Showing Spotlights 241 - 248 of 315 in category All (newest first):
 Granted, they don't sell them at Gap yet, but if current research undertaken by scientists in Australia is any indication, bullet-proof vests as light as T-shirts could become reality in the not too-distant future. Carbon nanotubes (CNTs) have great potential applications in making ballistic-resistance materials. The remarkable properties of CNTs makes them an ideal candidate for reinforcing polymers and other materials, and could lead to applications such as ligh-weight bullet-proof vests or shields for military vehicles and spacecraft. For these applications, thinner, lighter, and more flexible materials with superior dynamic mechanical properties are required than what is currently available. Ongoing research at the University of Sydney explores the energy absorption capacity of single-walled carbon nanotubes under a ballistic impact. CNT reinforced materials might not only be very effective in stopping ballistic penetration or high speed impact, like Kevlar vests, but they might also be able to prevent the blunt force trauma that still is a problem with today's body armor.
Granted, they don't sell them at Gap yet, but if current research undertaken by scientists in Australia is any indication, bullet-proof vests as light as T-shirts could become reality in the not too-distant future. Carbon nanotubes (CNTs) have great potential applications in making ballistic-resistance materials. The remarkable properties of CNTs makes them an ideal candidate for reinforcing polymers and other materials, and could lead to applications such as ligh-weight bullet-proof vests or shields for military vehicles and spacecraft. For these applications, thinner, lighter, and more flexible materials with superior dynamic mechanical properties are required than what is currently available. Ongoing research at the University of Sydney explores the energy absorption capacity of single-walled carbon nanotubes under a ballistic impact. CNT reinforced materials might not only be very effective in stopping ballistic penetration or high speed impact, like Kevlar vests, but they might also be able to prevent the blunt force trauma that still is a problem with today's body armor.
Nov 1st, 2007
 In our Spotlight on the issues of moving to hydrogen-powered cars (Nanotechnology could clean up the hydrogen car's dirty little secret) we briefly touched upon the problem of storing hydrogen onboard a vehicle. One gram of hydrogen gas will allow you to drive about 100 meters; unfortunately this single gram occupies almost 11 liters (2.9 gallons) of volume at room temperature and atmospheric pressure. In order to match today's cars' average reach of 400-500 kilometers per tank filling you would need to store 4 to 5 kg, or 40,000 to 50,000 liters, of hydrogen in your car. This is doable, but complicated and inconvenient, either by using intense pressure of several hundred atmospheres to store hydrogen as gas, or under cryogenic temperatures (minus 253 degrees centigrade) to store it in liquid form. Both alternatives have drawbacks. An intriguing nanotechnology approach to hydrogen storage is to encapsulate hydrogen inside hollow molecules, under room temperature. Fullerenes are ideal nanocages for this purpose, not only because they are hollow but also because hydrogen can be adsorbed on the fullerene surface. A new theoretical study provides the most accurate method to date for the structural optimization of such hydrogen-C60 composites, allowing to predict the hydrogen content in fullerene nanocages and their corresponding stability.
In our Spotlight on the issues of moving to hydrogen-powered cars (Nanotechnology could clean up the hydrogen car's dirty little secret) we briefly touched upon the problem of storing hydrogen onboard a vehicle. One gram of hydrogen gas will allow you to drive about 100 meters; unfortunately this single gram occupies almost 11 liters (2.9 gallons) of volume at room temperature and atmospheric pressure. In order to match today's cars' average reach of 400-500 kilometers per tank filling you would need to store 4 to 5 kg, or 40,000 to 50,000 liters, of hydrogen in your car. This is doable, but complicated and inconvenient, either by using intense pressure of several hundred atmospheres to store hydrogen as gas, or under cryogenic temperatures (minus 253 degrees centigrade) to store it in liquid form. Both alternatives have drawbacks. An intriguing nanotechnology approach to hydrogen storage is to encapsulate hydrogen inside hollow molecules, under room temperature. Fullerenes are ideal nanocages for this purpose, not only because they are hollow but also because hydrogen can be adsorbed on the fullerene surface. A new theoretical study provides the most accurate method to date for the structural optimization of such hydrogen-C60 composites, allowing to predict the hydrogen content in fullerene nanocages and their corresponding stability.
Oct 19th, 2007
 Modern nanotechnology researchers not only borrow extensively from nature to develop new materials and fabrication techniques, they also manage to transfer proven, and sometimes ancient, technologies into their nanotechnology laboratories. We've written about this before in our stories about welding ("Bronze Age technique works just fine in the nanotechnology era") and metal forging ("From Bronze Age shack to nanotechnology lab - metal forging techniques reach the bottom"). Today, our story deals with yet another ur-technology: spinning. Spinning is the process of creating yarn (or thread, rope, cable) from various raw fiber materials. The first spinning wheel was invented in India probably some 2,500-3,000 years ago, although some claim that the Chinese used similar devices as long as almost 5,000 years ago to spin silk threads. While spinning is one of the core technology foundations of our civilization, researchers have now begun to apply cotton-spinning techniques to fabricate carbon nanotube (CNT) "yarn."
Modern nanotechnology researchers not only borrow extensively from nature to develop new materials and fabrication techniques, they also manage to transfer proven, and sometimes ancient, technologies into their nanotechnology laboratories. We've written about this before in our stories about welding ("Bronze Age technique works just fine in the nanotechnology era") and metal forging ("From Bronze Age shack to nanotechnology lab - metal forging techniques reach the bottom"). Today, our story deals with yet another ur-technology: spinning. Spinning is the process of creating yarn (or thread, rope, cable) from various raw fiber materials. The first spinning wheel was invented in India probably some 2,500-3,000 years ago, although some claim that the Chinese used similar devices as long as almost 5,000 years ago to spin silk threads. While spinning is one of the core technology foundations of our civilization, researchers have now begun to apply cotton-spinning techniques to fabricate carbon nanotube (CNT) "yarn."
Oct 4th, 2007
 Just kidding - I always wanted to write a tabloid headline like that! In case you are expecting a story on the mysteries of crop circles caused by alien nanotechnology - stop reading right here; but the analogy is just too striking when you look at the amazing images coming out of the labs at the University of Southern California, where they developed a new technique to create three-dimensional carbon nanotube structures. While carbon nanotubes possess many exceptional properties which far exceed most known bulk materials, creating controlled nanotube (CNT) microstructures has always been a challenge. Overcoming this challenge is going to be key in developing useful and commercially viable CNT devices. Existing techniques for patterning three-dimensional CNT structures are based on the bottom-up growth of multiwalled CNTs (MWCNTs) from a patterned catalyst, which is limited to 2D-like geometries. Other, complex 3D microstructures have been fabricated with polymer-based resin materials but not with CNTs. The new technique developed by researchers in California uses a focused laser beam to selectively burn local regions of a dense forest of MWCNTs. This technique enables chemically sensitive fields to take advantage of nanotubes' exceptional properties and expand their possible applications into new areas.
Just kidding - I always wanted to write a tabloid headline like that! In case you are expecting a story on the mysteries of crop circles caused by alien nanotechnology - stop reading right here; but the analogy is just too striking when you look at the amazing images coming out of the labs at the University of Southern California, where they developed a new technique to create three-dimensional carbon nanotube structures. While carbon nanotubes possess many exceptional properties which far exceed most known bulk materials, creating controlled nanotube (CNT) microstructures has always been a challenge. Overcoming this challenge is going to be key in developing useful and commercially viable CNT devices. Existing techniques for patterning three-dimensional CNT structures are based on the bottom-up growth of multiwalled CNTs (MWCNTs) from a patterned catalyst, which is limited to 2D-like geometries. Other, complex 3D microstructures have been fabricated with polymer-based resin materials but not with CNTs. The new technique developed by researchers in California uses a focused laser beam to selectively burn local regions of a dense forest of MWCNTs. This technique enables chemically sensitive fields to take advantage of nanotubes' exceptional properties and expand their possible applications into new areas.
Sep 21st, 2007
 Firefighters and stuntmen certainly appreciate the fire resistant capabilities of modern textiles. Going far beyond such niche use, flame retardant materials have become a major business for the chemical industry and can be found practically everywhere in modern society. If you live in a country where houses are mostly built from wood (like in the U.S.; where, on the other hand, the things that used to be wood are now plastic - like christmas trees; flame retardant ones of course) most structural timber and wood elements such as paneling are treated to make them more fire resistant. Plastic materials are replacing traditional materials like wood and metal - just look at the toys you played with and the ones your kids have today. Unfortunately, the synthetic polymeric materials we group under the term 'plastic' are flammable. To decrease their flammability they require the addition of flame-retardant chemical compounds. The plastic casings, circuit boards and cables of your computers, electrical appliances or car are flame retardant. So is practically every material in airplanes, trains and ships from the fabric of seats to every kind of plastic structure found onboard. Name any plastic product and chances are it has been made flame retardant. Conventional methods for making plastic flame retardant involve a range of not exactly harmless chemicals. Improving the flame retardancy of polymeric materials without the use of toxic chemicals could now become possible thanks to the synergistic effect of carbon nanotubes and clay.
Firefighters and stuntmen certainly appreciate the fire resistant capabilities of modern textiles. Going far beyond such niche use, flame retardant materials have become a major business for the chemical industry and can be found practically everywhere in modern society. If you live in a country where houses are mostly built from wood (like in the U.S.; where, on the other hand, the things that used to be wood are now plastic - like christmas trees; flame retardant ones of course) most structural timber and wood elements such as paneling are treated to make them more fire resistant. Plastic materials are replacing traditional materials like wood and metal - just look at the toys you played with and the ones your kids have today. Unfortunately, the synthetic polymeric materials we group under the term 'plastic' are flammable. To decrease their flammability they require the addition of flame-retardant chemical compounds. The plastic casings, circuit boards and cables of your computers, electrical appliances or car are flame retardant. So is practically every material in airplanes, trains and ships from the fabric of seats to every kind of plastic structure found onboard. Name any plastic product and chances are it has been made flame retardant. Conventional methods for making plastic flame retardant involve a range of not exactly harmless chemicals. Improving the flame retardancy of polymeric materials without the use of toxic chemicals could now become possible thanks to the synergistic effect of carbon nanotubes and clay.
Aug 30th, 2007
 The fight against infections is as old as civilization. Silver, for instance, had already been recognized in ancient Greece and Rome for its infection-fighting properties and it has a long and intriguing history as an antibiotic in human health care. Modern day pharmaceutical companies developed powerful antibiotics - which also happen to be much more profitable than just plain old silver - an apparent high-tech solution to get nasty microbes such as bacteria under control. In the 1950s, penicillin was so successful that the U.S. surgeon general at the time, William H. Stewart, declared it was "time to close the book on infectious diseases, declare the war against pestilence won." Boy, was he wrong! These days, the U.S. Centers for Disease Control and Prevention (CDC) estimates that the infections acquired in hospitals alone (of all places! it's 2007 and we can't even make our hospitals safe - how scary is that?) affect approximately 2 million persons annually. In the U.S., between 44,000 and 98,000 people die every year from infections they picked up in hospitals. As our antibiotics become more and more ineffective researchers have begun to re-evaluate old antimicrobial substances such as silver. Antimicrobial nano-silver applications have become a very popular early commercial nanotechnology product. Researchers have now made a first step to add carbon nanotubes to our microbe-killing arsenal.
The fight against infections is as old as civilization. Silver, for instance, had already been recognized in ancient Greece and Rome for its infection-fighting properties and it has a long and intriguing history as an antibiotic in human health care. Modern day pharmaceutical companies developed powerful antibiotics - which also happen to be much more profitable than just plain old silver - an apparent high-tech solution to get nasty microbes such as bacteria under control. In the 1950s, penicillin was so successful that the U.S. surgeon general at the time, William H. Stewart, declared it was "time to close the book on infectious diseases, declare the war against pestilence won." Boy, was he wrong! These days, the U.S. Centers for Disease Control and Prevention (CDC) estimates that the infections acquired in hospitals alone (of all places! it's 2007 and we can't even make our hospitals safe - how scary is that?) affect approximately 2 million persons annually. In the U.S., between 44,000 and 98,000 people die every year from infections they picked up in hospitals. As our antibiotics become more and more ineffective researchers have begun to re-evaluate old antimicrobial substances such as silver. Antimicrobial nano-silver applications have become a very popular early commercial nanotechnology product. Researchers have now made a first step to add carbon nanotubes to our microbe-killing arsenal.
Aug 24th, 2007
 There you are - a romantic dinner for two, soft jazz music in the background, exquisite French cuisine served on finest porcelain. You are sniffing that 1986 Bordeaux you kept for this special occasion. The expensive floral bouquet that is the centerpiece of the table is warmly lit by several candles. "Wait!" you think, "wouldn't the soot from these candles make a great source of fluorescent carbon nanoparticles?" Or so works the mind of a nanoscientist. This is why they make great discoveries in nanotechnology while you and I just waste a few hours on dinner. Researchers actually have just demonstrated that fluorescent nanoparticles can be prepared from a common carbon source - candle soot. The whole process is so simple it could be carried out in a freshman chemical laboratory. So chances are the dinner actually was a midnight snack over pizza and diet coke, the music was Talib Kweli, and the mood was decidedly unromantic.
There you are - a romantic dinner for two, soft jazz music in the background, exquisite French cuisine served on finest porcelain. You are sniffing that 1986 Bordeaux you kept for this special occasion. The expensive floral bouquet that is the centerpiece of the table is warmly lit by several candles. "Wait!" you think, "wouldn't the soot from these candles make a great source of fluorescent carbon nanoparticles?" Or so works the mind of a nanoscientist. This is why they make great discoveries in nanotechnology while you and I just waste a few hours on dinner. Researchers actually have just demonstrated that fluorescent nanoparticles can be prepared from a common carbon source - candle soot. The whole process is so simple it could be carried out in a freshman chemical laboratory. So chances are the dinner actually was a midnight snack over pizza and diet coke, the music was Talib Kweli, and the mood was decidedly unromantic.
Aug 23rd, 2007
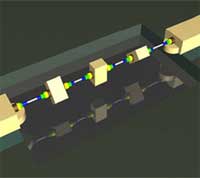 In the good old days, say 5,000 years ago, a bearing was simply the placement of tree trunks under the huge stone blocks that your worker army used to construct a pyramid. Since then, bearings have become a bit more sophisticated and are an essential part of much of today's machinery. Consequently, many kinds of bearings have been developed to suit particular purposes - sliding, rolling, fluid, or magnetic bearings, to name a few major categories. Bearings are now widely used for instance to reduce friction between shafts and axles or absorb the weight placed on moving parts and they are found in applications ranging from automobiles, trains and airplanes, computers, construction equipment, machine tools, to ceiling fans and roller skates. The same way that bearings have become an integral part of our modern world, they will also play an important role in the extremely miniaturized micro- and nanodevices of the future. Engineers will just have to come up with ingenuous ways to construct bearings at the nanoscale. Carbon nanotubes (CNTs) offer one possibility. Researchers have demonstrated that the relative displacements between the atomically smooth, nested shells in multiwalled carbon nanotubes (MWCNTs) can be used as a robust nanoscale motion-enabling mechanism. Even better, a group in Switzerland has demonstrated batch fabrication of such CNT bearings is possible.
In the good old days, say 5,000 years ago, a bearing was simply the placement of tree trunks under the huge stone blocks that your worker army used to construct a pyramid. Since then, bearings have become a bit more sophisticated and are an essential part of much of today's machinery. Consequently, many kinds of bearings have been developed to suit particular purposes - sliding, rolling, fluid, or magnetic bearings, to name a few major categories. Bearings are now widely used for instance to reduce friction between shafts and axles or absorb the weight placed on moving parts and they are found in applications ranging from automobiles, trains and airplanes, computers, construction equipment, machine tools, to ceiling fans and roller skates. The same way that bearings have become an integral part of our modern world, they will also play an important role in the extremely miniaturized micro- and nanodevices of the future. Engineers will just have to come up with ingenuous ways to construct bearings at the nanoscale. Carbon nanotubes (CNTs) offer one possibility. Researchers have demonstrated that the relative displacements between the atomically smooth, nested shells in multiwalled carbon nanotubes (MWCNTs) can be used as a robust nanoscale motion-enabling mechanism. Even better, a group in Switzerland has demonstrated batch fabrication of such CNT bearings is possible.
Aug 17th, 2007
 Granted, they don't sell them at Gap yet, but if current research undertaken by scientists in Australia is any indication, bullet-proof vests as light as T-shirts could become reality in the not too-distant future. Carbon nanotubes (CNTs) have great potential applications in making ballistic-resistance materials. The remarkable properties of CNTs makes them an ideal candidate for reinforcing polymers and other materials, and could lead to applications such as ligh-weight bullet-proof vests or shields for military vehicles and spacecraft. For these applications, thinner, lighter, and more flexible materials with superior dynamic mechanical properties are required than what is currently available. Ongoing research at the University of Sydney explores the energy absorption capacity of single-walled carbon nanotubes under a ballistic impact. CNT reinforced materials might not only be very effective in stopping ballistic penetration or high speed impact, like Kevlar vests, but they might also be able to prevent the blunt force trauma that still is a problem with today's body armor.
Granted, they don't sell them at Gap yet, but if current research undertaken by scientists in Australia is any indication, bullet-proof vests as light as T-shirts could become reality in the not too-distant future. Carbon nanotubes (CNTs) have great potential applications in making ballistic-resistance materials. The remarkable properties of CNTs makes them an ideal candidate for reinforcing polymers and other materials, and could lead to applications such as ligh-weight bullet-proof vests or shields for military vehicles and spacecraft. For these applications, thinner, lighter, and more flexible materials with superior dynamic mechanical properties are required than what is currently available. Ongoing research at the University of Sydney explores the energy absorption capacity of single-walled carbon nanotubes under a ballistic impact. CNT reinforced materials might not only be very effective in stopping ballistic penetration or high speed impact, like Kevlar vests, but they might also be able to prevent the blunt force trauma that still is a problem with today's body armor.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





