Showing Spotlights 113 - 120 of 315 in category All (newest first):
 Supercapacitors, also called electric double layer capacitors (EDLC), store energy in two closely spaced layers with opposing charges and offer fast charge/discharge rates and the ability to sustain millions of cycles. It is frequently stated that supercapacitors bridge the gap between batteries and electrolytic ('conventional') capacitors, but contemporary devices have a lower specific energy than Li-ion batteries and are orders of magnitude slower than electrolytic capacitors. A research team has now shown that by moving from porous carbon with a network of pores inside particles as electrode material to exposed surfaces of nanostructured carbon onions of 6-7 nm diameter, it is possible to reach the discharge rate (power) of electrolytic capacitors, but with volumetric capacitance about four orders of magnitude higher. Moreover, observed discharge rates up to 200 V/second are about three orders of magnitude higher than conventional supercapacitors.
Supercapacitors, also called electric double layer capacitors (EDLC), store energy in two closely spaced layers with opposing charges and offer fast charge/discharge rates and the ability to sustain millions of cycles. It is frequently stated that supercapacitors bridge the gap between batteries and electrolytic ('conventional') capacitors, but contemporary devices have a lower specific energy than Li-ion batteries and are orders of magnitude slower than electrolytic capacitors. A research team has now shown that by moving from porous carbon with a network of pores inside particles as electrode material to exposed surfaces of nanostructured carbon onions of 6-7 nm diameter, it is possible to reach the discharge rate (power) of electrolytic capacitors, but with volumetric capacitance about four orders of magnitude higher. Moreover, observed discharge rates up to 200 V/second are about three orders of magnitude higher than conventional supercapacitors.
Aug 19th, 2010
 Bullet-proof vests are basically made from high stiffness and toughness, woven or laminated, polymeric fibers stacked in a number of layers. Upon impact of the striking bullet, the fabric material absorbs the energy by stretching of the fibers and the stiff fibers ensure that the load is dispersed over a large area throughout the material. Carbon nanotube is an ideal candidate material for bulletproof vests due to its unique combination of exceptionally high elastic modulus and high yield strain. If one compares these values with those for other fibers suitable for ballistic applications, the enormous potential of CNTs as a candidate material for bullet-proof armor system is quite evident.
Bullet-proof vests are basically made from high stiffness and toughness, woven or laminated, polymeric fibers stacked in a number of layers. Upon impact of the striking bullet, the fabric material absorbs the energy by stretching of the fibers and the stiff fibers ensure that the load is dispersed over a large area throughout the material. Carbon nanotube is an ideal candidate material for bulletproof vests due to its unique combination of exceptionally high elastic modulus and high yield strain. If one compares these values with those for other fibers suitable for ballistic applications, the enormous potential of CNTs as a candidate material for bullet-proof armor system is quite evident.
Aug 6th, 2010
 Previous studies have revealed that single-walled carbon nanotubes (SWCNTs) strongly absorb light, especially in the near-infrared region, and convert it into heat. There even has been a report that fluffy SWCNTs can burst into flames when exposed to a camera flash, which means the local temperature has reached 600-700C. This effect has already been used to develop effective CNT-based cancer killers or extremely dark materials. In a new twist, researchers in China have now discovered that SWCNT buckypapers have a large Seebeck coefficient, indicating a strong capability to convert heat into electricity. Based on this, they have designed an opto-electronic power source which converts the incident light into electricity. While this has been discussed as a theoretical mechanism, the team at Tsinghua University in Beijing has actually fabricated an integrated device that outputs a macroscopic voltage, moving forward towards practical applications.
Previous studies have revealed that single-walled carbon nanotubes (SWCNTs) strongly absorb light, especially in the near-infrared region, and convert it into heat. There even has been a report that fluffy SWCNTs can burst into flames when exposed to a camera flash, which means the local temperature has reached 600-700C. This effect has already been used to develop effective CNT-based cancer killers or extremely dark materials. In a new twist, researchers in China have now discovered that SWCNT buckypapers have a large Seebeck coefficient, indicating a strong capability to convert heat into electricity. Based on this, they have designed an opto-electronic power source which converts the incident light into electricity. While this has been discussed as a theoretical mechanism, the team at Tsinghua University in Beijing has actually fabricated an integrated device that outputs a macroscopic voltage, moving forward towards practical applications.
Jul 29th, 2010
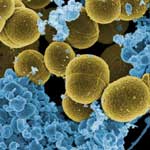 Life-threatening infectious diseases caused by antibiotic-resistant pathogens have been of great concern in both community and hospital settings. This increasing emergence of antibiotic-resistant strains of pathogens has necessitated the development of new antimicrobial surfaces and coatings. As antimicrobial surfaces have become popular in such areas as consumer products, public spaces such as schools and offices, and public transportation, the market for these coatings has quickly grown into a market worth hundreds of million of dollars. New work, by a team from Rensselaer Polytechnic Institute (RPI) has now combined the antimicrobial property of a cell lytic enzyme (lysostaphin) and the excellent properties of carbon nanotubes as an immobilization support in preparing nanocomposite paints that are highly effective against antibiotic-resistant strains of Staphylococcus aureus - methicillin-resistant Staphylococcus aureus (MRSA).
Life-threatening infectious diseases caused by antibiotic-resistant pathogens have been of great concern in both community and hospital settings. This increasing emergence of antibiotic-resistant strains of pathogens has necessitated the development of new antimicrobial surfaces and coatings. As antimicrobial surfaces have become popular in such areas as consumer products, public spaces such as schools and offices, and public transportation, the market for these coatings has quickly grown into a market worth hundreds of million of dollars. New work, by a team from Rensselaer Polytechnic Institute (RPI) has now combined the antimicrobial property of a cell lytic enzyme (lysostaphin) and the excellent properties of carbon nanotubes as an immobilization support in preparing nanocomposite paints that are highly effective against antibiotic-resistant strains of Staphylococcus aureus - methicillin-resistant Staphylococcus aureus (MRSA).
Jul 12th, 2010
 Glass fibers are a widely used reinforcing agent for many materials, from polymers to concrete. The most prominent glass fiber composite is fiberglass, a glass-reinforced plastic. The performance of the glass fiber composite over time depends on the durability of the polymer matrix and the fiber fracture behavior of the material. Since a conventional glass fiber is electrically insulating, traditionally, the monitoring for composite damage has been conducted by external sensors - a technique that degrades the mechanical properties of the material's structure and increases the cost. Researchers have therefore been working on the development of electrically conductive glass fiber plastics by adding conductive particles such as carbon blacks and carbon nanotubes to a polymer matrix. Researchers have now demonstrated a simple approach to deposit carbon nanotube networks onto glass fiber surfaces, thereby achieving semiconductive MWCNT-glass fibers.
Glass fibers are a widely used reinforcing agent for many materials, from polymers to concrete. The most prominent glass fiber composite is fiberglass, a glass-reinforced plastic. The performance of the glass fiber composite over time depends on the durability of the polymer matrix and the fiber fracture behavior of the material. Since a conventional glass fiber is electrically insulating, traditionally, the monitoring for composite damage has been conducted by external sensors - a technique that degrades the mechanical properties of the material's structure and increases the cost. Researchers have therefore been working on the development of electrically conductive glass fiber plastics by adding conductive particles such as carbon blacks and carbon nanotubes to a polymer matrix. Researchers have now demonstrated a simple approach to deposit carbon nanotube networks onto glass fiber surfaces, thereby achieving semiconductive MWCNT-glass fibers.
Jun 30th, 2010
 In a previous Nanowerk Spotlight we reported about work by a group of Chinese scientists that demonstrated that carbon nanotube sheets can act as powerful thermoacoustic loudspeakers. Moving experiments with carbon nanotube loudspeakers from air into water, researchers at the University of Texas at Dallas have now observed surprisingly high underwater sound generation efficiency using multi-walled carbon nanotubes sheets that are self-supported or attached to porous tissue. As a matter of fact, the nanotechnology speakers perform as well underwater as they do on land. The most surprising result they observed is that the carbon nanotubes immersed in water can still generate sound thermo-acoustically at frequencies 1 Hz - 100 KHz, despite the huge thermal capacity of water and its low thermal expansion.
In a previous Nanowerk Spotlight we reported about work by a group of Chinese scientists that demonstrated that carbon nanotube sheets can act as powerful thermoacoustic loudspeakers. Moving experiments with carbon nanotube loudspeakers from air into water, researchers at the University of Texas at Dallas have now observed surprisingly high underwater sound generation efficiency using multi-walled carbon nanotubes sheets that are self-supported or attached to porous tissue. As a matter of fact, the nanotechnology speakers perform as well underwater as they do on land. The most surprising result they observed is that the carbon nanotubes immersed in water can still generate sound thermo-acoustically at frequencies 1 Hz - 100 KHz, despite the huge thermal capacity of water and its low thermal expansion.
Jun 21st, 2010
 Flame retardant materials have become a major business for the chemical industry and can be found practically everywhere in modern society. Unfortunately, conventional methods for making plastic flame retardant involve a range of often very toxic chemicals. It has already been demonstrated that the flame retardancy of polymeric materials without the use of toxic chemicals could become possible thanks to the synergistic effect of nanoclay and carbon nanotubes. In a step further, researchers have now shown that the use of buckypaper is more efficient as a fire retardant in polymer composites in comparison to directly mixing carbon nanotubes into the composite matrix.
Flame retardant materials have become a major business for the chemical industry and can be found practically everywhere in modern society. Unfortunately, conventional methods for making plastic flame retardant involve a range of often very toxic chemicals. It has already been demonstrated that the flame retardancy of polymeric materials without the use of toxic chemicals could become possible thanks to the synergistic effect of nanoclay and carbon nanotubes. In a step further, researchers have now shown that the use of buckypaper is more efficient as a fire retardant in polymer composites in comparison to directly mixing carbon nanotubes into the composite matrix.
Jun 7th, 2010
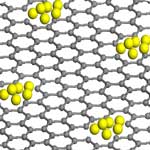 Among the various production methods for carbon nanotubes and graphene, currently only chemical vapor deposition techniques demonstrate a significant opportunity for mass production of CNT material. Using the CVD process, which is based on the catalytic action of metals, manufacturers can combine a metal catalyst such as iron with reaction gases such as hydrogen to form carbon nanotubes inside a high-temperature furnace. In order to optimize the synthesis process, a detailed understanding of the interaction between nanotubes or graphene and metal atoms is required - something that has been missing so far. Researchers in France have now shown that it is possible to create atomic-scale defects in carbon nanotubes and in graphene in preselected positions with a focused electron beam and to use these defects as trapping centers for foreign atoms.
Among the various production methods for carbon nanotubes and graphene, currently only chemical vapor deposition techniques demonstrate a significant opportunity for mass production of CNT material. Using the CVD process, which is based on the catalytic action of metals, manufacturers can combine a metal catalyst such as iron with reaction gases such as hydrogen to form carbon nanotubes inside a high-temperature furnace. In order to optimize the synthesis process, a detailed understanding of the interaction between nanotubes or graphene and metal atoms is required - something that has been missing so far. Researchers in France have now shown that it is possible to create atomic-scale defects in carbon nanotubes and in graphene in preselected positions with a focused electron beam and to use these defects as trapping centers for foreign atoms.
May 31st, 2010
 Supercapacitors, also called electric double layer capacitors (EDLC), store energy in two closely spaced layers with opposing charges and offer fast charge/discharge rates and the ability to sustain millions of cycles. It is frequently stated that supercapacitors bridge the gap between batteries and electrolytic ('conventional') capacitors, but contemporary devices have a lower specific energy than Li-ion batteries and are orders of magnitude slower than electrolytic capacitors. A research team has now shown that by moving from porous carbon with a network of pores inside particles as electrode material to exposed surfaces of nanostructured carbon onions of 6-7 nm diameter, it is possible to reach the discharge rate (power) of electrolytic capacitors, but with volumetric capacitance about four orders of magnitude higher. Moreover, observed discharge rates up to 200 V/second are about three orders of magnitude higher than conventional supercapacitors.
Supercapacitors, also called electric double layer capacitors (EDLC), store energy in two closely spaced layers with opposing charges and offer fast charge/discharge rates and the ability to sustain millions of cycles. It is frequently stated that supercapacitors bridge the gap between batteries and electrolytic ('conventional') capacitors, but contemporary devices have a lower specific energy than Li-ion batteries and are orders of magnitude slower than electrolytic capacitors. A research team has now shown that by moving from porous carbon with a network of pores inside particles as electrode material to exposed surfaces of nanostructured carbon onions of 6-7 nm diameter, it is possible to reach the discharge rate (power) of electrolytic capacitors, but with volumetric capacitance about four orders of magnitude higher. Moreover, observed discharge rates up to 200 V/second are about three orders of magnitude higher than conventional supercapacitors.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





