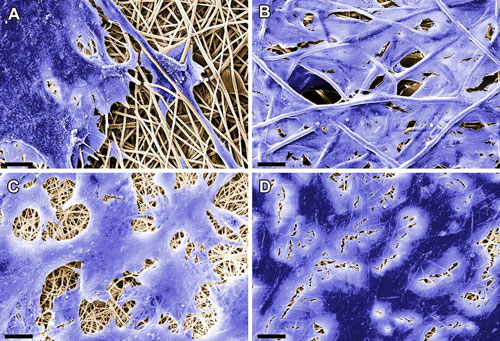
 Regenerative medicine uses cells harvested from the patient's own body to heal damaged tissue. Fraunhofer researchers have developed a cell-free substrate containing proteins to which autologous cells bind and grow only after implantation.
Regenerative medicine uses cells harvested from the patient's own body to heal damaged tissue. Fraunhofer researchers have developed a cell-free substrate containing proteins to which autologous cells bind and grow only after implantation.
Apr 1st, 2015
Read more
Researchers uncovered a new kind of synergy in the development of the nervous system, which explains an important mechanism required for neural circuits to form properly. Their breakthrough could eventually help develop tools to repair nerve cells following injuries to the nervous system (such as the brain and spinal cord).
Apr 1st, 2015
Read more
The decades worth of data that has been collected about the billions of neurons in the brain is astounding. To help scientists make sense of this 'brain big data', researchers have used data mining to create http://www.neuroelectro.org, a publicly available website that acts like Wikipedia, indexing physiological information about neurons.
Mar 30th, 2015
Read more
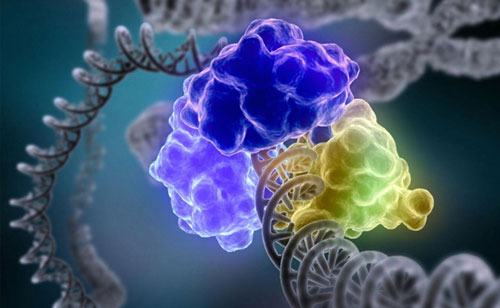 Researcher managed to clarify how DNA damage signaling works. The DNA molecule is chemically unstable giving rise to DNA lesions. That is why DNA damage detection, signaling and repair, collectively known as the DNA damage response, are needed. DNA repair consists of enzymes which find the damaged DNA and repair it. Some of them recognize the damaged bases and give signals to the other enzymes, which repair the DNA.
Researcher managed to clarify how DNA damage signaling works. The DNA molecule is chemically unstable giving rise to DNA lesions. That is why DNA damage detection, signaling and repair, collectively known as the DNA damage response, are needed. DNA repair consists of enzymes which find the damaged DNA and repair it. Some of them recognize the damaged bases and give signals to the other enzymes, which repair the DNA.
Mar 30th, 2015
Read more
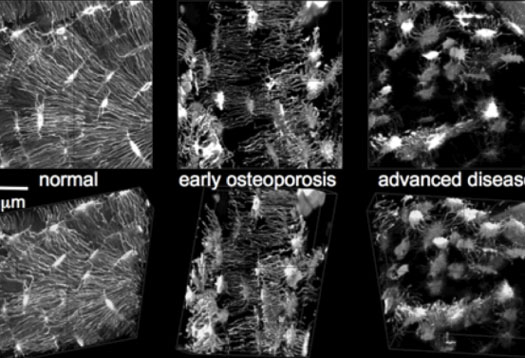 Researchers are using previously top-secret semiconductor technology to zoom through organs of the human body, down to the level of a single cell.
Researchers are using previously top-secret semiconductor technology to zoom through organs of the human body, down to the level of a single cell.
Mar 30th, 2015
Read more
Bioplastics show promise in search for antibacterial packaging.
Mar 27th, 2015
Read more
The study documents major investment opportunities to build UK excellence and leadership in translating industrial biotechnology and bioenergy research into industrial products and processes.
Mar 27th, 2015
Read more
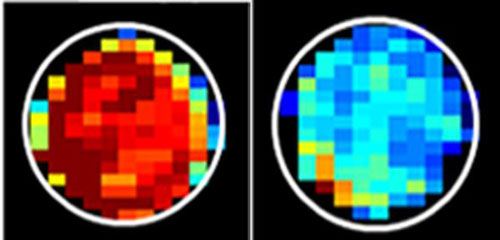 Preliminary study in lab-grown cells raises possibility of cancer diagnosis without biopsies.
Preliminary study in lab-grown cells raises possibility of cancer diagnosis without biopsies.
Mar 27th, 2015
Read more
 Using high-performance computing and genetic engineering to boost the photosynthetic efficiency of plants offers the best hope of increasing crop yields enough to feed a planet expected to have 9.5 billion people on it by 2050, researchers report in a new study.
Using high-performance computing and genetic engineering to boost the photosynthetic efficiency of plants offers the best hope of increasing crop yields enough to feed a planet expected to have 9.5 billion people on it by 2050, researchers report in a new study.
Mar 26th, 2015
Read more
A 'DNA chip card' employs electrochemical DNA chips and overcomes the complicated procedures associated with genetic testing of conventional methods.
Mar 26th, 2015
Read more
 A cross-disciplinary group of scientists is applying the tools of ecology and evolutionary biology to a new research frontier: indoor spaces.
A cross-disciplinary group of scientists is applying the tools of ecology and evolutionary biology to a new research frontier: indoor spaces.
Mar 25th, 2015
Read more
CRISPR-Cas9 is a powerful new tool for editing the genome. For researchers around the world, the CRISPR-Cas9 technique is an exciting innovation because it is faster and cheaper than previous methods.
Mar 25th, 2015
Read more
 Researchers have used a combination of metabolic engineering and directed evolution to develop a new, mutant yeast strain that could lead to a more efficient biofuel production process that would make biofuels more economically competitive with conventional fuels.
Researchers have used a combination of metabolic engineering and directed evolution to develop a new, mutant yeast strain that could lead to a more efficient biofuel production process that would make biofuels more economically competitive with conventional fuels.
Mar 24th, 2015
Read more
Researchers have developed a novel biotechnological solution for erectile dysfunction: a gene therapy that triggers reliable erections.
Mar 23rd, 2015
Read more
Researchers from the University of Copenhagen are the first in the world to develop a secure way of measuring the important protein apo-M. This could prove relevant for research into diseases such as diabetes, arteriosclerosis and sclerosis.
Mar 23rd, 2015
Read more
Scientists have successfully created 'mini-lungs' using stem cells derived from skin cells of patients with cystic fibrosis, and have shown that these can be used to test potential new drugs for this debilitating lung disease.
Mar 19th, 2015
Read more
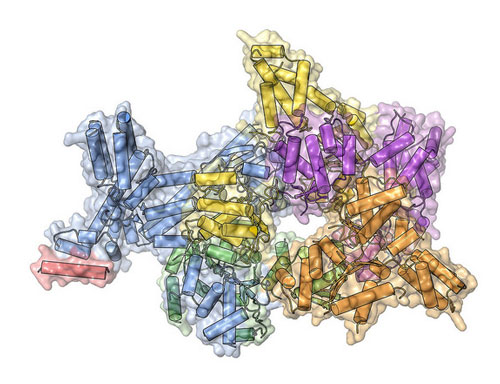 A clearer understanding of the origin recognition complex (ORC) - a protein complex that directs DNA replication - through its crystal structure offers new insight into fundamental mechanisms of DNA replication initiation. This will also provide insight into how ORC may be compromised in a subset of patients with Meier-Gorlin syndrome, a form of dwarfism in humans.
A clearer understanding of the origin recognition complex (ORC) - a protein complex that directs DNA replication - through its crystal structure offers new insight into fundamental mechanisms of DNA replication initiation. This will also provide insight into how ORC may be compromised in a subset of patients with Meier-Gorlin syndrome, a form of dwarfism in humans.
Mar 18th, 2015
Read more
Scientists develop world's first continuous purification method for valuable drugs. This will lead to significantly reduced production costs and to cheaper pharmaceuticals that are affordable for non-privileged health care systems.
Mar 18th, 2015
Read more
 Regenerative medicine uses cells harvested from the patient's own body to heal damaged tissue. Fraunhofer researchers have developed a cell-free substrate containing proteins to which autologous cells bind and grow only after implantation.
Regenerative medicine uses cells harvested from the patient's own body to heal damaged tissue. Fraunhofer researchers have developed a cell-free substrate containing proteins to which autologous cells bind and grow only after implantation.
 Subscribe to our Biotechnology News feed
Subscribe to our Biotechnology News feed





