Showing Spotlights 33 - 40 of 137 in category All (newest first):
 Colloidal silver is not a health elixir and should not be taken orally. Still, dubious online resources that sell silver dispersions or explain how to synthesize colloidal silver for nutritional purposes keep propagating mystic health effects of nano-silver. Whoever considers to "treat" themselves by taking colloidal silver certainly don't know what they want to treat themselves for. They should be aware that drinking an antimicrobial agent at any effectual dosage must inevitably cause harm to innumerable bacteria that are vital to our organism - especially in the alimentary canal. Drinking colloidal silver will either be noneffective or harmful. It is not medicine.
Colloidal silver is not a health elixir and should not be taken orally. Still, dubious online resources that sell silver dispersions or explain how to synthesize colloidal silver for nutritional purposes keep propagating mystic health effects of nano-silver. Whoever considers to "treat" themselves by taking colloidal silver certainly don't know what they want to treat themselves for. They should be aware that drinking an antimicrobial agent at any effectual dosage must inevitably cause harm to innumerable bacteria that are vital to our organism - especially in the alimentary canal. Drinking colloidal silver will either be noneffective or harmful. It is not medicine.
Nov 4th, 2011
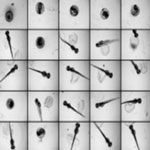 With the mass production of engineered nanoparticles, risk assessment efforts are in need of platforms that offer predictive value to human health and environment, and also possess high throughput screening capacity. Scientists, when turning to a model-organism to help answer genetic questions that cannot be easily addressed in humans, often chose the zebrafish. However, the current screening process in zebrafish involves mostly counting the survival rate, hatching and developmental abnormalities etc. through visual examination of each embryo and/or larvae under a dissecting microscope. Such process is time-consuming, labor-intensive and has limitations on data acquisition as well as statistics analysis. Researchers have now successfully demonstrated two high content imaging platforms to enhance the ability to screen the toxicological effects of nanoparticles in zebrafish embryos.
With the mass production of engineered nanoparticles, risk assessment efforts are in need of platforms that offer predictive value to human health and environment, and also possess high throughput screening capacity. Scientists, when turning to a model-organism to help answer genetic questions that cannot be easily addressed in humans, often chose the zebrafish. However, the current screening process in zebrafish involves mostly counting the survival rate, hatching and developmental abnormalities etc. through visual examination of each embryo and/or larvae under a dissecting microscope. Such process is time-consuming, labor-intensive and has limitations on data acquisition as well as statistics analysis. Researchers have now successfully demonstrated two high content imaging platforms to enhance the ability to screen the toxicological effects of nanoparticles in zebrafish embryos.
Sep 9th, 2011
 Until more information becomes available on the mechanisms underlying nanomaterial toxicity, it is uncertain what measurement technique should be used to monitor exposures in the workplace. Many of the sampling techniques that are available for measuring airborne nano aerosols vary in complexity but can provide useful information for evaluating occupational exposures with respect to particle size, mass, surface area, number concentration, and composition. Unfortunately, relatively few of these techniques are readily applicable to routine exposure monitoring. That's why researchers have now developed a unique new sampler design that collects nanoparticles separately from larger particles in a way that mimics the respiratory system.
Until more information becomes available on the mechanisms underlying nanomaterial toxicity, it is uncertain what measurement technique should be used to monitor exposures in the workplace. Many of the sampling techniques that are available for measuring airborne nano aerosols vary in complexity but can provide useful information for evaluating occupational exposures with respect to particle size, mass, surface area, number concentration, and composition. Unfortunately, relatively few of these techniques are readily applicable to routine exposure monitoring. That's why researchers have now developed a unique new sampler design that collects nanoparticles separately from larger particles in a way that mimics the respiratory system.
Aug 4th, 2011
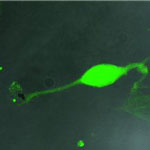 There is a growing body of research on using carbon nanotubes (CNTs) and other nanomaterials in neural engineering. Scientists are already exploring the feasibility of using CNTs to probe neural activity. With this research comes the need to develop a unified approach when assessing the toxicity of CNT in neurons. However, a complex picture emerges from the reported data: is it feasible to develop CNT-based devices as drug delivery vectors? Ultimately, are soluble CNT neurotoxic, and, if yes, to what degree? Given the often conflicting results of research reports on the biocompatibility of soluble CNT when administered to neurons in the central nervous system, a review article helps to clarify which aspects (technical or methodological) of these studies may be responsible for their heterogeneous conclusions.
There is a growing body of research on using carbon nanotubes (CNTs) and other nanomaterials in neural engineering. Scientists are already exploring the feasibility of using CNTs to probe neural activity. With this research comes the need to develop a unified approach when assessing the toxicity of CNT in neurons. However, a complex picture emerges from the reported data: is it feasible to develop CNT-based devices as drug delivery vectors? Ultimately, are soluble CNT neurotoxic, and, if yes, to what degree? Given the often conflicting results of research reports on the biocompatibility of soluble CNT when administered to neurons in the central nervous system, a review article helps to clarify which aspects (technical or methodological) of these studies may be responsible for their heterogeneous conclusions.
Jul 14th, 2011
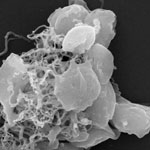 Blood platelets are the structural and chemical foundation of blood clotting and they play a vital role in minor injuries when coagulation prevents the loss of blood at the injury site. If the proper function of these platelets gets disturbed, blood clotting can lead to thrombosis, which is a leading cause of death and disability in the developed world. In view of the rapid development of nanotechnology, the impact of the newly engineered nanomaterials as an additional thrombosis risk factor is not yet known but should not be underestimated. In fact, it has been reported that carbon nanotubes induce platelet aggregation and potentiate arterial thrombosis in animal model. However, a mechanism of thrombogenic effects of carbon nanotubes was not known. Researchers have now shown that show the molecular mechanism of carbon nanotubes' induced platelets activation.
Blood platelets are the structural and chemical foundation of blood clotting and they play a vital role in minor injuries when coagulation prevents the loss of blood at the injury site. If the proper function of these platelets gets disturbed, blood clotting can lead to thrombosis, which is a leading cause of death and disability in the developed world. In view of the rapid development of nanotechnology, the impact of the newly engineered nanomaterials as an additional thrombosis risk factor is not yet known but should not be underestimated. In fact, it has been reported that carbon nanotubes induce platelet aggregation and potentiate arterial thrombosis in animal model. However, a mechanism of thrombogenic effects of carbon nanotubes was not known. Researchers have now shown that show the molecular mechanism of carbon nanotubes' induced platelets activation.
Jul 7th, 2011
 Engineered nanomaterials present regulators with a conundrum - there is a gut feeling that these materials present a new regulatory challenge, yet the nature and resolution of this challenge remains elusive. But as the debate over the regulation of nanomaterials continues, there are worrying signs that discussions are being driven less by the science of how these materials might cause harm, and more by the politics of confusion and uncertainty. Yet the more we learn about how materials interact with biology, the less clear it becomes where the boundaries of this class of materials called "nanomaterials" lie, or even whether this is a legitimate class of material at all from a regulatory perspective.
Engineered nanomaterials present regulators with a conundrum - there is a gut feeling that these materials present a new regulatory challenge, yet the nature and resolution of this challenge remains elusive. But as the debate over the regulation of nanomaterials continues, there are worrying signs that discussions are being driven less by the science of how these materials might cause harm, and more by the politics of confusion and uncertainty. Yet the more we learn about how materials interact with biology, the less clear it becomes where the boundaries of this class of materials called "nanomaterials" lie, or even whether this is a legitimate class of material at all from a regulatory perspective.
Apr 15th, 2011
 Life cycle assessment is an essential tool for ensuring the safe, responsible, and sustainable commercialization of a new technology. With missing data about the large scale impact of nanotechnology, life cycle assessments of potential nanoproducts should form an integral part of nanotechnology research at early stages of decision making as it can help in the screening of different process alternatives. Part of any meaningful results from a life cycle assessment is the total quantity of the material under investigation. Especially exposure assessments often begin with estimates based on total amounts of a material produced with the assumption that some fraction of the material in question will ultimately released to the environment. As it turns out, nobody - no research institution, no government agency, no industry association - knows even vaguely how much nanomaterials are manufactured today.
Life cycle assessment is an essential tool for ensuring the safe, responsible, and sustainable commercialization of a new technology. With missing data about the large scale impact of nanotechnology, life cycle assessments of potential nanoproducts should form an integral part of nanotechnology research at early stages of decision making as it can help in the screening of different process alternatives. Part of any meaningful results from a life cycle assessment is the total quantity of the material under investigation. Especially exposure assessments often begin with estimates based on total amounts of a material produced with the assumption that some fraction of the material in question will ultimately released to the environment. As it turns out, nobody - no research institution, no government agency, no industry association - knows even vaguely how much nanomaterials are manufactured today.
Apr 11th, 2011
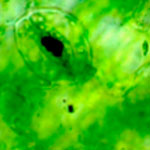 At the core of research efforts to determine the impact of synthetic nanoparticles on the environment and living systems is a fundamental understanding of the interactions between man-made nanoparticles and natural living systems that have evolved over millions of years. To describe nanoparticles at large, it may be beneficial to acknowledge that 1) biological systems are part of the food chain and therefore an essential component of the ecosystems and 2) collaborations are essential for such interdisciplinary research. Researchers have now presented a biophysical perspective that describes the fate of nanoparticles in both the aqueous phase and in living systems.
At the core of research efforts to determine the impact of synthetic nanoparticles on the environment and living systems is a fundamental understanding of the interactions between man-made nanoparticles and natural living systems that have evolved over millions of years. To describe nanoparticles at large, it may be beneficial to acknowledge that 1) biological systems are part of the food chain and therefore an essential component of the ecosystems and 2) collaborations are essential for such interdisciplinary research. Researchers have now presented a biophysical perspective that describes the fate of nanoparticles in both the aqueous phase and in living systems.
Mar 16th, 2011
 Colloidal silver is not a health elixir and should not be taken orally. Still, dubious online resources that sell silver dispersions or explain how to synthesize colloidal silver for nutritional purposes keep propagating mystic health effects of nano-silver. Whoever considers to "treat" themselves by taking colloidal silver certainly don't know what they want to treat themselves for. They should be aware that drinking an antimicrobial agent at any effectual dosage must inevitably cause harm to innumerable bacteria that are vital to our organism - especially in the alimentary canal. Drinking colloidal silver will either be noneffective or harmful. It is not medicine.
Colloidal silver is not a health elixir and should not be taken orally. Still, dubious online resources that sell silver dispersions or explain how to synthesize colloidal silver for nutritional purposes keep propagating mystic health effects of nano-silver. Whoever considers to "treat" themselves by taking colloidal silver certainly don't know what they want to treat themselves for. They should be aware that drinking an antimicrobial agent at any effectual dosage must inevitably cause harm to innumerable bacteria that are vital to our organism - especially in the alimentary canal. Drinking colloidal silver will either be noneffective or harmful. It is not medicine.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





