Showing Spotlights 17 - 24 of 44 in category All (newest first):
 Using quantum dots as the basis for solar cells is not a new idea, but attempts to make such devices have not yet achieved sufficiently high efficiency in converting sunlight to power. Although these performance levels are promising, all high-performing device results to date have relied on a multiple-layer-by-layer strategy for film fabrication rather than employing a single-layer deposition process. Now, though, researchers have developed a semiconductor ink with the goal of enabling the coating of large areas of solar cell substrates in a single deposition step and thereby eliminating tens of deposition steps necessary with the previous layer-by-layer method.
Using quantum dots as the basis for solar cells is not a new idea, but attempts to make such devices have not yet achieved sufficiently high efficiency in converting sunlight to power. Although these performance levels are promising, all high-performing device results to date have relied on a multiple-layer-by-layer strategy for film fabrication rather than employing a single-layer deposition process. Now, though, researchers have developed a semiconductor ink with the goal of enabling the coating of large areas of solar cell substrates in a single deposition step and thereby eliminating tens of deposition steps necessary with the previous layer-by-layer method.
Aug 26th, 2013
 Quantum dots are expected to deliver lower cost, higher energy efficiency and greater wavelength control for a wide range of products, including lamps, displays and photovoltaics. Unfortunately, the toxicity of the elements used for efficient quantum dot based LEDs is a severe drawback for many applications. Therefore, light-emitting devices which are based on the non-toxic element silicon are extraordinary promising candidates for future QD-lighting applications. Researchers have now demonstrated highly efficient and widely color-tunable silicon light-emitting diodes (SiLEDs). The emission wavelength of the devices can easily be tuned from the deep red (680 nm) down to the orange/yellow (625 nm) spectral region by simply changing the size of the used size-separated silicon nanocrystals.
Quantum dots are expected to deliver lower cost, higher energy efficiency and greater wavelength control for a wide range of products, including lamps, displays and photovoltaics. Unfortunately, the toxicity of the elements used for efficient quantum dot based LEDs is a severe drawback for many applications. Therefore, light-emitting devices which are based on the non-toxic element silicon are extraordinary promising candidates for future QD-lighting applications. Researchers have now demonstrated highly efficient and widely color-tunable silicon light-emitting diodes (SiLEDs). The emission wavelength of the devices can easily be tuned from the deep red (680 nm) down to the orange/yellow (625 nm) spectral region by simply changing the size of the used size-separated silicon nanocrystals.
Feb 8th, 2013
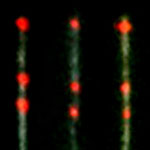 Knowing the distribution of DNA binding proteins along the genome is very informative and can tell scientists about the state of gene expression at the time of measurement. These DNA-binding proteins include transcription factors which modulate the process of transcription, various polymerases, nucleases which cleave DNA molecules, and histones which are involved in chromosome packaging in the cell nucleus. Previously, researchers demonstrated the viability of a single-molecule approach to directly visualize and map protein binding sites on DNA using fluorescent quantum dots, allowing multicolor, nanometer-resolution localization. Now, they have shown that proteins bound to DNA can be located very accurately by direct imaging. The precision of these measurement presents new opportunities for contextual genomic research on the single-molecule level.
Knowing the distribution of DNA binding proteins along the genome is very informative and can tell scientists about the state of gene expression at the time of measurement. These DNA-binding proteins include transcription factors which modulate the process of transcription, various polymerases, nucleases which cleave DNA molecules, and histones which are involved in chromosome packaging in the cell nucleus. Previously, researchers demonstrated the viability of a single-molecule approach to directly visualize and map protein binding sites on DNA using fluorescent quantum dots, allowing multicolor, nanometer-resolution localization. Now, they have shown that proteins bound to DNA can be located very accurately by direct imaging. The precision of these measurement presents new opportunities for contextual genomic research on the single-molecule level.
Mar 12th, 2012
 In January 2007, China successfully tested an Anti-satellite (ASAT) missile system by destroying their own defunct LEO satellite, which generated huge amounts of space debris. This ASAT test raised worldwide concerns about the vulnerability of satellites and other space assets and possibility of triggering an arms race in space. In order to meet emerging challenges posed by such ASAT missile systems, military strategists and researchers are developing novel technologies to protect their space assets. In view of the above, Raytheon Company has developed a counter measure system using quantum dots to protect space assets such as satellites from missile attacks. They have developed a decoy consisting of quantum dots of different sizes and shapes that are engineered to emit radiation having a radiation profile similar to that of the asset.
In January 2007, China successfully tested an Anti-satellite (ASAT) missile system by destroying their own defunct LEO satellite, which generated huge amounts of space debris. This ASAT test raised worldwide concerns about the vulnerability of satellites and other space assets and possibility of triggering an arms race in space. In order to meet emerging challenges posed by such ASAT missile systems, military strategists and researchers are developing novel technologies to protect their space assets. In view of the above, Raytheon Company has developed a counter measure system using quantum dots to protect space assets such as satellites from missile attacks. They have developed a decoy consisting of quantum dots of different sizes and shapes that are engineered to emit radiation having a radiation profile similar to that of the asset.
Jan 23rd, 2012
 A University of Ulster laboratory has found a simple, low cost and environmentally friendly way to turn common graphite flakes into bulk amounts of either high quality graphene nanosheets or quantum dots. Such structures could lead to new nanoelectronics and energy conversion technologies. The scientists discovered a simple process, which is quicker and environmentally friendlier than currently established techniques for making high quality graphene nanosheets and quantum dots at an industrial scale. The most important attribute of the produced graphene nanosheets and quantum dots compared to those reported in the literature is that they are clean from any solvent contamination and possess a low concentration of oxygen, which is inherited from the starting graphite powder.
A University of Ulster laboratory has found a simple, low cost and environmentally friendly way to turn common graphite flakes into bulk amounts of either high quality graphene nanosheets or quantum dots. Such structures could lead to new nanoelectronics and energy conversion technologies. The scientists discovered a simple process, which is quicker and environmentally friendlier than currently established techniques for making high quality graphene nanosheets and quantum dots at an industrial scale. The most important attribute of the produced graphene nanosheets and quantum dots compared to those reported in the literature is that they are clean from any solvent contamination and possess a low concentration of oxygen, which is inherited from the starting graphite powder.
Jan 18th, 2012
 Quantum dots, because they are both photoluminescent and electroluminescent and have unique physical properties, will be at the core of next-generation displays. Compared to organic luminescent materials used in organic light emitting diodes (OLEDs), QD-based materials have purer colors, longer lifetime, lower manufacturing cost, and lower power consumption. Another key advantage is that, because QDs can be deposited on virtually any substrate, you can expect printable and flexible displays of all sizes. To date, the integration of QDs into a full-color LED structure has not been possible due to the difficulty in patterning individual red-green-blue (RGB) QDs onto the pixelated display panel. Now, a Samsung team has demonstrated a novel transfer printing approach which enables fine patterning of high-quality QD films for large-area (4-inch diagonal), full-color displays mounted on glass as well as on flexible plastic substrates.
Quantum dots, because they are both photoluminescent and electroluminescent and have unique physical properties, will be at the core of next-generation displays. Compared to organic luminescent materials used in organic light emitting diodes (OLEDs), QD-based materials have purer colors, longer lifetime, lower manufacturing cost, and lower power consumption. Another key advantage is that, because QDs can be deposited on virtually any substrate, you can expect printable and flexible displays of all sizes. To date, the integration of QDs into a full-color LED structure has not been possible due to the difficulty in patterning individual red-green-blue (RGB) QDs onto the pixelated display panel. Now, a Samsung team has demonstrated a novel transfer printing approach which enables fine patterning of high-quality QD films for large-area (4-inch diagonal), full-color displays mounted on glass as well as on flexible plastic substrates.
Feb 28th, 2011
 Researchers are faced with a paradox in quantum dot-based particle/molecule tracking: on one hand, blinking is a problem for quantum dot-based tracking as it breaks up tracking trajectories; on the other hand, blinking is also a very useful indicator as it offers the best - and often only practical - in situ indication of aggregation status. Thus, when a researcher conducts a tracking experiment, s/he is annoyed by blinking but at the same time would be equally disappointed if blinking was not present to confirm single, or near single, particle status. Previous strategies to manipulate blinking dynamics involve changing the structure (surface or core chemistry) of a quantum dot. Scientists have now taken a fundamentally different approach: quantum dots employed remain unchanged.
Researchers are faced with a paradox in quantum dot-based particle/molecule tracking: on one hand, blinking is a problem for quantum dot-based tracking as it breaks up tracking trajectories; on the other hand, blinking is also a very useful indicator as it offers the best - and often only practical - in situ indication of aggregation status. Thus, when a researcher conducts a tracking experiment, s/he is annoyed by blinking but at the same time would be equally disappointed if blinking was not present to confirm single, or near single, particle status. Previous strategies to manipulate blinking dynamics involve changing the structure (surface or core chemistry) of a quantum dot. Scientists have now taken a fundamentally different approach: quantum dots employed remain unchanged.
Feb 24th, 2011
 Quantum dots have been receiving extensive attention from researchers because they can be widely used for basic physics study, quantum computing, biological imaging, nanoelectronics, and photonics applications. Current major fabrication methods for semiconductor quantum dots all have certain drawbacks. Comparing all these methods, the electrical depletion method has many advantages, such as electrical tunability by gate contacts, smooth confinement boundaries, good control and uniformity if the top gate patterns are uniform enough. Researchers have now, for the first time, applied the electrical depletion method to quantum wells and generate a large area of uniform quantum dots using a uniform metallic nanoholes array on top of quantum wells. This design for forming quantum dots has inherited all the advantages of the electrical depletion method. Furthermore, it can produce millions of uniform quantum dots easily, precisely, and controllably, which will help realize the wide applications of quantum dots in many areas.
Quantum dots have been receiving extensive attention from researchers because they can be widely used for basic physics study, quantum computing, biological imaging, nanoelectronics, and photonics applications. Current major fabrication methods for semiconductor quantum dots all have certain drawbacks. Comparing all these methods, the electrical depletion method has many advantages, such as electrical tunability by gate contacts, smooth confinement boundaries, good control and uniformity if the top gate patterns are uniform enough. Researchers have now, for the first time, applied the electrical depletion method to quantum wells and generate a large area of uniform quantum dots using a uniform metallic nanoholes array on top of quantum wells. This design for forming quantum dots has inherited all the advantages of the electrical depletion method. Furthermore, it can produce millions of uniform quantum dots easily, precisely, and controllably, which will help realize the wide applications of quantum dots in many areas.
Jul 30th, 2010
 Using quantum dots as the basis for solar cells is not a new idea, but attempts to make such devices have not yet achieved sufficiently high efficiency in converting sunlight to power. Although these performance levels are promising, all high-performing device results to date have relied on a multiple-layer-by-layer strategy for film fabrication rather than employing a single-layer deposition process. Now, though, researchers have developed a semiconductor ink with the goal of enabling the coating of large areas of solar cell substrates in a single deposition step and thereby eliminating tens of deposition steps necessary with the previous layer-by-layer method.
Using quantum dots as the basis for solar cells is not a new idea, but attempts to make such devices have not yet achieved sufficiently high efficiency in converting sunlight to power. Although these performance levels are promising, all high-performing device results to date have relied on a multiple-layer-by-layer strategy for film fabrication rather than employing a single-layer deposition process. Now, though, researchers have developed a semiconductor ink with the goal of enabling the coating of large areas of solar cell substrates in a single deposition step and thereby eliminating tens of deposition steps necessary with the previous layer-by-layer method.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





