Showing Spotlights 41 - 48 of 65 in category All (newest first):
 Semiconducting nanowires are known to be extremely sensitive to chemical species adsorbed on their surfaces. For a nanowire device, the binding of a charged analyte to the surface of the nanowire leads to a conductance change, or a change in current flowing through these tinny wires. Their one-dimensional nanoscale morphology and their extremely high surface-to-volume ratio make this conductance change to be much greater for nanowire-based sensors versus planar field-effect transistors, increasing the sensitivity to a point that single molecule detection is possible. In the last decade, it has been demonstrated that these new nanostructures can be used for the detection of multiple biomolecular species of medical diagnostic relevance, such as DNA and proteins. In recent work, researchers have used the ultrasensitive recognition properties of semiconducting silicon nanowires to demonstrate the most sensitive ever published sensing of explosives reported so far.
Semiconducting nanowires are known to be extremely sensitive to chemical species adsorbed on their surfaces. For a nanowire device, the binding of a charged analyte to the surface of the nanowire leads to a conductance change, or a change in current flowing through these tinny wires. Their one-dimensional nanoscale morphology and their extremely high surface-to-volume ratio make this conductance change to be much greater for nanowire-based sensors versus planar field-effect transistors, increasing the sensitivity to a point that single molecule detection is possible. In the last decade, it has been demonstrated that these new nanostructures can be used for the detection of multiple biomolecular species of medical diagnostic relevance, such as DNA and proteins. In recent work, researchers have used the ultrasensitive recognition properties of semiconducting silicon nanowires to demonstrate the most sensitive ever published sensing of explosives reported so far.
Sep 14th, 2010
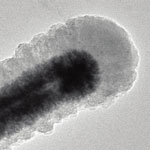 Most of today's lithium-ion batteries rely on anodes made from graphite, a form of carbon. There are several candidate electrodes to replace graphite as the anode for lithium-ion batteries. Such electrodes like silicon or tin have very high capacities but suffer from poor efficiency and cyclic stability as they experience large volume change and particle pulverization during repeated cycling. Silicon-based anodes, for instance, theoretically offer as much as a ten-fold capacity improvement over graphite, but silicon-based anodes have so far not been stable enough for practical use. Researchers have now used the macromolecular structure of self-assembled Tobacco mosaic virus as templates to fabricate stable three-dimensional current collectors for high power and high energy density Li-ion batteries.
Most of today's lithium-ion batteries rely on anodes made from graphite, a form of carbon. There are several candidate electrodes to replace graphite as the anode for lithium-ion batteries. Such electrodes like silicon or tin have very high capacities but suffer from poor efficiency and cyclic stability as they experience large volume change and particle pulverization during repeated cycling. Silicon-based anodes, for instance, theoretically offer as much as a ten-fold capacity improvement over graphite, but silicon-based anodes have so far not been stable enough for practical use. Researchers have now used the macromolecular structure of self-assembled Tobacco mosaic virus as templates to fabricate stable three-dimensional current collectors for high power and high energy density Li-ion batteries.
Aug 24th, 2010
 Nanotechnology-enabled tissue engineering is a rapidly growing field. At the core of tissue engineering is the construction of scaffolds out of biomaterials to provide mechanical support and guide cell growth into new tissues or organs. In particular, electrospun biodegradable polymeric nanofibers are being used in scaffolds for engineering various tissues such as nerves, cartilages or bone. Electrospinning is a fabrication technique which can produce nanoscale fibers from more than 100 different polymers. The electrospun nanofibers are typically collected as nonwoven mats with random orientation. A new study has now demonstrated the fabrication of a novel class of nanofiber scaffold composed of radially-aligned, electrospun nanofibers and also demonstrated the unique application of these materials as effective biomedical patches/scaffolds that could prove to be beneficial during neurosurgery.
Nanotechnology-enabled tissue engineering is a rapidly growing field. At the core of tissue engineering is the construction of scaffolds out of biomaterials to provide mechanical support and guide cell growth into new tissues or organs. In particular, electrospun biodegradable polymeric nanofibers are being used in scaffolds for engineering various tissues such as nerves, cartilages or bone. Electrospinning is a fabrication technique which can produce nanoscale fibers from more than 100 different polymers. The electrospun nanofibers are typically collected as nonwoven mats with random orientation. A new study has now demonstrated the fabrication of a novel class of nanofiber scaffold composed of radially-aligned, electrospun nanofibers and also demonstrated the unique application of these materials as effective biomedical patches/scaffolds that could prove to be beneficial during neurosurgery.
Aug 17th, 2010
 All existing transistors are based on junctions - obtained by changing the polarity of silicon from positive to negative. Researchers have now demonstrated a new type of transistor in which there are no junctions and no doping concentration gradients. The key to fabricating a junctionless gated resistor is the formation of a semiconductor layer that is thin and narrow enough to allow for full depletion of carriers when the device is turned off - something that was achieved by fabricating silicon nanowires with a diameter of a few dozens of atomic planes. The electrical current flows in this silicon nanowire, and the flow of current is perfectly controlled by a ring structure that electrically squeezes the silicon wire in the same way that you might stop the flow of water in a hose by squeezing it.
All existing transistors are based on junctions - obtained by changing the polarity of silicon from positive to negative. Researchers have now demonstrated a new type of transistor in which there are no junctions and no doping concentration gradients. The key to fabricating a junctionless gated resistor is the formation of a semiconductor layer that is thin and narrow enough to allow for full depletion of carriers when the device is turned off - something that was achieved by fabricating silicon nanowires with a diameter of a few dozens of atomic planes. The electrical current flows in this silicon nanowire, and the flow of current is perfectly controlled by a ring structure that electrically squeezes the silicon wire in the same way that you might stop the flow of water in a hose by squeezing it.
Mar 1st, 2010
 One of the outstanding challenges in nanotechnology generally, and in the exploitation of so-called 'bottom-up' assembly of basic nanoscale building blocks such as nanowires, is the development of techniques for assembling large numbers of such nanostructures into more complex systems and precisely specified patterns in an accurate, deterministic manner. For instance, it is possible to build transistors, optical devices, and sensors with very specific properties using nanowires. Thus many useful applications of nanowires will depend on the ability to take these building blocks and organize them in some deterministic way in order to ultimately construct and interface with a nanowire-based system. New work demonstrates the basic capability for, and elucidates some of the guiding principles in, the use of dielectrophoretic behavior to direct the placement of large numbers of nanowires on complex, pre-patterned structures as might be required for integration of nanowires with, for example, silicon-based microelectronic circuitry. It shows that a high level of placement precision can be achieved by paying careful attention to the signal frequency as well as the macroscopic electrode architecture that is employed.
One of the outstanding challenges in nanotechnology generally, and in the exploitation of so-called 'bottom-up' assembly of basic nanoscale building blocks such as nanowires, is the development of techniques for assembling large numbers of such nanostructures into more complex systems and precisely specified patterns in an accurate, deterministic manner. For instance, it is possible to build transistors, optical devices, and sensors with very specific properties using nanowires. Thus many useful applications of nanowires will depend on the ability to take these building blocks and organize them in some deterministic way in order to ultimately construct and interface with a nanowire-based system. New work demonstrates the basic capability for, and elucidates some of the guiding principles in, the use of dielectrophoretic behavior to direct the placement of large numbers of nanowires on complex, pre-patterned structures as might be required for integration of nanowires with, for example, silicon-based microelectronic circuitry. It shows that a high level of placement precision can be achieved by paying careful attention to the signal frequency as well as the macroscopic electrode architecture that is employed.
Jun 19th, 2009
 The fight against infections is as old as civilization. Silver, for instance, had already been recognized in ancient Greece and Rome for its infection-fighting properties and it has a long and intriguing history as an antibiotic in human health care. Modern day pharmaceutical companies developed powerful antibiotics - which also happen to be much more profitable than just plain old silver - an apparent high-tech solution to get nasty microbes such as harmful bacteria under control. However, thanks to emerging nanotechnology applications, silver is making a comeback in the form of antimicrobial nanoparticle coatings. As even the most powerful antibiotics become less and less effective, researchers have begun to re-evaluate old antimicrobial substances such as silver and as a result, antimicrobial nano-silver applications have become a very popular early commercial nanotechnology product. Researchers in China have now further advanced the nanotechnology application of silver be developing a novel multi-action nanofiber membrane containing four active components, each playing a different role in the membrane's excellent antibacterial function.
The fight against infections is as old as civilization. Silver, for instance, had already been recognized in ancient Greece and Rome for its infection-fighting properties and it has a long and intriguing history as an antibiotic in human health care. Modern day pharmaceutical companies developed powerful antibiotics - which also happen to be much more profitable than just plain old silver - an apparent high-tech solution to get nasty microbes such as harmful bacteria under control. However, thanks to emerging nanotechnology applications, silver is making a comeback in the form of antimicrobial nanoparticle coatings. As even the most powerful antibiotics become less and less effective, researchers have begun to re-evaluate old antimicrobial substances such as silver and as a result, antimicrobial nano-silver applications have become a very popular early commercial nanotechnology product. Researchers in China have now further advanced the nanotechnology application of silver be developing a novel multi-action nanofiber membrane containing four active components, each playing a different role in the membrane's excellent antibacterial function.
Jun 2nd, 2009
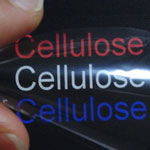 Synthetic fibers are ubiquitous in modern society and their manufacture represents a huge, multi-billion dollar worldwide industry. Synthetic fibers - carbon fibers, nylon, polyester, kevlar, spandex, etc. - are manufactured from fossil fuels, usually from oil, but sometimes from coal or natural gas. Most of these materials are not biodegradable and, in addition to their significant carbon footprint during production, they pose environmental problems at the end of their life cycle. Natural fibers, on the other hand, such as wool and cotton, come from renewable animal or plant sources but they usually lack the high-performance characteristics of many synthetic fibers. This may change, as the new field of bio-based nanomaterials promises to deliver environmentally friendly, high-performance bio-fiber materials that can replace some of the synthetic materials.
Synthetic fibers are ubiquitous in modern society and their manufacture represents a huge, multi-billion dollar worldwide industry. Synthetic fibers - carbon fibers, nylon, polyester, kevlar, spandex, etc. - are manufactured from fossil fuels, usually from oil, but sometimes from coal or natural gas. Most of these materials are not biodegradable and, in addition to their significant carbon footprint during production, they pose environmental problems at the end of their life cycle. Natural fibers, on the other hand, such as wool and cotton, come from renewable animal or plant sources but they usually lack the high-performance characteristics of many synthetic fibers. This may change, as the new field of bio-based nanomaterials promises to deliver environmentally friendly, high-performance bio-fiber materials that can replace some of the synthetic materials.
Dec 18th, 2008
 Optical imaging of materials is full with rich physical, chemical and biological information about the sample, because the optical energies in the visible range coincide with the atomic and molecular transition energies of many materials. Apart from the topographical information, the optical image therefore contains information about intrinsic properties of a material. However, the wave nature of light prevents the light to focus in a volume smaller than half of the wavelength, which is about 200-300 nm for visible light. Therefore, it is almost impossible to image nanomaterials, which could be a few nanometers in size, using optical imaging process. A typical lens made of, for example glass, will not be sufficient to image a nanomaterial. In work that gives rise to a new concept of a lens for optical imaging, scientists in Japan have proposed a lens made of silver nanorods, rather than a curved glass surface. This metallic nanolens is capable of manipulating light in such a way that an optical image of nanoscale objects can be obtained in the visible range.
Optical imaging of materials is full with rich physical, chemical and biological information about the sample, because the optical energies in the visible range coincide with the atomic and molecular transition energies of many materials. Apart from the topographical information, the optical image therefore contains information about intrinsic properties of a material. However, the wave nature of light prevents the light to focus in a volume smaller than half of the wavelength, which is about 200-300 nm for visible light. Therefore, it is almost impossible to image nanomaterials, which could be a few nanometers in size, using optical imaging process. A typical lens made of, for example glass, will not be sufficient to image a nanomaterial. In work that gives rise to a new concept of a lens for optical imaging, scientists in Japan have proposed a lens made of silver nanorods, rather than a curved glass surface. This metallic nanolens is capable of manipulating light in such a way that an optical image of nanoscale objects can be obtained in the visible range.
Jun 17th, 2008
 Semiconducting nanowires are known to be extremely sensitive to chemical species adsorbed on their surfaces. For a nanowire device, the binding of a charged analyte to the surface of the nanowire leads to a conductance change, or a change in current flowing through these tinny wires. Their one-dimensional nanoscale morphology and their extremely high surface-to-volume ratio make this conductance change to be much greater for nanowire-based sensors versus planar field-effect transistors, increasing the sensitivity to a point that single molecule detection is possible. In the last decade, it has been demonstrated that these new nanostructures can be used for the detection of multiple biomolecular species of medical diagnostic relevance, such as DNA and proteins. In recent work, researchers have used the ultrasensitive recognition properties of semiconducting silicon nanowires to demonstrate the most sensitive ever published sensing of explosives reported so far.
Semiconducting nanowires are known to be extremely sensitive to chemical species adsorbed on their surfaces. For a nanowire device, the binding of a charged analyte to the surface of the nanowire leads to a conductance change, or a change in current flowing through these tinny wires. Their one-dimensional nanoscale morphology and their extremely high surface-to-volume ratio make this conductance change to be much greater for nanowire-based sensors versus planar field-effect transistors, increasing the sensitivity to a point that single molecule detection is possible. In the last decade, it has been demonstrated that these new nanostructures can be used for the detection of multiple biomolecular species of medical diagnostic relevance, such as DNA and proteins. In recent work, researchers have used the ultrasensitive recognition properties of semiconducting silicon nanowires to demonstrate the most sensitive ever published sensing of explosives reported so far.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





