| Posted: Mar 05, 2013 | |
Toward next-generation nanomedicines for cancer therapy |
|
| (Nanowerk Spotlight) The ultimate goal of drug delivery, especially with regard to cancer therapy, is to ferry most of the administered drug to the target, while eliminating or minimizing the accumulation of the drug at any non-target tissues. Nanomedicine applications with targeted nanoparticles are expected to revolutionize cancer therapy. The use of such nanoparticles to deliver therapeutic agents is currently being studied as a promising method by which drugs can be effectively targeted to specific cells in the body, such as tumor cells. | |
| Already, almost 250 nanomedicine products approved or in clinical study and the majority of current commercial applications of nanotechnology to medicine is geared towards drug delivery to enable new modes of action, as well as better targeting and bioavailability of existing medicinal substances. | |
| "However, the currently developed nanotechnology-based drug delivery systems, either 'passively targeted' or 'actively targeted', do not really improve the delivery of drugs to target tumors," Hong Tan, a professor at the College of Polymer Science and Engineering at Sichuan University, tells Nanowerk. "Therefore, we propose a new-generation nanocarrier that integrates various desired functions into a single nanosystem, which can harmonize with the complex physiological environment and display different properties sequentially, thus resulting in an excellent targeting effect and satisfactory biodistribution of drugs." | |
| Tan and his team, together with collaborators from the university's Laboratory Animal Center and the Ningbo Institute of Material Technology and Engineering, have designed and prepared an intelligent nanoscale delivery system based on multifunctional multiblock polyurethanes for targeted intracellular delivery of the anticancer drug paclitaxel (PTX) into tumors. | |
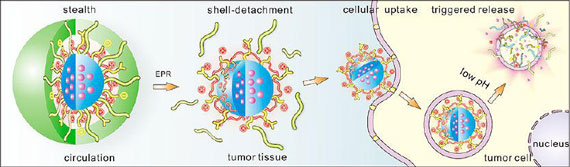
| Tan explains that these nanocarriers demonstrate a variety of attractive properties in a smart fashion, such as stealth character and long circulation, active targeting, pH-dependent shell-detachment on arriving at tumor site, improved cellular internalization, and triggered intracellular drug release in response to acidity within tumor cells. | |
| The researchers reported their findings in the February 14, 2013 online edition of ACS Nano ("Toward the Next-Generation Nanomedicines: Design of Multifunctional Multiblock Polyurethanes for Effective Cancer Treatment"). | |
 |
|
| Schematic mechanism of targeted intracellular delivery of anticancer drugs into tumor cells using multifunctional multiblock polyurethane nanocarriers. | |
| According to the team, recent progress in nanoparticle engineering has improved drug targeting to some extent, but their use has not always translated into improved clinical outcomes. This is largely due to the fact that current target/multifunctional nanoparticles, regardless of how advanced they are, reach the target as a result of blood circulation, just like conventional drug delivery systems do. | |
| "The traditional 'active targeting' approaches using antibodies or other active ligands on the particle surface do not really improve the accumulation of drugs into the target, because a large portion of the nanoparticles are taken up by the reticuloendothelial system (RES)," explains Tan. "Moreover, even if nanoparticles with prolonged circulation time can reach the tumor site by the enhanced permeation and retention (EPR) effect, the dense extracellular matrix and elevated interstitial pressure of tumors make drug penetration more difficult than in normal tissues, further leading to the clearance of nanoparticles from tumor sites." | |
| Motivated by this rationale, Tan and his collaborators incorporated the targeting antibody molecules and tumor penetrating quaternary ammonium (GQA) cationic groups into the inner shell of multilayered polyurethane nanomicelles. | |
| These active ligands can be screened and protected by PEG outer corona during their circulation in blood in order to prevent RES uptake. When exposed to the acidic tumor environment, they detach to maximize the targeting effect and intracellular delivery of antitumor drugs. | |
| "Such an intelligent nanovehicle can significantly improve the biodistribution of drugs through which the drug concentration in tumor tissue can be greatly increased while those in other tissues – e.g., liver, spleen – are significantly decreased," notes Tan. | |
| The team's work changes the traditional understanding of targeted drug delivery and provides a novel concept that nanoscale drug delivery systems need to harmonize with and respond to the highly complex biological environment inside the body and display specific required properties when necessary in order to achieve improved tumor accumulation and biodistribution of therapeutics. | |
| In addition, this work by the Chinese team has also broadened the area of polyurethane applications and opens a new era in the development of biodegradable polyurethanes for next-generation nanodelivery systems. | |
| Going forward, the researchers plan to investigate the detailed mechanisms of cellular targeting and uptake of nanocarriers to understand whether each of the multiple functions will also work as well as expected in vivo. A next step then would be to introduce such multifunctional nanomedicines into the clinical domain. However, the additional complexity and cost, as well as regulatory hurdles, may make multifunctional nanomedicines difficult to be approved and commercially manufactured. | |
| Reference: "Toward the Next-Generation Nanomedicines: Design of Multifunctional Multiblock Polyurethanes for Effective Cancer Treatment" | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
