| Posted: Sep 13, 2011 | |
Abundant inorganic material could replace platinum in dye-sensitized solar cells |
|
| (Nanowerk Spotlight) Dye-sensitized solar cells (DSSCs) are among the most promising photovoltaic devices for low-cost light-to-energy conversion with relatively high efficiency. While the DSSC is a fairly mature design, researchers are still trying to improve its efficiency with various techniques (see for instance: "Improved design for dye-sensitized solar cells includes quantum dot antennas"). | |
| To date, the most commonly used counter electrode in DSSC is fluorine doped tin oxide (FTO) glass coated with a thin layer of platinum. However, as a noble metal, the low abundance (0.0037 ppm) and high cost (currently around $50-$60/gram) hinder platinum from being used for large-scale manufacturing. Considerable efforts have been made to replace platinum with earth-abundant low-cost alternatives, including carbon-based materials (e.g., carbon nanotube, carbon black, and graphite), conjugated polymers, and inorganic materials as counter electrode (see for instance: "An alternative to platinum counter electrode for dye-sensitized solar cells"). | |
| In comparison to carbon materials and polymers, inorganic compounds carry many advantageous characteristics, for example, simple preparation and a diversity of materials that can be used. In recent years, a variety of binary metal oxide, metal sulfide, metal nitride and metal carbide have been developed as counter electrodes. | |
| In the quest to seek alternative counter electrode materials for expensive and scarce platinum, a group of scientists has now explored the use of abundant ternary or quaternary materials as potential substitutes for platinum. | |
| "We demonstrate the first study of developing a novel platinum-free counter electrode for dye-sensitized solar cells based on low-cost quaternary copper zinc tin sulfide (CZTS) nanocrystals," Zhiqun Lin, an Associate Professor in the School of Materials Science and Engineering at the Georgia Institute of Technology, tells Nanowerk. "With a simple wet chemistry synthesis of CZTS and a viable spin-coating fabrication of electrodes, the resulting CZTS film after selenization exhibited an impressive electrocatalytic performance as counter electrodes to promote the regeneration of iodide from triiodide in electrolyte, yielding a power conversion efficiency of 7.37%, remarkably comparable to that with platinum electrodes (PCE = 7.04%)." | |
 |
|
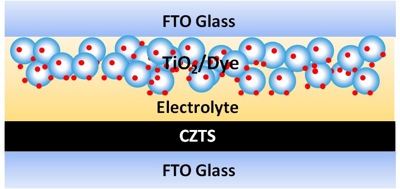
| A copper zinc tin sulfide (CZTS) semiconductor can be used as an effective counter-electrode material in place of platinum metal, yielding low-cost, high-efficiency dye-sensitized solar cells. CZTS nanocrystals were synthesized and then spin-coated on fluorine-doped tin oxide (FTO) glass. (Image: Lin Research Group, Georgia Tech College of Engineering) | |
| As the research team explains in their paper in the September 7, 2011 online edition of Angewandte Chemie International Edition ("Low-Cost Copper Zinc Tin Sulfide Counter Electrodes for High-Efficiency Dye-Sensitized Solar Cells"), the use of CZTS as counter electrode may expand the possibilities for developing low-cost and scalable DSSCs that dispose of the need for expensive and scarce platinum. | |
| "A quaternary chalcogenide semiconductor, CZTS is most widely known as one of the most promising photovoltaic materials and is widely used in thin film solar cells," explains Lin. "Notably, CZTS is composed of naturally abundant elements in the Earth's crust and has very low toxicity, i.e. it is environmentally friendly compared to two high-efficiency thin film solar cells, CdTe and CIGS that have toxic elements and rare metals." | |
| Recently, high efficiency thin-film solar cells have been demonstrated based on the superior photovoltaic performance of CZTS as a p-type semiconductor (see for instance: "High-Efficiency Solar Cell with Earth-Abundant Liquid-Processed Absorber"). However, no studies have centered on the electrocatalytic activity of CZTS for use in DSSCs. | |
| The research team from Georgia Tech, Iowa State University, and Fudan University, led by Lin, employed a solution-base synthesis approach to prepare CZTS nanocrystals. | |
| Lin says it is worth noting that "compared to conventional costly and low-throughput high-vacuum sputtering and vapor deposition of CZTS, the ability to produce a CZTS nanocrystal dispersion (that is, a nanocrystal 'ink') that can be sprayed and coated on surface and then thermally annealed into larger-grain thin film would substantially lower the manufacturing cost and allow high-throughput solar-cell production." | |
| The CZTS ink was then either spin-coated (resulting in an approx. 180 nm thick layer) or drop-cast (resulting in an approx. 2.3 µm thick layer) onto clean FTO glass and sintered at 540 °C for one hour in selenium vapor. | |
| When comparing the performance of several dye-sensitized solar cells with different counter electrodes, the team found that when their spin-coated CZTS sintered in selenium vapor (CZTSSe) was employed, the highest power conversion efficiency of 7.37% was obtained. | |
| "We expect further improvement of the photovoltaic performance as we haven't yet optimized many parameters of the counter electrode preparation (e.g., CZTS composition and fabrication method)" says Lin. "Rational treatment of the photo anode – e.g., adding a scattering layer – would further increase the efficiency of both CZTSSe-coated and platinum-coated solar cells; this is the subject of future work." | |
| He also notes that by reducing the thickness of CZTS and CZTSSe layers, and thereby reducing resistance, a further increase in the performance of solar cells may be achieved. | |
| "The initial recognition of CZTS material is its photovoltaic property" says Lin. "In this work we revealed its electrocatalytic property. We envision that combining these two properties of CZTS in a solar cell, that is, capitalizing on CZTS as both a photovoltaic material and a catalyst at the same time, may be a challenging yet promising research in the future." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
