Showing Spotlights 17 - 24 of 65 in category All (newest first):
 Most printed electronics applications rely on some kind of ink formulated with conductive nanomaterials. Researchers have now introduced a rapid and facile method to fabricate a foldable capacitive touch pad using silver nanowire inks. The team developed a technique that uses a 2D programmed printing machine with postdeposition sintering using a camera flash light to harden the deposited silver nanowire ink. resulting paper-based touchpads produced by direct writing with silver nanowire inks offer several distinct advantages over existing counterparts.
Most printed electronics applications rely on some kind of ink formulated with conductive nanomaterials. Researchers have now introduced a rapid and facile method to fabricate a foldable capacitive touch pad using silver nanowire inks. The team developed a technique that uses a 2D programmed printing machine with postdeposition sintering using a camera flash light to harden the deposited silver nanowire ink. resulting paper-based touchpads produced by direct writing with silver nanowire inks offer several distinct advantages over existing counterparts.
Nov 24th, 2014
 The food chemistry Maillard reaction is responsible for many colors and flavors in foods - roasting of coffee, baking of bread and sizzling of meat. Scientists have made use of this ingenious food chemistry to 'cook' their copper nanowires. This green approach that formulates copper atoms in water to form untangled metallic state nanowires. Naturally, a lingering chocolate-like aroma was detected during the copper nanowires synthesis.
The food chemistry Maillard reaction is responsible for many colors and flavors in foods - roasting of coffee, baking of bread and sizzling of meat. Scientists have made use of this ingenious food chemistry to 'cook' their copper nanowires. This green approach that formulates copper atoms in water to form untangled metallic state nanowires. Naturally, a lingering chocolate-like aroma was detected during the copper nanowires synthesis.
Nov 21st, 2014
 Over the years, researchers have developed a large number of techniques to synthesize nanowires and nanotubes in the laboratory. These procedures vary widely in their hardware requirements and methodology. Nevertheless, they all share a set of common goals: simplicity of protocol; fast execution; and low energy input. Now, an international group of scientists has reported a breakthrough in all three of these areas, leading to a revolutionary and remarkably simple technique for preparing one-dimensional nanostructures. As an example, they demonstrate a unique approach to growing amorphous boron nanowires.
Over the years, researchers have developed a large number of techniques to synthesize nanowires and nanotubes in the laboratory. These procedures vary widely in their hardware requirements and methodology. Nevertheless, they all share a set of common goals: simplicity of protocol; fast execution; and low energy input. Now, an international group of scientists has reported a breakthrough in all three of these areas, leading to a revolutionary and remarkably simple technique for preparing one-dimensional nanostructures. As an example, they demonstrate a unique approach to growing amorphous boron nanowires.
Feb 4th, 2014
 Nanowire field-effect sensors show significant advantages of real-time, label-free and highly sensitive detection of a wide range of analytes in liquid phase, including proteins, nucleic acids, small molecules, and viruses in single-element or multiplexed formats. Motivated by the unique features of these sensors and the ease to integrate them in the currently available VLSI technology, researchers used molecularly modified silicon nanowire FETs to detect volatile organic compounds (VOCs) that are associated with environmental pollution, quality control, explosive materials, or various diseases.
Nanowire field-effect sensors show significant advantages of real-time, label-free and highly sensitive detection of a wide range of analytes in liquid phase, including proteins, nucleic acids, small molecules, and viruses in single-element or multiplexed formats. Motivated by the unique features of these sensors and the ease to integrate them in the currently available VLSI technology, researchers used molecularly modified silicon nanowire FETs to detect volatile organic compounds (VOCs) that are associated with environmental pollution, quality control, explosive materials, or various diseases.
Jan 30th, 2014
 Nanowires are considered a major building block for future nanotechnology devices, with great potential for applications in transistors, solar cells, lasers, sensors, etc. Now, for the first time, nanotechnology researchers have utilized nanowires as a 'storage' device for biochemical species such as ATP. The team demonstrated that their nano-storage wire structure can be deposited onto virtually any substrate to build nanostorage devices for the real-time controlled release of biochemical molecules upon the application of electrical stimuli.
Nanowires are considered a major building block for future nanotechnology devices, with great potential for applications in transistors, solar cells, lasers, sensors, etc. Now, for the first time, nanotechnology researchers have utilized nanowires as a 'storage' device for biochemical species such as ATP. The team demonstrated that their nano-storage wire structure can be deposited onto virtually any substrate to build nanostorage devices for the real-time controlled release of biochemical molecules upon the application of electrical stimuli.
Jul 31st, 2013
 Nanoscale materials like quantum dots, carbon nanotubes, graphene, or nanowires, have intriguing properties, but unless they can be assembled in to larger structures it is difficult to take advantage of these properties. Figuring out how to assemble nanostructures into functional macroscale assemblies is one of the key challenges that nanoscientists around the world are faced with. In the area of nanowires, this has led to researchers exploring various nanowire assembly techniques ranging from Langmuir Blodgett alignment to electrospinning. Researchers have now developed a novel approach for assembling nanowires into macroscopic yarns that consist of millions of nanowires bundled together. The team found that light can be used to charge inorganic semiconducting nanowires. Once charged, the nanowires can be manipulated with electric fields.
Nanoscale materials like quantum dots, carbon nanotubes, graphene, or nanowires, have intriguing properties, but unless they can be assembled in to larger structures it is difficult to take advantage of these properties. Figuring out how to assemble nanostructures into functional macroscale assemblies is one of the key challenges that nanoscientists around the world are faced with. In the area of nanowires, this has led to researchers exploring various nanowire assembly techniques ranging from Langmuir Blodgett alignment to electrospinning. Researchers have now developed a novel approach for assembling nanowires into macroscopic yarns that consist of millions of nanowires bundled together. The team found that light can be used to charge inorganic semiconducting nanowires. Once charged, the nanowires can be manipulated with electric fields.
Oct 18th, 2012
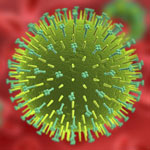 Breath analysis of exhaled breath condensate has been increasingly recognized as a promising diagnostic method to link specific gaseous components in human breath to medical conditions and exposure to chemical compounds. Sampling breath is also much less invasive than testing blood, can be done very quickly, and creates as good as no biohazard waste. Studies have shown that exhaled breath from a flu patient contains influenza viruses but, although the use of silicon nanowire (SiNW) sensors for virus detection is not new, so far no studies have been conducted to apply silicon nanowire technology to the diagnosis of flu. Now, new research suggests that a SiNW sensor device, when calibrated by virus standards and exhaled breath condensate controls, can be reliably applied to the diagnosis of flu in a clinical setting with two orders of magnitude less time compared to the gold standard method RT-qPCR.
Breath analysis of exhaled breath condensate has been increasingly recognized as a promising diagnostic method to link specific gaseous components in human breath to medical conditions and exposure to chemical compounds. Sampling breath is also much less invasive than testing blood, can be done very quickly, and creates as good as no biohazard waste. Studies have shown that exhaled breath from a flu patient contains influenza viruses but, although the use of silicon nanowire (SiNW) sensors for virus detection is not new, so far no studies have been conducted to apply silicon nanowire technology to the diagnosis of flu. Now, new research suggests that a SiNW sensor device, when calibrated by virus standards and exhaled breath condensate controls, can be reliably applied to the diagnosis of flu in a clinical setting with two orders of magnitude less time compared to the gold standard method RT-qPCR.
Jun 29th, 2012
 Burn injuries are one of the major global health problems. Every year 195,000 people from all over the world die because of fire alone. A burn injury may damage some or all skin layers and is caused by a hot solid, a hot liquid, or a flame. However, injuries related to electricity, radioactivity, ultraviolet radiation, chemicals and respiratory damage due to smoking are also considered as burn injuries. Besides cleaning the wound and applying various topical anti-microbial agents, wound dressings could be an effective solution in preventing microbial infections for burn care. The suitability of a burn wound dressing depends on a burn type. Conventional dressings are not efficient enough to induce haemostasis, adherence and in holding a moist environment around wound. Due to the advances in the field of nanotechnology, it is now possible to design nanofiber-based wound dressings where an electrospun-nanofibrous layer is applied to a basic support fabric material.
Burn injuries are one of the major global health problems. Every year 195,000 people from all over the world die because of fire alone. A burn injury may damage some or all skin layers and is caused by a hot solid, a hot liquid, or a flame. However, injuries related to electricity, radioactivity, ultraviolet radiation, chemicals and respiratory damage due to smoking are also considered as burn injuries. Besides cleaning the wound and applying various topical anti-microbial agents, wound dressings could be an effective solution in preventing microbial infections for burn care. The suitability of a burn wound dressing depends on a burn type. Conventional dressings are not efficient enough to induce haemostasis, adherence and in holding a moist environment around wound. Due to the advances in the field of nanotechnology, it is now possible to design nanofiber-based wound dressings where an electrospun-nanofibrous layer is applied to a basic support fabric material.
Jun 13th, 2012
 Most printed electronics applications rely on some kind of ink formulated with conductive nanomaterials. Researchers have now introduced a rapid and facile method to fabricate a foldable capacitive touch pad using silver nanowire inks. The team developed a technique that uses a 2D programmed printing machine with postdeposition sintering using a camera flash light to harden the deposited silver nanowire ink. resulting paper-based touchpads produced by direct writing with silver nanowire inks offer several distinct advantages over existing counterparts.
Most printed electronics applications rely on some kind of ink formulated with conductive nanomaterials. Researchers have now introduced a rapid and facile method to fabricate a foldable capacitive touch pad using silver nanowire inks. The team developed a technique that uses a 2D programmed printing machine with postdeposition sintering using a camera flash light to harden the deposited silver nanowire ink. resulting paper-based touchpads produced by direct writing with silver nanowire inks offer several distinct advantages over existing counterparts.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





