Showing Spotlights 41 - 48 of 77 in category All (newest first):
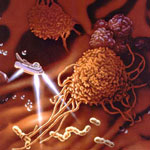 Off-targeting remains a key challenge of researchers working on nanoparticle drug delivery - the majority of intravenously administered therapeutic nanoparticles are also reaching normal tissues, resulting in considerable adverse side effects. Another challenge of nanoparticle drug delivery includes the limited penetration depth of particles into the tumors. While extensive efforts have been devoted for designing therapeutic nanoparticles, a new study - echoing the journey through the human body in Fantastic Voyage - represents the first example of coupling such drug nanocarriers with self-propelled nanoshuttles. The ability of synthetic nanomotors to carry 'cargo' has already been demonstrated; but not in connection to common drug-loaded particles. In a new study, researchers demonstrate that catalytic nanoshuttles can readily pickup common biocompatible and biodegradable drug-loaded particles and liposomes and transport them over predefined routes towards predetermined destination.
Off-targeting remains a key challenge of researchers working on nanoparticle drug delivery - the majority of intravenously administered therapeutic nanoparticles are also reaching normal tissues, resulting in considerable adverse side effects. Another challenge of nanoparticle drug delivery includes the limited penetration depth of particles into the tumors. While extensive efforts have been devoted for designing therapeutic nanoparticles, a new study - echoing the journey through the human body in Fantastic Voyage - represents the first example of coupling such drug nanocarriers with self-propelled nanoshuttles. The ability of synthetic nanomotors to carry 'cargo' has already been demonstrated; but not in connection to common drug-loaded particles. In a new study, researchers demonstrate that catalytic nanoshuttles can readily pickup common biocompatible and biodegradable drug-loaded particles and liposomes and transport them over predefined routes towards predetermined destination.
Dec 10th, 2010
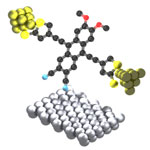 For the visionary goals of nanotechnology, functional and perhaps autonomous molecular motors will play an essential part, just like electric motors can be found in many appliances today. These nanomachines could perform functions similar to the biological molecular motors found in living cells, things like transporting and assembling molecules, or facilitating chemical reactions by pumping protons through membranes. Although applications of molecular motors are still in the future, the results of early-day studies are already spectacular: well-designed molecules or supramolecules show different kinds of motion - fueled by different driving forces such as light, heat, or chemical reactions - resulting in molecular shuttles, molecular elevators and rotating motors. A team of researchers is now proposing a conceptually new design of molecular motor based on electric field actuation and electric current detection of the rotational motion of a molecular dipole embedded in a three-terminal single-molecule device.
For the visionary goals of nanotechnology, functional and perhaps autonomous molecular motors will play an essential part, just like electric motors can be found in many appliances today. These nanomachines could perform functions similar to the biological molecular motors found in living cells, things like transporting and assembling molecules, or facilitating chemical reactions by pumping protons through membranes. Although applications of molecular motors are still in the future, the results of early-day studies are already spectacular: well-designed molecules or supramolecules show different kinds of motion - fueled by different driving forces such as light, heat, or chemical reactions - resulting in molecular shuttles, molecular elevators and rotating motors. A team of researchers is now proposing a conceptually new design of molecular motor based on electric field actuation and electric current detection of the rotational motion of a molecular dipole embedded in a three-terminal single-molecule device.
Dec 2nd, 2010
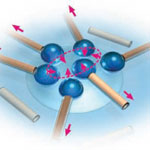 One of the (many) major challenges in getting closer to realizing visions of skillful nanomachines and ubiquitous nanofactories is the construction of synthetic nanomotors and other nanoscale propulsion systems that power these devices. At issue is not only the small scale of these systems but also the ability to precisely control their motion. Complicating the issue is that navigation principles used in the macroscale world are not applicable for nanoscale propulsion. The precise navigation of nanoscale objects is extremely challenging because of the combination of Brownian motion (random movement of particles) and low Reynolds number (where viscous forces dominate). Researchers in Germany have now demonstrated artificial water-walking devices in the form of self-powered microstriders at the air-liquid interface made of rolled-up catalytic microtubes.
One of the (many) major challenges in getting closer to realizing visions of skillful nanomachines and ubiquitous nanofactories is the construction of synthetic nanomotors and other nanoscale propulsion systems that power these devices. At issue is not only the small scale of these systems but also the ability to precisely control their motion. Complicating the issue is that navigation principles used in the macroscale world are not applicable for nanoscale propulsion. The precise navigation of nanoscale objects is extremely challenging because of the combination of Brownian motion (random movement of particles) and low Reynolds number (where viscous forces dominate). Researchers in Germany have now demonstrated artificial water-walking devices in the form of self-powered microstriders at the air-liquid interface made of rolled-up catalytic microtubes.
Sep 30th, 2010
 For nanotechnology researchers, movement at the nanoscale is a challenging problem and there is much to be learned from nature's motor systems. There are various approaches to creating self-propelled micro- and nanosized motors and one promising approach rests on catalytic conversion of chemical to mechanical energy - a process that is ubiquitous in biology, powering such important and diverse processes as cell division, skeletal muscle movement, protein synthesis, and transport of cargo within cells. Self-propelled motion of synthetic materials can be useful in applications such as bottom-up assembly of structures, pattern formation, drug delivery at specific locations, etc. Researchers have now presented a novel and versatile light-driven catalytic micromotor system, which is the cleanest and simplest of its kind.
For nanotechnology researchers, movement at the nanoscale is a challenging problem and there is much to be learned from nature's motor systems. There are various approaches to creating self-propelled micro- and nanosized motors and one promising approach rests on catalytic conversion of chemical to mechanical energy - a process that is ubiquitous in biology, powering such important and diverse processes as cell division, skeletal muscle movement, protein synthesis, and transport of cargo within cells. Self-propelled motion of synthetic materials can be useful in applications such as bottom-up assembly of structures, pattern formation, drug delivery at specific locations, etc. Researchers have now presented a novel and versatile light-driven catalytic micromotor system, which is the cleanest and simplest of its kind.
Jun 2nd, 2010
 Many nanotechnology applications are plagued by very poor wear resistance of device components at the nanoscale. Gears, bearings, and liquid lubricants can reduce friction in the macroscopic world, but the origins of friction for small devices such as micro- or nanoelectromechanical systems require other solutions. Despite the unprecedented accuracy by which these devices are nowadays designed and fabricated, their enormous surface-volume ratio leads to severe friction and wear issues, which dramatically reduce their applicability and lifetime. Although there is a significant amount of research work going on in the area of nanoscale friction, at present there is much less research conducted on nanoscale wear. Researchers have now demonstrated extremely low wear rates at the nanoscale, representing a technological breakthrough for numerous applications in emerging fields such as nanolithography, nanometrology, and nanomanufacturing.
Many nanotechnology applications are plagued by very poor wear resistance of device components at the nanoscale. Gears, bearings, and liquid lubricants can reduce friction in the macroscopic world, but the origins of friction for small devices such as micro- or nanoelectromechanical systems require other solutions. Despite the unprecedented accuracy by which these devices are nowadays designed and fabricated, their enormous surface-volume ratio leads to severe friction and wear issues, which dramatically reduce their applicability and lifetime. Although there is a significant amount of research work going on in the area of nanoscale friction, at present there is much less research conducted on nanoscale wear. Researchers have now demonstrated extremely low wear rates at the nanoscale, representing a technological breakthrough for numerous applications in emerging fields such as nanolithography, nanometrology, and nanomanufacturing.
Feb 15th, 2010
 A very promising field of nanomotor research are DNA nanomachines. These are synthetic DNA assemblies that switch between defined molecular shapes upon stimulation by external triggers. They can be controlled by a variety of methods that include pH changes and the addition of other molecular components, such as small molecule effectors, proteins and DNA strands. Researchers have now designed and built a simple DNA machine that is capable of continuous rotation with controlled speed and direction - a function that might be very useful for example for molecular transport. This machine is driven by an externally controlled electric field. When this field is oscillated between four directions, it continuously reorients a rotor DNA that is asymmetrically attached to a DNA axle.
A very promising field of nanomotor research are DNA nanomachines. These are synthetic DNA assemblies that switch between defined molecular shapes upon stimulation by external triggers. They can be controlled by a variety of methods that include pH changes and the addition of other molecular components, such as small molecule effectors, proteins and DNA strands. Researchers have now designed and built a simple DNA machine that is capable of continuous rotation with controlled speed and direction - a function that might be very useful for example for molecular transport. This machine is driven by an externally controlled electric field. When this field is oscillated between four directions, it continuously reorients a rotor DNA that is asymmetrically attached to a DNA axle.
Jan 12th, 2010
 One challenge in designing nanomachines is being able to establish how well they work and optimize their performance. This is where single molecule techniques will play an important role. With advances in nanotechnologies, it is possible to construct simple nanomachines that can perform simple functions such as opening and closing of a DNA device (e.g. DNA tweezers or DNA switches), small rotational and translational motors and energy transfer cascades. Using single-molecule techniques researchers can watch individual nanomachines working and determine the functionality of their design. Researchers in Germany now have incorporated optical addressability to these nanomachines. Hence, they can optically detect and eventually control the state of the nanodevice.
One challenge in designing nanomachines is being able to establish how well they work and optimize their performance. This is where single molecule techniques will play an important role. With advances in nanotechnologies, it is possible to construct simple nanomachines that can perform simple functions such as opening and closing of a DNA device (e.g. DNA tweezers or DNA switches), small rotational and translational motors and energy transfer cascades. Using single-molecule techniques researchers can watch individual nanomachines working and determine the functionality of their design. Researchers in Germany now have incorporated optical addressability to these nanomachines. Hence, they can optically detect and eventually control the state of the nanodevice.
Dec 23rd, 2009
 Reciprocating devices are a common part of the macroscopic world. Examples of reciprocating machines are petrol and diesel engines or a hydraulic pump. At the core of these machines is a piston and cylinder assembly where the piston executes a reciprocating motion inside the cylinder. Reciprocating motion like that in a piston has not been available in a nanoscale machine until now. Ned Seeman and his team at New York University have designed a DNA device that exhibits reciprocal motion. They have used the PX-JX2 device, a robust sequence-dependent nanomechanical DNA machine, as the basis for constructing a pair of reciprocal devices, wherein one device assumes one state, while the other device assumes the opposite state.
Reciprocating devices are a common part of the macroscopic world. Examples of reciprocating machines are petrol and diesel engines or a hydraulic pump. At the core of these machines is a piston and cylinder assembly where the piston executes a reciprocating motion inside the cylinder. Reciprocating motion like that in a piston has not been available in a nanoscale machine until now. Ned Seeman and his team at New York University have designed a DNA device that exhibits reciprocal motion. They have used the PX-JX2 device, a robust sequence-dependent nanomechanical DNA machine, as the basis for constructing a pair of reciprocal devices, wherein one device assumes one state, while the other device assumes the opposite state.
Jun 11th, 2009
 Off-targeting remains a key challenge of researchers working on nanoparticle drug delivery - the majority of intravenously administered therapeutic nanoparticles are also reaching normal tissues, resulting in considerable adverse side effects. Another challenge of nanoparticle drug delivery includes the limited penetration depth of particles into the tumors. While extensive efforts have been devoted for designing therapeutic nanoparticles, a new study - echoing the journey through the human body in Fantastic Voyage - represents the first example of coupling such drug nanocarriers with self-propelled nanoshuttles. The ability of synthetic nanomotors to carry 'cargo' has already been demonstrated; but not in connection to common drug-loaded particles. In a new study, researchers demonstrate that catalytic nanoshuttles can readily pickup common biocompatible and biodegradable drug-loaded particles and liposomes and transport them over predefined routes towards predetermined destination.
Off-targeting remains a key challenge of researchers working on nanoparticle drug delivery - the majority of intravenously administered therapeutic nanoparticles are also reaching normal tissues, resulting in considerable adverse side effects. Another challenge of nanoparticle drug delivery includes the limited penetration depth of particles into the tumors. While extensive efforts have been devoted for designing therapeutic nanoparticles, a new study - echoing the journey through the human body in Fantastic Voyage - represents the first example of coupling such drug nanocarriers with self-propelled nanoshuttles. The ability of synthetic nanomotors to carry 'cargo' has already been demonstrated; but not in connection to common drug-loaded particles. In a new study, researchers demonstrate that catalytic nanoshuttles can readily pickup common biocompatible and biodegradable drug-loaded particles and liposomes and transport them over predefined routes towards predetermined destination.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





