| Posted: Aug 16, 2018 | |
Novel tissue engineering scaffolds fabricated via controlled ice crystallization |
|
| (Nanowerk Spotlight) A large number of people around the world suffer from chronic skin wounds each year. Often, chronic wounds such as skin ulcers are seen in older people suffering from circulation disorders and diabetic patients whose skin tissue has a poor capacity of regeneration. Currently, many treatment approaches focus primarily on managing the wounds. | |
| Researchers have now taken a nanotechnology-based tissue engineering approach to accelerate the regeneration and repair of damaged tissues at the wound site by directing cells and tissues to grow towards the target site. Their hope is that this leads to the development of affordable and functional biodegradable wound dressings for accelerated healing of chronic skin wounds by promoting regeneration of local tissues. | |
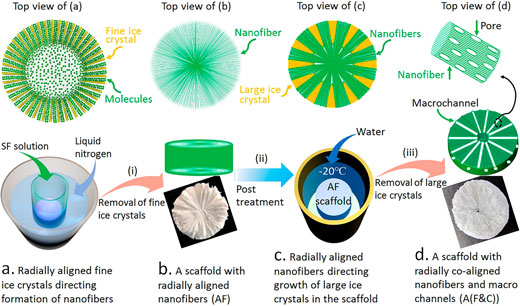
| "We show that 3D scaffolds with both aligned nanofibers and aligned interconnected macrochannels can be created with various biomacromolecules, including silk fibroin, using a facile guided ice-crystal growth and nanofiber assembly strategy," Dr. Linpeng Fan, first author of a paper on this work (ACS Nano, "Creating Biomimetic Anisotropic Architectures with Co-Aligned Nanofibers and Macrochannels by Manipulating Ice Crystallization"), tells Nanowerk. | |
| In this work, a research team at the Institute for Frontier Materials at Deakin University, Australia, developed a facile assembly strategy based on the growth of ice crystals to produce biomimetic 3D active nanofibrous scaffolds. They show that this cell- and tissue-inducing scaffold can capture and direct periphereal healthy cells and tissues to grow towards the target site and thus accelerate regeneration of damaged tissues. | |
| "So far, it has been an enormous challenge to couple aligned nanofibers with aligned macrochannels, which always go in the opposite direction," Dr. Fan points out. "We found that this can be achieved by manipulating ice crystallization in such a way that it guides the assembly of oriented nanofibers." | |
| In this process, the oriented nanofibers guide the oriented growth of large ice crystals along the fiber direction into the scaffold, which in turn leads to further assembly of the nanofibers to form aligned macrochannels in their long-axis direction. | |
 |
|
| Fabricating 3D Scaffolds with Radially Co-Aligned Nanofibers and Interconnected Macrochannels (A(F&C) Scaffolds) by a Facilely Guided Ice-Crystal Growth and Nanofiber Assembly Strategy, with Silk Fibroin (SF) as an Example. (Reprinted with permission by American Chemical Society) (click on image to enlarge) | |
| Once the aligned ice crystals in the scaffold were removed, the resulting biomimetic anisotropic 3D scaffold had both radially co-aligned nanofibers and interconnected macrochannels. | |
| "More importantly" adds Dr. Fan, "the scaffold can direct cell filtration and migration, deposition of collagen proteins of cells, as well as vascularization and 3D neurite growth." | |
| The strategy could inspire the design and development of multifunctional 3D active nanofibrous scaffolds for engineering different tissues. | |
| For example, these 3D scaffolds could be used for accelerated wound healing by recruiting and directing peripheral healthy cells to grow inwards radially. | |
| This work could also inspire the development of novel multifunctional scaffolds for engineering some complex tissues such as blood vessels. | |
| The team's preliminary in vivo results demonstrate that this kind of 3D active scaffold can significantly accelerate tissue regeneration compared to traditional 3D scaffolds. | |
| "Based on this, currently, we are developing a kind of novel wound-dressings for the healing of chronic wounds," Dr. Fan concludes. "We hope we can produce affordable and functional wound dressings to help people who need, especially the diabetics for healing of chronic ulcers by promoting regeneration of local tissues." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
