Showing Spotlights 1377 - 1384 of 2851 in category All (newest first):
 In the past few years, dialogues have become increasingly important for politics and science as well as scientific communication. More and more, they serve as an important feature for the responsible handling of nanotechnology at the national and European level. German speaking states have therefore laid emphasis on dialogues as a tool for communication and information in their nanotechnology action plans. The projects described in the following were largely initiated by the respective authorities as implementation measures of the national nanotechnology action plans.
In the past few years, dialogues have become increasingly important for politics and science as well as scientific communication. More and more, they serve as an important feature for the responsible handling of nanotechnology at the national and European level. German speaking states have therefore laid emphasis on dialogues as a tool for communication and information in their nanotechnology action plans. The projects described in the following were largely initiated by the respective authorities as implementation measures of the national nanotechnology action plans.
Jul 10th, 2013
 New research shows that ordered intermetallic core-shell nanocatalysts are highly promising designs for fuel cells. These are the newest members to platinum-iron alloy nanocatalysts with such intermetallic core-shell (IMCS) design. Furthermore, on characterizing them after 10,000 cycles, they still retain their structural ordering at the core while the platinum shell got thicker and thicker. Such a static core-dynamic shell (SCDS) regime is being reported for the first time.
New research shows that ordered intermetallic core-shell nanocatalysts are highly promising designs for fuel cells. These are the newest members to platinum-iron alloy nanocatalysts with such intermetallic core-shell (IMCS) design. Furthermore, on characterizing them after 10,000 cycles, they still retain their structural ordering at the core while the platinum shell got thicker and thicker. Such a static core-dynamic shell (SCDS) regime is being reported for the first time.
Jul 8th, 2013
 One of the most restricting parameters in nanofabrication is the difficulty involved with controllably patterning materials at precise locations in a repeatable manner over relatively large areas. A novel microelectromechanical system (MEMS)-based mask writer has now been developed by a team of researchers. The device allows to directly write structures at the nanoscale without the need to use photoresist, lift-off techniques or other complex and expensive approaches. The technique uses a MEMS plate with apertures drilled into it and a shutter so that one can, in effect, spray paint with atoms.
One of the most restricting parameters in nanofabrication is the difficulty involved with controllably patterning materials at precise locations in a repeatable manner over relatively large areas. A novel microelectromechanical system (MEMS)-based mask writer has now been developed by a team of researchers. The device allows to directly write structures at the nanoscale without the need to use photoresist, lift-off techniques or other complex and expensive approaches. The technique uses a MEMS plate with apertures drilled into it and a shutter so that one can, in effect, spray paint with atoms.
Jul 3rd, 2013
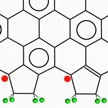 Edge magnetism is a unique property of graphene ribbons that has been predicted by theory, but not yet directly confirmed experimentally. If researchers want to utilize zigzag graphene nanoribbons (zGNRs) in spintronics, they first need to figure out a suitable termination group for zGNRs. The often used hydrogen atom termination is not a good choice since hydrogen terminated zGNRs can only be stabilized at extremely low hydrogen concentrations. In new work, researchers designed special boundaries for zGNRs that could become both stable and maintain the edge magnetism.
Edge magnetism is a unique property of graphene ribbons that has been predicted by theory, but not yet directly confirmed experimentally. If researchers want to utilize zigzag graphene nanoribbons (zGNRs) in spintronics, they first need to figure out a suitable termination group for zGNRs. The often used hydrogen atom termination is not a good choice since hydrogen terminated zGNRs can only be stabilized at extremely low hydrogen concentrations. In new work, researchers designed special boundaries for zGNRs that could become both stable and maintain the edge magnetism.
Jul 2nd, 2013
 A new publication outlines nanoscience and nanotechnology dedicated research expenditure in Europe over recent years, in particular via the 7th research framework programme (FP7). It highlights a selection of exemplary projects financed through FP7 that are representative of major research themes, such as nanoelectronics, nanophotonics, nanobiotechnology, nanomedicine, self-assembly, catalysts, industrial applications, energy and environment, tools for investing the nanoscale, health/safety/environment and communication/societal impact.
A new publication outlines nanoscience and nanotechnology dedicated research expenditure in Europe over recent years, in particular via the 7th research framework programme (FP7). It highlights a selection of exemplary projects financed through FP7 that are representative of major research themes, such as nanoelectronics, nanophotonics, nanobiotechnology, nanomedicine, self-assembly, catalysts, industrial applications, energy and environment, tools for investing the nanoscale, health/safety/environment and communication/societal impact.
Jul 1st, 2013
 Utilization of graphene may help realize innovative low power replacements for III-V materials based high electron mobility transistors while extending operational frequencies closer to the THz regime for superior wireless communications, imaging, and other novel applications. Device architectures explored to date suffer a fundamental performance roadblock due to lack of compatible deposition techniques for nanoscale dielectrics required to efficiently modulate graphene transconductance while maintaining low gate capacitance-voltage product. In new work, researchers have shown that a double layer of graphene with structural transformation to a striped channel architecture can produce high field effect mobility at a very low operation voltage.
Utilization of graphene may help realize innovative low power replacements for III-V materials based high electron mobility transistors while extending operational frequencies closer to the THz regime for superior wireless communications, imaging, and other novel applications. Device architectures explored to date suffer a fundamental performance roadblock due to lack of compatible deposition techniques for nanoscale dielectrics required to efficiently modulate graphene transconductance while maintaining low gate capacitance-voltage product. In new work, researchers have shown that a double layer of graphene with structural transformation to a striped channel architecture can produce high field effect mobility at a very low operation voltage.
Jun 26th, 2013
 In a seminal paper more than 20 years ago, scientists described a device that was capable of causing drops of water placed on it to move uphill. However, as it turned out in subsequent research, drops of water are notoriously difficult to move from where they lie, unless they are large enough to be moved by gravity. In the absence of a microtube or of a channel - as they are required by most microfluidic devices - it usually is not possible to apply the pressure needed to induce liquid movement. An alternative approach, developed by researchers in Italy, is to pattern a gradient on a surface which allows a droplet to move in order to minimize its free energy.
In a seminal paper more than 20 years ago, scientists described a device that was capable of causing drops of water placed on it to move uphill. However, as it turned out in subsequent research, drops of water are notoriously difficult to move from where they lie, unless they are large enough to be moved by gravity. In the absence of a microtube or of a channel - as they are required by most microfluidic devices - it usually is not possible to apply the pressure needed to induce liquid movement. An alternative approach, developed by researchers in Italy, is to pattern a gradient on a surface which allows a droplet to move in order to minimize its free energy.
Jun 25th, 2013
 While researchers are working on developing more cost-effective nanolithographic tools such as for instance superlens lithography, one of the key problems with nanofabrication is how to generate ever-challenging patterns with high resolution - especially for 3D nanostructures - and at the same time substantially reduce the cost of the process. A novel nanolithography method is based on light scattering from nanoparticles, which can generate 3D hollow-core structures that resembles 'nano-volcanoes'. Different from traditional lithography methods that are typically based on complex systems, this process relies solely on the light interaction with a single nanoparticle. No masks and external optics are needed in this approach, and light is manipulated into the desired optical pattern solely by the colloids.
While researchers are working on developing more cost-effective nanolithographic tools such as for instance superlens lithography, one of the key problems with nanofabrication is how to generate ever-challenging patterns with high resolution - especially for 3D nanostructures - and at the same time substantially reduce the cost of the process. A novel nanolithography method is based on light scattering from nanoparticles, which can generate 3D hollow-core structures that resembles 'nano-volcanoes'. Different from traditional lithography methods that are typically based on complex systems, this process relies solely on the light interaction with a single nanoparticle. No masks and external optics are needed in this approach, and light is manipulated into the desired optical pattern solely by the colloids.
Jun 24th, 2013
 In the past few years, dialogues have become increasingly important for politics and science as well as scientific communication. More and more, they serve as an important feature for the responsible handling of nanotechnology at the national and European level. German speaking states have therefore laid emphasis on dialogues as a tool for communication and information in their nanotechnology action plans. The projects described in the following were largely initiated by the respective authorities as implementation measures of the national nanotechnology action plans.
In the past few years, dialogues have become increasingly important for politics and science as well as scientific communication. More and more, they serve as an important feature for the responsible handling of nanotechnology at the national and European level. German speaking states have therefore laid emphasis on dialogues as a tool for communication and information in their nanotechnology action plans. The projects described in the following were largely initiated by the respective authorities as implementation measures of the national nanotechnology action plans.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





