Showing Spotlights 1433 - 1440 of 2795 in category All (newest first):
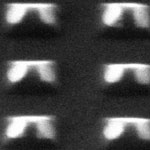 Reducing the size of photonic and electronic elements is critical for ultra-fast data processing and ultra-dense information storage. The miniaturization of a key, workhorse optical instrument - the laser - is no exception. Coherent light sources at the nanometer scale are important not only for exploring phenomena in small dimensions but also for realizing optical devices with sizes that can beat the diffraction limit of light. Researchers at Northwestern University have now found a way to manufacture single laser devices that are the size of a virus particle and that operate at room temperature. They show that subdiffraction nanoresonators based on metallic bowties, when coupled to a gain material, can generate coherent and directional light emission.
Reducing the size of photonic and electronic elements is critical for ultra-fast data processing and ultra-dense information storage. The miniaturization of a key, workhorse optical instrument - the laser - is no exception. Coherent light sources at the nanometer scale are important not only for exploring phenomena in small dimensions but also for realizing optical devices with sizes that can beat the diffraction limit of light. Researchers at Northwestern University have now found a way to manufacture single laser devices that are the size of a virus particle and that operate at room temperature. They show that subdiffraction nanoresonators based on metallic bowties, when coupled to a gain material, can generate coherent and directional light emission.
Oct 4th, 2012
 The unique energy band structure in graphene allows it to actively respond to photons with ultra-wide spectrum range - from the visible to the infrared - with record strong inter-band transition efficiency. As a consequence, graphene based ultra-fast photonics has been rising fast in various aspects of ultra-fast photonics - an ultra-fast graphene photo-detector with bandwidth exceeding 500 GHz; a broadband graphene optical modulator; a broadband graphene polarizer etc, which all benefit from the material's broadband photonics property. Researchers have now experimentally demonstrated for the first time that graphene, besides its well-known optical saturable absorption, also shows microwave and terahertz saturable absorption. The results lead to the expectation that graphene may show potential applications in microwave photonics.
The unique energy band structure in graphene allows it to actively respond to photons with ultra-wide spectrum range - from the visible to the infrared - with record strong inter-band transition efficiency. As a consequence, graphene based ultra-fast photonics has been rising fast in various aspects of ultra-fast photonics - an ultra-fast graphene photo-detector with bandwidth exceeding 500 GHz; a broadband graphene optical modulator; a broadband graphene polarizer etc, which all benefit from the material's broadband photonics property. Researchers have now experimentally demonstrated for the first time that graphene, besides its well-known optical saturable absorption, also shows microwave and terahertz saturable absorption. The results lead to the expectation that graphene may show potential applications in microwave photonics.
Oct 2nd, 2012
 The size of pixels is one of the key limiting features in the state of the art of holographic displays systems. Holography is a technique that enables a light field to be recorded and later reconstructed when the original light field is no longer present, due to the absence of the original objects. The resolution and field of view in these holographic systems are dictated by the size of the pixel, i.e. the smallest light scattering element. To address the limitations of current holographic systems due to their pixel size, a research team set out to use nanostructures as the smallest possible light-scattering elements for producing holograms. They harnessed the extraordinary conductive and light scattering abilities of nanotubes and patterned an array of carbon nanotubes to produce a high resolution hologram.
The size of pixels is one of the key limiting features in the state of the art of holographic displays systems. Holography is a technique that enables a light field to be recorded and later reconstructed when the original light field is no longer present, due to the absence of the original objects. The resolution and field of view in these holographic systems are dictated by the size of the pixel, i.e. the smallest light scattering element. To address the limitations of current holographic systems due to their pixel size, a research team set out to use nanostructures as the smallest possible light-scattering elements for producing holograms. They harnessed the extraordinary conductive and light scattering abilities of nanotubes and patterned an array of carbon nanotubes to produce a high resolution hologram.
Oct 1st, 2012
 It was previously thought that carbon nanotubes and other carbon nanomaterials are not well suited to make efficient solar cells. The main reason for this is that nanotubes are hard to isolate in single chiralities or in a given diameter range and only of semiconducting or metallic type, and thus it is hard to use them in a controlled way. New work has now shown that thin film solar cells made entirely out of carbon nanomaterials can achieve an efficiency similar to that of polymer solar cells at their initial research stages (a decade ago), but with much improved photostability. As a result, the use of carbon materials holds great promise towards the realization of photostable thin film solar cells.
It was previously thought that carbon nanotubes and other carbon nanomaterials are not well suited to make efficient solar cells. The main reason for this is that nanotubes are hard to isolate in single chiralities or in a given diameter range and only of semiconducting or metallic type, and thus it is hard to use them in a controlled way. New work has now shown that thin film solar cells made entirely out of carbon nanomaterials can achieve an efficiency similar to that of polymer solar cells at their initial research stages (a decade ago), but with much improved photostability. As a result, the use of carbon materials holds great promise towards the realization of photostable thin film solar cells.
Sep 27th, 2012
 One way to use engineered nanoparticles in the real world is in thin films. Such nanoparticulate films are thin layers, sometimes only a few nanometers thick, of composite materials that contain nanoparticles. These new materials have a wide range of applications in drug delivery, nanoelectronics, magnetic storage devices, sensors, or optical coating. However, most processes used to fabricate thin nanocomposite films with high nanoparticle fillings suffer from random nanoparticle agglomeration causing formation of irregularly shaped nanostructured features within the composite. Another complication arises from cracks that develop during the fabrication of the films. Researchers have now described a simple method for fabricating thick, crack-free silica nanoparticle films by subsequent deposition of thin, crack-free silica nanoparticle multilayers.
One way to use engineered nanoparticles in the real world is in thin films. Such nanoparticulate films are thin layers, sometimes only a few nanometers thick, of composite materials that contain nanoparticles. These new materials have a wide range of applications in drug delivery, nanoelectronics, magnetic storage devices, sensors, or optical coating. However, most processes used to fabricate thin nanocomposite films with high nanoparticle fillings suffer from random nanoparticle agglomeration causing formation of irregularly shaped nanostructured features within the composite. Another complication arises from cracks that develop during the fabrication of the films. Researchers have now described a simple method for fabricating thick, crack-free silica nanoparticle films by subsequent deposition of thin, crack-free silica nanoparticle multilayers.
Sep 26th, 2012
 Smart phones, tablets and other electronic gadgets have become an integral part of our daily life. To maintain the steady development to even faster and smaller devices, it is desirable to replace their slow electrical data interconnects with fast optical connections. Photonic crystals are an ideal tool for such a purpose as they can guide and bend light on a nanometer scale. Surprisingly, researchers have so far been unable to truly probe inside these crystals how the light intensity is distributed - this, however, is a requirement for being able to accurately characterize the local density of electromagnetic states inside the crystal which is the key for controlling the interaction of light with matter. Researchers have now demonstrated a new concept to measure the intensity distribution of light inside photonic crystals. This method, for the first time, allows researchers to map the absolute strength of an electromagnetic field component inside a photonic crystal.
Smart phones, tablets and other electronic gadgets have become an integral part of our daily life. To maintain the steady development to even faster and smaller devices, it is desirable to replace their slow electrical data interconnects with fast optical connections. Photonic crystals are an ideal tool for such a purpose as they can guide and bend light on a nanometer scale. Surprisingly, researchers have so far been unable to truly probe inside these crystals how the light intensity is distributed - this, however, is a requirement for being able to accurately characterize the local density of electromagnetic states inside the crystal which is the key for controlling the interaction of light with matter. Researchers have now demonstrated a new concept to measure the intensity distribution of light inside photonic crystals. This method, for the first time, allows researchers to map the absolute strength of an electromagnetic field component inside a photonic crystal.
Sep 25th, 2012
 The remarkable properties of some natural materials have motivated many researchers to synthesize biomimetic nanocomposites that attempt to reproduce Nature's achievements and to understand the toughening and deformation mechanisms of natural nanocomposite materials. One of the best examples is nacre, the pearly internal layer of many mollusc shells. CaCO3 has long been considered to be a genuine chemical component to approximate nacre. However, only supported polycrystalline CaCO3 (calcite) films have been reported so far. With rapid advancement of nanotechnology, it would be highly desirable to synthesize freestanding CaCO3 tablet building blocks in large quantities, ideally identical or similar to those in natural nacre, using solution-based methods. Now, for the first time, scientists have devised a facile chemical method to synthesize single-crystalline CaCO3 nanotablets in large quantities and provided genuine primary building blocks for the fabrication of nacreous inorganic-organic hybrids.
The remarkable properties of some natural materials have motivated many researchers to synthesize biomimetic nanocomposites that attempt to reproduce Nature's achievements and to understand the toughening and deformation mechanisms of natural nanocomposite materials. One of the best examples is nacre, the pearly internal layer of many mollusc shells. CaCO3 has long been considered to be a genuine chemical component to approximate nacre. However, only supported polycrystalline CaCO3 (calcite) films have been reported so far. With rapid advancement of nanotechnology, it would be highly desirable to synthesize freestanding CaCO3 tablet building blocks in large quantities, ideally identical or similar to those in natural nacre, using solution-based methods. Now, for the first time, scientists have devised a facile chemical method to synthesize single-crystalline CaCO3 nanotablets in large quantities and provided genuine primary building blocks for the fabrication of nacreous inorganic-organic hybrids.
Sep 20th, 2012
 A look at what is happening in the field of artificial nanoscale motors and molecular machinery. These nanomachines could one day perform functions similar to the biological molecular motors found in living cells, things like transporting and assembling molecules, or facilitating chemical reactions by pumping protons through membranes. Researchers have reported a light-powered DNA locomotion device that is capable of autonomous and reversible motion along an oligonucleotide track. The direction of motion can be switched using different wavelengths of light. Compared with other reported DNA walkers, this new strategy not only preserves the autonomous and controllable movement but also provides a reusable track, making it feasible to reset the device after the complete trip, as observed in nature for kinesin and myosin.
A look at what is happening in the field of artificial nanoscale motors and molecular machinery. These nanomachines could one day perform functions similar to the biological molecular motors found in living cells, things like transporting and assembling molecules, or facilitating chemical reactions by pumping protons through membranes. Researchers have reported a light-powered DNA locomotion device that is capable of autonomous and reversible motion along an oligonucleotide track. The direction of motion can be switched using different wavelengths of light. Compared with other reported DNA walkers, this new strategy not only preserves the autonomous and controllable movement but also provides a reusable track, making it feasible to reset the device after the complete trip, as observed in nature for kinesin and myosin.
Sep 19th, 2012
 Reducing the size of photonic and electronic elements is critical for ultra-fast data processing and ultra-dense information storage. The miniaturization of a key, workhorse optical instrument - the laser - is no exception. Coherent light sources at the nanometer scale are important not only for exploring phenomena in small dimensions but also for realizing optical devices with sizes that can beat the diffraction limit of light. Researchers at Northwestern University have now found a way to manufacture single laser devices that are the size of a virus particle and that operate at room temperature. They show that subdiffraction nanoresonators based on metallic bowties, when coupled to a gain material, can generate coherent and directional light emission.
Reducing the size of photonic and electronic elements is critical for ultra-fast data processing and ultra-dense information storage. The miniaturization of a key, workhorse optical instrument - the laser - is no exception. Coherent light sources at the nanometer scale are important not only for exploring phenomena in small dimensions but also for realizing optical devices with sizes that can beat the diffraction limit of light. Researchers at Northwestern University have now found a way to manufacture single laser devices that are the size of a virus particle and that operate at room temperature. They show that subdiffraction nanoresonators based on metallic bowties, when coupled to a gain material, can generate coherent and directional light emission.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





