these produce a mixture of isomers.
|
|
The team is continuing to work on designing new catalysts based on two or more metal centers. “We hope to find catalysts for new reactions, or that can provide access to new materials not available using known catalysts,” says Hou.
|

