Showing Spotlights 361 - 368 of 544 in category All (newest first):
 Continuing miniaturization has moved the semiconductor industry well into the nano realm with leading chip manufacturers on their way to CMOS using 22nm process technology. With transistors the size of tens of nanometers, researchers have begun to explore the interface of biology and electronics by integrating nanoelectronic components and living cells. While researchers have already experimented with integrating living cells into semiconductor materials other research is exploring the opposite way, i.e. integrating nanoelectronics into living cells. Researchers in Spain have demonstrated that silicon chips smaller than cells can be produced, collected, and internalized inside living cells by different techniques (lipofection, phagocytosis or microinjection) and, most significantly, they can be used as intracellular sensors.
Continuing miniaturization has moved the semiconductor industry well into the nano realm with leading chip manufacturers on their way to CMOS using 22nm process technology. With transistors the size of tens of nanometers, researchers have begun to explore the interface of biology and electronics by integrating nanoelectronic components and living cells. While researchers have already experimented with integrating living cells into semiconductor materials other research is exploring the opposite way, i.e. integrating nanoelectronics into living cells. Researchers in Spain have demonstrated that silicon chips smaller than cells can be produced, collected, and internalized inside living cells by different techniques (lipofection, phagocytosis or microinjection) and, most significantly, they can be used as intracellular sensors.
Mar 15th, 2010
 Nanobiotechnology is essentially different in many aspects from other areas of nanotechnology such as nanoelectronics or nanomaterials. It is certainly the most complex sub-area of nanotechnology. Last month, EuroNanoBio - a Support Action funded under the 7th Framework Programme of the European Union - has issued its report on a conceptual framework and a tool box to structure the European capacity in nanobiotechnology. The EuroNanoBio partners explored the definition, establishment and further development of a European scale infrastructure on nanobiotechnology and the associated realistic implementation plan. It aimed at defining not only the key features of a potential European infrastructure in nanobiotechnology, but it has also established the way it should be designed.
Nanobiotechnology is essentially different in many aspects from other areas of nanotechnology such as nanoelectronics or nanomaterials. It is certainly the most complex sub-area of nanotechnology. Last month, EuroNanoBio - a Support Action funded under the 7th Framework Programme of the European Union - has issued its report on a conceptual framework and a tool box to structure the European capacity in nanobiotechnology. The EuroNanoBio partners explored the definition, establishment and further development of a European scale infrastructure on nanobiotechnology and the associated realistic implementation plan. It aimed at defining not only the key features of a potential European infrastructure in nanobiotechnology, but it has also established the way it should be designed.
Feb 19th, 2010
 In many diagnostic processes, the detection of several protein markers is required. Rather than performing several sequential analysis steps, a multiplexed approach allows the simultaneous measurement of multiple biomarkers from the same blood sample. The convergence of nanotechnology, microtechnology, microfluidics, photonics, signal processing, and proteomics allows for the development of increasingly sophisticated and effective multiplexed point-of-care diagnostic devices. The detection of protein biomarkers can done with an optical multiplexing approach that uses dye particles of different colors. In contrast to conventional fluorescence dyes, quantum dots generate a much more powerful fluorescent signal which provides a large increase in sensitivity compared to other methods. Quantum dots are also available in multiple colors, allowing the investigators to tag each antibody with a uniquely colored quantum dot. Researchers have now demonstrated a novel and fast quantum dot-based FRET technique that is suitable for multiplexed ultrasensitive detection.
In many diagnostic processes, the detection of several protein markers is required. Rather than performing several sequential analysis steps, a multiplexed approach allows the simultaneous measurement of multiple biomarkers from the same blood sample. The convergence of nanotechnology, microtechnology, microfluidics, photonics, signal processing, and proteomics allows for the development of increasingly sophisticated and effective multiplexed point-of-care diagnostic devices. The detection of protein biomarkers can done with an optical multiplexing approach that uses dye particles of different colors. In contrast to conventional fluorescence dyes, quantum dots generate a much more powerful fluorescent signal which provides a large increase in sensitivity compared to other methods. Quantum dots are also available in multiple colors, allowing the investigators to tag each antibody with a uniquely colored quantum dot. Researchers have now demonstrated a novel and fast quantum dot-based FRET technique that is suitable for multiplexed ultrasensitive detection.
Feb 17th, 2010
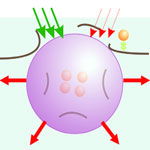 Multifunctional nanoparticles are at the core of a growing field called theranostics that develops technologies physicians can use to diagnose and treat diseases in a single procedure. The major promise of theranostics is to bring together key stages of a medical treatment, such as the diagnosis and therapy, and thus to make a treatment shorter, safer and more efficient. Theranostic approaches require adequate tools with a high multi-functionality and selectivity. The initial phase of the development of theranostics has already revealed the two general challenges: lack of multifunctional methods and agents, and the lack of selectivity and specificity of available agents - that ultimately requires cell and molecular levels. Researchers have now developed a novel method based on gold nanoparticle-generated transient photothermal vapor nanobubbles, a structure they refer to as plasmonic nanobubbles.
Multifunctional nanoparticles are at the core of a growing field called theranostics that develops technologies physicians can use to diagnose and treat diseases in a single procedure. The major promise of theranostics is to bring together key stages of a medical treatment, such as the diagnosis and therapy, and thus to make a treatment shorter, safer and more efficient. Theranostic approaches require adequate tools with a high multi-functionality and selectivity. The initial phase of the development of theranostics has already revealed the two general challenges: lack of multifunctional methods and agents, and the lack of selectivity and specificity of available agents - that ultimately requires cell and molecular levels. Researchers have now developed a novel method based on gold nanoparticle-generated transient photothermal vapor nanobubbles, a structure they refer to as plasmonic nanobubbles.
Feb 1st, 2010
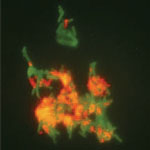 About two years ago we reported on the concept of a biological nanofactory that comprises multiple functional modules: a targeting module specifically targets cells; a sensing module senses and transports raw materials that are present in their vicinity; a biosynthesis module converts raw materials to useful molecules, transport them back to the cell surface, and self-destructs upon completion of this sequence (self-destruct module).
Scientists at the University of Maryland have now demonstrated what was conceptualized in this earlier vision. Moreover they have added a quality that was not originally conceived - the nanofactory needs to have modalities that enable its own assembly. The scientists used the principles of synthetic biology to create the enzyme pathway that has as a part of it an assembly domain. Then, they used 'biofabrication' to assemble antibodies on to the synthesis domain, which enables targeting.
About two years ago we reported on the concept of a biological nanofactory that comprises multiple functional modules: a targeting module specifically targets cells; a sensing module senses and transports raw materials that are present in their vicinity; a biosynthesis module converts raw materials to useful molecules, transport them back to the cell surface, and self-destructs upon completion of this sequence (self-destruct module).
Scientists at the University of Maryland have now demonstrated what was conceptualized in this earlier vision. Moreover they have added a quality that was not originally conceived - the nanofactory needs to have modalities that enable its own assembly. The scientists used the principles of synthetic biology to create the enzyme pathway that has as a part of it an assembly domain. Then, they used 'biofabrication' to assemble antibodies on to the synthesis domain, which enables targeting.
Jan 29th, 2010
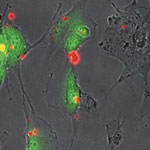 Oncogenes are genes that are associated with the development of cancer - when mutated or expressed at high levels, they can help turn a normal cell into a tumor cell. Promising new chemotherapeutic strategies have therefore focused on suppressing oncogenes. One such approach is based on RNA interference (RNAi), a technique wherein small double-stranded RNA molecules can sequence-specifically inhibit the expression of targeted oncogenes. The idea here is that with the help of small interfering RNA (siRNA), key oncogenes that modulate signaling pathways and thereby regulate the behavior of malignant tumor cells can be manipulated. To harness the full potential of this approach, the prime requirements are to deliver the siRNA molecules with high selectivity and efficiency into tumor cells and to monitor both siRNA delivery and the resulting knockdown effects at the single-cell level.
Oncogenes are genes that are associated with the development of cancer - when mutated or expressed at high levels, they can help turn a normal cell into a tumor cell. Promising new chemotherapeutic strategies have therefore focused on suppressing oncogenes. One such approach is based on RNA interference (RNAi), a technique wherein small double-stranded RNA molecules can sequence-specifically inhibit the expression of targeted oncogenes. The idea here is that with the help of small interfering RNA (siRNA), key oncogenes that modulate signaling pathways and thereby regulate the behavior of malignant tumor cells can be manipulated. To harness the full potential of this approach, the prime requirements are to deliver the siRNA molecules with high selectivity and efficiency into tumor cells and to monitor both siRNA delivery and the resulting knockdown effects at the single-cell level.
Jan 19th, 2010
 Emerging nanotechnology applications in the fields of medicine and biology often involve the use of nanoparticles for probing biological processes and structures or for constructing sophisticated nanoscale drug delivery mechanisms. Nanoparticles are already being used with dramatic success in biomedical applications. However, relatively little is known about the potential biological risks from these nanoparticle applications inside the body. The identity of nanoparticles in a biological medium, in terms of their interaction with that medium, is largely determined by the proteins that dress the particles. Since many of the toxic and therapeutic uses of nanoparticles involve the introduction of nanoparticles into the bloodstream of humans and other animals, it is particularly important to know how nanoparticles interact with blood proteins. New research performed in the Polymers Division at the National Institute of Standards and Technology (NIST) directly addresses this issue and explores the effects of nanoparticle size (5nm to 100nm) and a whole range of important blood proteins.
Emerging nanotechnology applications in the fields of medicine and biology often involve the use of nanoparticles for probing biological processes and structures or for constructing sophisticated nanoscale drug delivery mechanisms. Nanoparticles are already being used with dramatic success in biomedical applications. However, relatively little is known about the potential biological risks from these nanoparticle applications inside the body. The identity of nanoparticles in a biological medium, in terms of their interaction with that medium, is largely determined by the proteins that dress the particles. Since many of the toxic and therapeutic uses of nanoparticles involve the introduction of nanoparticles into the bloodstream of humans and other animals, it is particularly important to know how nanoparticles interact with blood proteins. New research performed in the Polymers Division at the National Institute of Standards and Technology (NIST) directly addresses this issue and explores the effects of nanoparticle size (5nm to 100nm) and a whole range of important blood proteins.
Jan 13th, 2010
 In many biomedical applications, protein nanotubes present several advantages over nanospheres. The layer-by-layer (LbL) deposition technique for the preparation of protein nanotubes has attracted considerable attention because of their potential nanotechnology applications in enzymatic nanocatalysts, bioseparation nanofilters, and targeting nanocarriers. A drawback is that in template synthesis the extraction process often results in physical deformation of the nanotubes. Researchers in Japan have now developed a new procedure using specific solvent and freeze-drying technique. They describe for the first time molecular capturing properties of protein nanotubes with a controllable affinity and size selectivity.
In many biomedical applications, protein nanotubes present several advantages over nanospheres. The layer-by-layer (LbL) deposition technique for the preparation of protein nanotubes has attracted considerable attention because of their potential nanotechnology applications in enzymatic nanocatalysts, bioseparation nanofilters, and targeting nanocarriers. A drawback is that in template synthesis the extraction process often results in physical deformation of the nanotubes. Researchers in Japan have now developed a new procedure using specific solvent and freeze-drying technique. They describe for the first time molecular capturing properties of protein nanotubes with a controllable affinity and size selectivity.
Jan 11th, 2010
 Continuing miniaturization has moved the semiconductor industry well into the nano realm with leading chip manufacturers on their way to CMOS using 22nm process technology. With transistors the size of tens of nanometers, researchers have begun to explore the interface of biology and electronics by integrating nanoelectronic components and living cells. While researchers have already experimented with integrating living cells into semiconductor materials other research is exploring the opposite way, i.e. integrating nanoelectronics into living cells. Researchers in Spain have demonstrated that silicon chips smaller than cells can be produced, collected, and internalized inside living cells by different techniques (lipofection, phagocytosis or microinjection) and, most significantly, they can be used as intracellular sensors.
Continuing miniaturization has moved the semiconductor industry well into the nano realm with leading chip manufacturers on their way to CMOS using 22nm process technology. With transistors the size of tens of nanometers, researchers have begun to explore the interface of biology and electronics by integrating nanoelectronic components and living cells. While researchers have already experimented with integrating living cells into semiconductor materials other research is exploring the opposite way, i.e. integrating nanoelectronics into living cells. Researchers in Spain have demonstrated that silicon chips smaller than cells can be produced, collected, and internalized inside living cells by different techniques (lipofection, phagocytosis or microinjection) and, most significantly, they can be used as intracellular sensors. 
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





