Showing Spotlights 57 - 64 of 316 in category All (newest first):
 The degree of competitiveness in sports has been remarkably impacted by nanotechnology like any other innovative idea in materials science. Within the niche of sport equipments, nanotechnology offers a number of advantages and immense potential to improve sporting equipments making athletes safer, comfortble and more agile than ever. Baseball bats, tennis and badminton racquets, hockey sticks, racing bicycles, golf balls/clubs, skis, fly-fishing rods, archery arrows, etc. are some of the sporting equipments, whose performance and durability are being improved with the help of nanotechnology. Nanomaterials such as carbon nanotubes, silica nanoparticles, nanoclays fullerenes, etc. are being incorporated into various sports equipment to improve the performance of athletes as well as equipments.
The degree of competitiveness in sports has been remarkably impacted by nanotechnology like any other innovative idea in materials science. Within the niche of sport equipments, nanotechnology offers a number of advantages and immense potential to improve sporting equipments making athletes safer, comfortble and more agile than ever. Baseball bats, tennis and badminton racquets, hockey sticks, racing bicycles, golf balls/clubs, skis, fly-fishing rods, archery arrows, etc. are some of the sporting equipments, whose performance and durability are being improved with the help of nanotechnology. Nanomaterials such as carbon nanotubes, silica nanoparticles, nanoclays fullerenes, etc. are being incorporated into various sports equipment to improve the performance of athletes as well as equipments.
May 27th, 2013
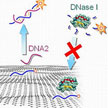 Carbon nanomaterials such as nanotubes or graphene not only are widely researched for their potential uses in industrial applications, they also are of great interest to biomedical engineers working on nanotechnology applications. These researchers found that incorporating carbon-based nanomaterials is effective not only as injectable nanoscale devices but also as components to enhance the function of existing biomaterials significantly. A recent article highlights different types of carbon-based nanomaterials currently used in biomedical applications.
Carbon nanomaterials such as nanotubes or graphene not only are widely researched for their potential uses in industrial applications, they also are of great interest to biomedical engineers working on nanotechnology applications. These researchers found that incorporating carbon-based nanomaterials is effective not only as injectable nanoscale devices but also as components to enhance the function of existing biomaterials significantly. A recent article highlights different types of carbon-based nanomaterials currently used in biomedical applications.
Apr 25th, 2013
 Carbon is the fourth-most-abundant element in the universe and, depending on the arrangements of carbon atoms, takes on a wide variety of forms, called allotropes. Carbon allotropes exhibit unique properties of strength and electrical conductivity. Solid carbon at room temperature has two classical structures: diamond and graphite. In 1985 the discovery of the existence of a third and new carbon allotrope containing sixty perfectly symmetrically arranged carbon atoms (C60) meant a major breakthrough and opened a novel field of carbon nanochemistry. Then, in 1991, carbon nanotubes were discovered and graphene in 2004. Now, a research group in China has designed a novel carbon allotrope they've named D-carbon.
Carbon is the fourth-most-abundant element in the universe and, depending on the arrangements of carbon atoms, takes on a wide variety of forms, called allotropes. Carbon allotropes exhibit unique properties of strength and electrical conductivity. Solid carbon at room temperature has two classical structures: diamond and graphite. In 1985 the discovery of the existence of a third and new carbon allotrope containing sixty perfectly symmetrically arranged carbon atoms (C60) meant a major breakthrough and opened a novel field of carbon nanochemistry. Then, in 1991, carbon nanotubes were discovered and graphene in 2004. Now, a research group in China has designed a novel carbon allotrope they've named D-carbon.
Mar 15th, 2013
 The coming age of wearable, highly flexible and transparent electronic devices will rely on essentially invisible electronic and optoelectronic circuits. In order to have close to invisible circuitry, one must have optically transparent thin-film transistors. In order to have flexibility, one needs bendable substrates. Researchers have now now fabricated transistors on specially designed nanopaper. They show that flexible organic field-effect transistors (OFETs) with high transparency and excellent mechanical properties can be fabricated on tailored nanopapers.
The coming age of wearable, highly flexible and transparent electronic devices will rely on essentially invisible electronic and optoelectronic circuits. In order to have close to invisible circuitry, one must have optically transparent thin-film transistors. In order to have flexibility, one needs bendable substrates. Researchers have now now fabricated transistors on specially designed nanopaper. They show that flexible organic field-effect transistors (OFETs) with high transparency and excellent mechanical properties can be fabricated on tailored nanopapers.
Feb 21st, 2013
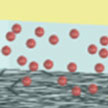 Carbon nanotubes, like the nervous cells of our brain, are excellent electrical signal conductors and can form intimate mechanical contacts with cellular membranes, thereby establishing a functional link to neuronal structures. There is a growing body of research on using nanomaterials in neural engineering. Carbon nanotube (CNT) synapse circuits are a first step in this direction. In new work, researchers at the University of California, Los Angeles, have developed a CNT synapse with the elementary dynamic logic, learning, and memory functions of a biological synapse.
Carbon nanotubes, like the nervous cells of our brain, are excellent electrical signal conductors and can form intimate mechanical contacts with cellular membranes, thereby establishing a functional link to neuronal structures. There is a growing body of research on using nanomaterials in neural engineering. Carbon nanotube (CNT) synapse circuits are a first step in this direction. In new work, researchers at the University of California, Los Angeles, have developed a CNT synapse with the elementary dynamic logic, learning, and memory functions of a biological synapse.
Feb 12th, 2013
 Materials engineers are keen to exploit the outstanding mechanical properties of carbon nanotubes (CNTs) for applications in fibers, composites, fabrics and other larger-scale structures and devices. The ability to fabricate continuous, multifunctional yarns represents an important step in this direction. The development of a continuous, weavable multilayered CNT yarn with superior mechanical, structural, surface, and electrical properties would open the way for a wide range of structural and functional applications, including composites, intelligent fabrics, catalyst supports, and sensors. Researchers in China have now found that carbon nanotubes can be self-assembled into a stable double-helix structure by a controlled over-twisting process, and this novel structure has unique mechanical properties.
Materials engineers are keen to exploit the outstanding mechanical properties of carbon nanotubes (CNTs) for applications in fibers, composites, fabrics and other larger-scale structures and devices. The ability to fabricate continuous, multifunctional yarns represents an important step in this direction. The development of a continuous, weavable multilayered CNT yarn with superior mechanical, structural, surface, and electrical properties would open the way for a wide range of structural and functional applications, including composites, intelligent fabrics, catalyst supports, and sensors. Researchers in China have now found that carbon nanotubes can be self-assembled into a stable double-helix structure by a controlled over-twisting process, and this novel structure has unique mechanical properties.
Jan 22nd, 2013
 The ideal drug carrier may be something out of science fiction. In principle, it is injected into the body and transports itself to the correct target, such as a tumor, and delivers the required dose at this target. This idealized concept was first proposed by Paul Ehrlich at the beginning of the 20th century and was nicknamed the 'magic bullet' concept. With the advent of nanotechnology and nanomedicine this dream is rapidly becoming a reality. Researchers have already demonstrated that unctionalized carbon nanotubes (CNTs) might be able to target specific cells, become ingested, and then release their contents in response to a chemical trigger. A group of researchers has now essentially achieved this goal. They have encapsulated drugs inside carbon nanotubes for drug delivery and shown that these drugs can be released 'on command' by inductive heating with an external alternating current or pulsed magnetic field.
The ideal drug carrier may be something out of science fiction. In principle, it is injected into the body and transports itself to the correct target, such as a tumor, and delivers the required dose at this target. This idealized concept was first proposed by Paul Ehrlich at the beginning of the 20th century and was nicknamed the 'magic bullet' concept. With the advent of nanotechnology and nanomedicine this dream is rapidly becoming a reality. Researchers have already demonstrated that unctionalized carbon nanotubes (CNTs) might be able to target specific cells, become ingested, and then release their contents in response to a chemical trigger. A group of researchers has now essentially achieved this goal. They have encapsulated drugs inside carbon nanotubes for drug delivery and shown that these drugs can be released 'on command' by inductive heating with an external alternating current or pulsed magnetic field.
Oct 30th, 2012
 Nitrogen-doped carbon nanotubes (CNTs) have been extensively investigated for fuel cell applications due to their excellent electrocatalytic properties. However, their biomedical applications were comparatively less investigated despite reports of their better biocompatibility. When considering carbon nanotubes for drug delivery applications, it is desirable to develop strategies that allow utilize their hollow inner cavities for maximum loading capacity. Small size and facile surface modification are also preferable with regard to their biomedical compatibility. Nitrogen-doped CNTs have been already previously demonstrated to have better biocompatibility and mitigated cytotoxicity as compared to traditional undoped pristine CNTs. Taking advantage of this, researchers used nitrogen doping of CNTs which resulted in formation of cup-shaped compartments in CNTs uniquely suitable for encapsulation. The resulting nitrogen-doped carbon nanotube cups can be corked by gold nanoparticles to form enclosed nanocapsules.
Nitrogen-doped carbon nanotubes (CNTs) have been extensively investigated for fuel cell applications due to their excellent electrocatalytic properties. However, their biomedical applications were comparatively less investigated despite reports of their better biocompatibility. When considering carbon nanotubes for drug delivery applications, it is desirable to develop strategies that allow utilize their hollow inner cavities for maximum loading capacity. Small size and facile surface modification are also preferable with regard to their biomedical compatibility. Nitrogen-doped CNTs have been already previously demonstrated to have better biocompatibility and mitigated cytotoxicity as compared to traditional undoped pristine CNTs. Taking advantage of this, researchers used nitrogen doping of CNTs which resulted in formation of cup-shaped compartments in CNTs uniquely suitable for encapsulation. The resulting nitrogen-doped carbon nanotube cups can be corked by gold nanoparticles to form enclosed nanocapsules.
Aug 2nd, 2012
 The degree of competitiveness in sports has been remarkably impacted by nanotechnology like any other innovative idea in materials science. Within the niche of sport equipments, nanotechnology offers a number of advantages and immense potential to improve sporting equipments making athletes safer, comfortble and more agile than ever. Baseball bats, tennis and badminton racquets, hockey sticks, racing bicycles, golf balls/clubs, skis, fly-fishing rods, archery arrows, etc. are some of the sporting equipments, whose performance and durability are being improved with the help of nanotechnology. Nanomaterials such as carbon nanotubes, silica nanoparticles, nanoclays fullerenes, etc. are being incorporated into various sports equipment to improve the performance of athletes as well as equipments.
The degree of competitiveness in sports has been remarkably impacted by nanotechnology like any other innovative idea in materials science. Within the niche of sport equipments, nanotechnology offers a number of advantages and immense potential to improve sporting equipments making athletes safer, comfortble and more agile than ever. Baseball bats, tennis and badminton racquets, hockey sticks, racing bicycles, golf balls/clubs, skis, fly-fishing rods, archery arrows, etc. are some of the sporting equipments, whose performance and durability are being improved with the help of nanotechnology. Nanomaterials such as carbon nanotubes, silica nanoparticles, nanoclays fullerenes, etc. are being incorporated into various sports equipment to improve the performance of athletes as well as equipments.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





