Showing Spotlights 1361 - 1368 of 2786 in category All (newest first):
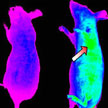 Currently developed nanotechnology-based drug delivery systems, either 'passively targeted' or 'actively targeted', do not significantly improve the delivery of drugs to target tumors. Scientists in China now propose a new-generation nanocarrier that integrates various desired functions into a single nanosystem, which can harmonize with the complex physiological environment and display different properties sequentially, thus resulting in an excellent targeting effect and satisfactory biodistribution of drugs.
Currently developed nanotechnology-based drug delivery systems, either 'passively targeted' or 'actively targeted', do not significantly improve the delivery of drugs to target tumors. Scientists in China now propose a new-generation nanocarrier that integrates various desired functions into a single nanosystem, which can harmonize with the complex physiological environment and display different properties sequentially, thus resulting in an excellent targeting effect and satisfactory biodistribution of drugs.
Mar 5th, 2013
 So-called shape memory polymers have the ability to reassume their original shape following temporary deformation. This function can be activated by means of external stimuli such as temperature change, light, or magnetic fields. Researchers have now shown that they can mold shape memory polymers into shapes relevant for micro-optics, and that they can exploit shape memory effects in this context to develop new kinds of programmable optical components. They demonstrate a series of deformable, shape-memorizing micro-optics using a shape memory elastomer.
So-called shape memory polymers have the ability to reassume their original shape following temporary deformation. This function can be activated by means of external stimuli such as temperature change, light, or magnetic fields. Researchers have now shown that they can mold shape memory polymers into shapes relevant for micro-optics, and that they can exploit shape memory effects in this context to develop new kinds of programmable optical components. They demonstrate a series of deformable, shape-memorizing micro-optics using a shape memory elastomer.
Mar 4th, 2013
 With all the rapid progress going on in research and commercialization of flexible and transparent electronics, the obvious question is not if, but when it will be possible to build a flexible and transparent truly high performance computer. A research team has now shown, for the first time, a generic batch fabrication process to obtain mechanically flexible and transparent mono-crystalline silicon (100) from bulk wafers. The researchers demonstrate a pragmatic pathway for a truly high performance computation systems on flexible and transparent platform.
With all the rapid progress going on in research and commercialization of flexible and transparent electronics, the obvious question is not if, but when it will be possible to build a flexible and transparent truly high performance computer. A research team has now shown, for the first time, a generic batch fabrication process to obtain mechanically flexible and transparent mono-crystalline silicon (100) from bulk wafers. The researchers demonstrate a pragmatic pathway for a truly high performance computation systems on flexible and transparent platform.
Mar 1st, 2013
 Harvesting unexploited energy in the living environment is increasingly becoming an intense research area as the global push to replace fossil fuels with clean and renewable energy sources heats up. There is an almost infinite number of mechanical energy sources all around us - basically, anything that moves can be harvested for energy. This ranges from the very large, like wave power in the oceans, to the very small like rain drops or biomechanical energy from heart beat, breathing, and blood flow. In an intriguing demonstration, researchers at Georgia Tech have now demonstrated that the technology offered by nanogenerators can also be used for large-scale energy harvesting.
Harvesting unexploited energy in the living environment is increasingly becoming an intense research area as the global push to replace fossil fuels with clean and renewable energy sources heats up. There is an almost infinite number of mechanical energy sources all around us - basically, anything that moves can be harvested for energy. This ranges from the very large, like wave power in the oceans, to the very small like rain drops or biomechanical energy from heart beat, breathing, and blood flow. In an intriguing demonstration, researchers at Georgia Tech have now demonstrated that the technology offered by nanogenerators can also be used for large-scale energy harvesting.
Feb 28th, 2013
 The low-frequency fluctuations in electrical current attract particular attention among researchers. The low-frequency electronic 1/f noise was first discovered in vacuum tubes, in 1925, and later observed in a wide variety of electronic materials and devices. The importance of this noise for electronic and communication devices motivated numerous studies of its physical mechanisms and methods for its control. Researchers were now able to shed light on 1/f noise origin and mechanisms using a set of multi-layered graphene samples with the thickness continuously varied from around 15 atomic planes to a single layer of graphene.
The low-frequency fluctuations in electrical current attract particular attention among researchers. The low-frequency electronic 1/f noise was first discovered in vacuum tubes, in 1925, and later observed in a wide variety of electronic materials and devices. The importance of this noise for electronic and communication devices motivated numerous studies of its physical mechanisms and methods for its control. Researchers were now able to shed light on 1/f noise origin and mechanisms using a set of multi-layered graphene samples with the thickness continuously varied from around 15 atomic planes to a single layer of graphene.
Feb 27th, 2013
 The coming age of wearable, highly flexible and transparent electronic devices will rely on essentially invisible electronic and optoelectronic circuits. In order to have close to invisible circuitry, one must have optically transparent thin-film transistors. In order to have flexibility, one needs bendable substrates. Researchers have now now fabricated transistors on specially designed nanopaper. They show that flexible organic field-effect transistors (OFETs) with high transparency and excellent mechanical properties can be fabricated on tailored nanopapers.
The coming age of wearable, highly flexible and transparent electronic devices will rely on essentially invisible electronic and optoelectronic circuits. In order to have close to invisible circuitry, one must have optically transparent thin-film transistors. In order to have flexibility, one needs bendable substrates. Researchers have now now fabricated transistors on specially designed nanopaper. They show that flexible organic field-effect transistors (OFETs) with high transparency and excellent mechanical properties can be fabricated on tailored nanopapers.
Feb 21st, 2013
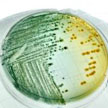 A bacterium which causes disease reacts to the antibiotics used as treatment by becoming resistant to them, sooner or later. This natural process of adaptation, antimicrobial resistance, means that the effective lifespan of antibiotics is limited. Unnecessary use and inappropriate use of antibiotics favors the emergence and spread of resistant bacteria. New research uses a graphene-based photothermal agent to trap and kill bacteria.
A bacterium which causes disease reacts to the antibiotics used as treatment by becoming resistant to them, sooner or later. This natural process of adaptation, antimicrobial resistance, means that the effective lifespan of antibiotics is limited. Unnecessary use and inappropriate use of antibiotics favors the emergence and spread of resistant bacteria. New research uses a graphene-based photothermal agent to trap and kill bacteria.
Feb 19th, 2013
 The purpose of the emerging field of nanotoxicity is to recognize and evaluate the hazards and risks of engineered nanomaterials and evaluate safety. Today, we don't even know what the impact of most chemicals is, and that includes products that have been produced by industry for many years. Nevertheless, a general understanding about nanotoxicity is slowly emerging as the body of research on cytotoxicity, genotoxicity, and ecotoxicity of nanomaterials grows. A new review summarizes and discusses recent reports derived from cell lines or animal models concerning the effects of nanomaterials on, and their application in, the endocrine system of mammalian and other species.
The purpose of the emerging field of nanotoxicity is to recognize and evaluate the hazards and risks of engineered nanomaterials and evaluate safety. Today, we don't even know what the impact of most chemicals is, and that includes products that have been produced by industry for many years. Nevertheless, a general understanding about nanotoxicity is slowly emerging as the body of research on cytotoxicity, genotoxicity, and ecotoxicity of nanomaterials grows. A new review summarizes and discusses recent reports derived from cell lines or animal models concerning the effects of nanomaterials on, and their application in, the endocrine system of mammalian and other species.
Feb 18th, 2013
 Currently developed nanotechnology-based drug delivery systems, either 'passively targeted' or 'actively targeted', do not significantly improve the delivery of drugs to target tumors. Scientists in China now propose a new-generation nanocarrier that integrates various desired functions into a single nanosystem, which can harmonize with the complex physiological environment and display different properties sequentially, thus resulting in an excellent targeting effect and satisfactory biodistribution of drugs.
Currently developed nanotechnology-based drug delivery systems, either 'passively targeted' or 'actively targeted', do not significantly improve the delivery of drugs to target tumors. Scientists in China now propose a new-generation nanocarrier that integrates various desired functions into a single nanosystem, which can harmonize with the complex physiological environment and display different properties sequentially, thus resulting in an excellent targeting effect and satisfactory biodistribution of drugs.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





