Showing Spotlights 113 - 120 of 137 in category All (newest first):
 The question if certain engineered nanoparticles are toxic, and if yes to what degree, is still one of the major issues that hasn't been properly answered yet. Most studies in the literature thus far have focused on the environmental aspects of nanoparticle toxicity, and these studies have been conducted primarily on industrial or natural/incidental nanoparticles. However, engineered nanoparticles are at the forefront of the rapidly developing field of nanomedicine; and here they are deliberately injected into the body to perform a specific medical application: fluorescent agents for imaging; drug delivery vehicles; or therapeutic agents for the destruction of cancer cells (for instance in thermolysis); just to name a few. A brand new review article provides the first comprehensive summary of the properties of engineered nanoparticles which determine their interaction with components of the immune system. It concludes that nanoparticle-based therapeutics are no more intrinsically immunotoxic than traditional pharmaceuticals, such as biotechnology-derived or small molecules. Moreover, incorporation of traditional drugs into nanotechnology formulations frequently results in a decrease in immunotoxicity compared to the native drug. Although many questions still require thorough investigation, the available data suggest that nanoparticles can be engineered to become the next generation of biocompatible drug delivery platforms.
The question if certain engineered nanoparticles are toxic, and if yes to what degree, is still one of the major issues that hasn't been properly answered yet. Most studies in the literature thus far have focused on the environmental aspects of nanoparticle toxicity, and these studies have been conducted primarily on industrial or natural/incidental nanoparticles. However, engineered nanoparticles are at the forefront of the rapidly developing field of nanomedicine; and here they are deliberately injected into the body to perform a specific medical application: fluorescent agents for imaging; drug delivery vehicles; or therapeutic agents for the destruction of cancer cells (for instance in thermolysis); just to name a few. A brand new review article provides the first comprehensive summary of the properties of engineered nanoparticles which determine their interaction with components of the immune system. It concludes that nanoparticle-based therapeutics are no more intrinsically immunotoxic than traditional pharmaceuticals, such as biotechnology-derived or small molecules. Moreover, incorporation of traditional drugs into nanotechnology formulations frequently results in a decrease in immunotoxicity compared to the native drug. Although many questions still require thorough investigation, the available data suggest that nanoparticles can be engineered to become the next generation of biocompatible drug delivery platforms.
Aug 2nd, 2007
 A quantum dot (QD), also called a nanocrystal, is a semiconductor nanostructure that can be as small as 2 to 10 nm. The usefulness of quantum dots comes from their peak emission frequency's extreme sensitivity - quantum mechanical in nature - to both the dot's size and composition. QDs have been touted as possible replacements for organic dyes in the imaging of biological systems, due to their excellent fluorescent properties, good chemical stability, broad excitation ranges and high photobleaching thresholds. However, the main drawback of QDs is their toxicity and therefore their application is problematic. If this toxicity problem could be addressed, QDs may one day be safely utilized in many areas. For instance, cadmium telluride (CdTe - which is toxic) QD based nanocomposites can be used as fluorescent probes for biological imaging, they can also be utilized to monitor targeted drug delivery and for controlled modification of structural and functional properties of intracellular components. Scientists in Ireland have been using gelatin during the production of CdTe QDs thereby reducing the toxicity of the particles. Their approach could be useful for the development of other nanoparticle composites with low toxicity as well.
A quantum dot (QD), also called a nanocrystal, is a semiconductor nanostructure that can be as small as 2 to 10 nm. The usefulness of quantum dots comes from their peak emission frequency's extreme sensitivity - quantum mechanical in nature - to both the dot's size and composition. QDs have been touted as possible replacements for organic dyes in the imaging of biological systems, due to their excellent fluorescent properties, good chemical stability, broad excitation ranges and high photobleaching thresholds. However, the main drawback of QDs is their toxicity and therefore their application is problematic. If this toxicity problem could be addressed, QDs may one day be safely utilized in many areas. For instance, cadmium telluride (CdTe - which is toxic) QD based nanocomposites can be used as fluorescent probes for biological imaging, they can also be utilized to monitor targeted drug delivery and for controlled modification of structural and functional properties of intracellular components. Scientists in Ireland have been using gelatin during the production of CdTe QDs thereby reducing the toxicity of the particles. Their approach could be useful for the development of other nanoparticle composites with low toxicity as well.
Jul 13th, 2007
 New technology, whether it is a novel cancer treatment or an innovative approach to farming, almost always comes with risk. Those risks are often first - and most severely - felt by industry workers, and nanotechnology is no different. Today, workers around the world are exposed to nanoparticles on a daily basis. There is much speculation, yet so far, little definitive information about how exposure affects workers. A report released by the International Council on Nanotechnology in November 2006, offers a clear picture of the situation. "The properties for which novel nanoscale materials are designed may generate new risks to workers, consumers, the public, and the environment. While some of these risks can be anticipated from experiences with other synthetic chemicals and with existing knowledge of ambient and manufactured fine particles, novel risks associated with new properties cannot easily be anticipated based on existing data." Questions, such as how to measure toxicity and how to monitor and control exposure, remain unanswered.
New technology, whether it is a novel cancer treatment or an innovative approach to farming, almost always comes with risk. Those risks are often first - and most severely - felt by industry workers, and nanotechnology is no different. Today, workers around the world are exposed to nanoparticles on a daily basis. There is much speculation, yet so far, little definitive information about how exposure affects workers. A report released by the International Council on Nanotechnology in November 2006, offers a clear picture of the situation. "The properties for which novel nanoscale materials are designed may generate new risks to workers, consumers, the public, and the environment. While some of these risks can be anticipated from experiences with other synthetic chemicals and with existing knowledge of ambient and manufactured fine particles, novel risks associated with new properties cannot easily be anticipated based on existing data." Questions, such as how to measure toxicity and how to monitor and control exposure, remain unanswered.
Jul 9th, 2007
 In our May 7 spotlight "The potential and the pitfalls of nanomedicine" we took a general look at the potential implications of nanomedicine and addressed some ethical issues that arise as the technology develops. In part two of this article we now take a closer look at emerging nanomedical techniques such as nanosurgery, tissue engineering, nanoparticle-enabled diagnostics, and targeted drug delivery. Again, the ethical issues inherent in these emerging medical technologies need to be considered. There are established principals for ethical assessment of existing, conventional, medical technologies and a new research article examines if and how these principals can be extended to nanomedicine.
In our May 7 spotlight "The potential and the pitfalls of nanomedicine" we took a general look at the potential implications of nanomedicine and addressed some ethical issues that arise as the technology develops. In part two of this article we now take a closer look at emerging nanomedical techniques such as nanosurgery, tissue engineering, nanoparticle-enabled diagnostics, and targeted drug delivery. Again, the ethical issues inherent in these emerging medical technologies need to be considered. There are established principals for ethical assessment of existing, conventional, medical technologies and a new research article examines if and how these principals can be extended to nanomedicine.
May 23rd, 2007
 Engineered nanoparticles are at the forefront of the rapidly developing field of nanomedicine. Their unique size-dependent properties, of which optical and magnetic effects are the most used for biological applications, makes them suitable for a wide range of biomedical applications such as cell labeling and targeting, tissue engineering, drug delivery and drug targeting, magnetic resonance imaging, probing of DNA structure, tumor destruction via heating (hyperthermia), and detection and analysis of biomolecules such as proteins or pathogens. Many of these applications can also be tailored to target skin to help in the early diagnosis of a skin disease, which then could also be treated via nanocarriers. In addition, a tissue engineering approach could be useful for skin wound healing therapies and the magnetic properties of these particles might help in directing and localizing these agents in a particular layer of the skin where their action is desired. Unfortunately, if nanoparticles are able to penetrate layers of skin for therapeutic purposes, they might equally be able to penetrate skin unintentionally. This raises the question if people, who are exposed to such nanomaterials, could accidentally be contaminated and thus exposed to a potential local and/or systemic health risk. Researchers in Italy now have begun to systematically evaluate both risks and applications of nanoparticle skin absorption.
Engineered nanoparticles are at the forefront of the rapidly developing field of nanomedicine. Their unique size-dependent properties, of which optical and magnetic effects are the most used for biological applications, makes them suitable for a wide range of biomedical applications such as cell labeling and targeting, tissue engineering, drug delivery and drug targeting, magnetic resonance imaging, probing of DNA structure, tumor destruction via heating (hyperthermia), and detection and analysis of biomolecules such as proteins or pathogens. Many of these applications can also be tailored to target skin to help in the early diagnosis of a skin disease, which then could also be treated via nanocarriers. In addition, a tissue engineering approach could be useful for skin wound healing therapies and the magnetic properties of these particles might help in directing and localizing these agents in a particular layer of the skin where their action is desired. Unfortunately, if nanoparticles are able to penetrate layers of skin for therapeutic purposes, they might equally be able to penetrate skin unintentionally. This raises the question if people, who are exposed to such nanomaterials, could accidentally be contaminated and thus exposed to a potential local and/or systemic health risk. Researchers in Italy now have begun to systematically evaluate both risks and applications of nanoparticle skin absorption.
Apr 24th, 2007
 Nano-this and nano-that. These days it seems you need the prefix "nano" for products or applications if you want to be either very trendy or incredibly scary. This "nanotrend" has assumed "mega" proportions: Patent offices around the world are swamped with nanotechnology-related applications; investment advisors compile nanotechnology stock indices and predict a coming boom in nanotechnology stocks with misleading estimates floating around of a trillion-dollar industry within 10 years; pundits promise a new world with radically different medical procedures, manufacturing technologies and solutions to environmental problems; nano conferences and trade shows are thriving all over the world; scientific journals are awash in articles dealing with nanoscience discoveries and nano- technology breakthroughs. Nanotechnology has been plagued by a lot of hype, but cynicism and criticism have not been far behind. Science fiction writers exploit fears of nanorobots turning into killers; the media can run amok when news about potential health problems with nanoproducts surface (as happened last year with a product recall for a bathroom cleaner in Germany). Some see doomsday scenarios of molecular self-assembly turning the world into "grey goo." The emerging polarization of opinions on nanotechnology is reminiscent of controversies about genetically modified plants or nuclear energy. Vague promises of a better life are met by equally vague, generalized fears about a worse future. These debates have some aspects in common: the subject is complex and not easy to explain; there is no consensus on risks and benefits; scientists and corporations seem able to proceed unchecked, and it is unclear who is in control.
Nano-this and nano-that. These days it seems you need the prefix "nano" for products or applications if you want to be either very trendy or incredibly scary. This "nanotrend" has assumed "mega" proportions: Patent offices around the world are swamped with nanotechnology-related applications; investment advisors compile nanotechnology stock indices and predict a coming boom in nanotechnology stocks with misleading estimates floating around of a trillion-dollar industry within 10 years; pundits promise a new world with radically different medical procedures, manufacturing technologies and solutions to environmental problems; nano conferences and trade shows are thriving all over the world; scientific journals are awash in articles dealing with nanoscience discoveries and nano- technology breakthroughs. Nanotechnology has been plagued by a lot of hype, but cynicism and criticism have not been far behind. Science fiction writers exploit fears of nanorobots turning into killers; the media can run amok when news about potential health problems with nanoproducts surface (as happened last year with a product recall for a bathroom cleaner in Germany). Some see doomsday scenarios of molecular self-assembly turning the world into "grey goo." The emerging polarization of opinions on nanotechnology is reminiscent of controversies about genetically modified plants or nuclear energy. Vague promises of a better life are met by equally vague, generalized fears about a worse future. These debates have some aspects in common: the subject is complex and not easy to explain; there is no consensus on risks and benefits; scientists and corporations seem able to proceed unchecked, and it is unclear who is in control.
Apr 16th, 2007
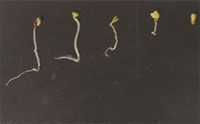 Nanomaterials, with at least one dimension of 100 nanometers or less, are increasingly being used for commercial purposes such as fillers, opacifiers, catalysts, semiconductors, cosmetics, microelectronics, and drug carriers. Nanoparticles with a size of between 1 and 100 nanometers fall in the transitional zone between individual atoms (or molecules) and the bulk material. Because the physicochemical properties of material on this scale can greatly differ from the corresponding bulk material, these nanomaterials can have the potential to generate unknown biological effects in living cells. As the discussion on potentially undesired side effects of engineered nanoparticles heats up there is an increasing amount of nanotoxicology research that gets undertaken and published. However, very few studies have been conducted to assess the toxicity of nanomaterials to ecological terrestrial species, particularly plants. In order to develop a comprehensive toxicity profile for manufactured nanoparticles, their phytotoxicity - the ability to cause injury to plants - has to be investigated. A new study examined the effects of five types of nanoparticles on seed germination and root growth of six higher plant species and observed that several types of the particles had significant inhibition on seed germination and root growth of the six plants. If confirmed, these results are significant in terms of use and disposal of engineered nanoparticles.
Nanomaterials, with at least one dimension of 100 nanometers or less, are increasingly being used for commercial purposes such as fillers, opacifiers, catalysts, semiconductors, cosmetics, microelectronics, and drug carriers. Nanoparticles with a size of between 1 and 100 nanometers fall in the transitional zone between individual atoms (or molecules) and the bulk material. Because the physicochemical properties of material on this scale can greatly differ from the corresponding bulk material, these nanomaterials can have the potential to generate unknown biological effects in living cells. As the discussion on potentially undesired side effects of engineered nanoparticles heats up there is an increasing amount of nanotoxicology research that gets undertaken and published. However, very few studies have been conducted to assess the toxicity of nanomaterials to ecological terrestrial species, particularly plants. In order to develop a comprehensive toxicity profile for manufactured nanoparticles, their phytotoxicity - the ability to cause injury to plants - has to be investigated. A new study examined the effects of five types of nanoparticles on seed germination and root growth of six higher plant species and observed that several types of the particles had significant inhibition on seed germination and root growth of the six plants. If confirmed, these results are significant in terms of use and disposal of engineered nanoparticles.
Mar 27th, 2007
 It seems that with every new study on the toxicity of nanomaterials there remain more questions afterwards than before. Environmental, occupational and public exposure to engineered nanoparticles will increase dramatically in the near future as a result of the widespread use of nanoparticles for consumer and industrial products. The extent of future exposure to nanoparticles associated with these new products is still unknown. So far only limited data is available regarding carbon nanotube (CNT) toxicity. As a result still not much is known about their impact on biological systems including humans. Discussions regarding the potential risks of their widespread use, as well as their possible positive impact are just beginning to take place. In order to provide a basis for comparison to existing epidemiological data, a group of researchers in Switzerland and Germany have investigated CNTs at various degrees of agglomeration using an in vitro cytotoxicity study with human cancer cells. The cytotoxic effects of well-dispersed CNT were compared with that of conventionally purified rope-like agglomerated CNTs and asbestos as a reference. While suspended CNT-bundles were less cytotoxic than asbestos, rope-like agglomerates induced more pronounced cytotoxic effects than asbestos fibers at the same concentrations. The study underlines the need for thorough materials characterization prior to toxicological studies and corroborates the role of agglomeration in the cytotoxic effect of nanomaterials.
It seems that with every new study on the toxicity of nanomaterials there remain more questions afterwards than before. Environmental, occupational and public exposure to engineered nanoparticles will increase dramatically in the near future as a result of the widespread use of nanoparticles for consumer and industrial products. The extent of future exposure to nanoparticles associated with these new products is still unknown. So far only limited data is available regarding carbon nanotube (CNT) toxicity. As a result still not much is known about their impact on biological systems including humans. Discussions regarding the potential risks of their widespread use, as well as their possible positive impact are just beginning to take place. In order to provide a basis for comparison to existing epidemiological data, a group of researchers in Switzerland and Germany have investigated CNTs at various degrees of agglomeration using an in vitro cytotoxicity study with human cancer cells. The cytotoxic effects of well-dispersed CNT were compared with that of conventionally purified rope-like agglomerated CNTs and asbestos as a reference. While suspended CNT-bundles were less cytotoxic than asbestos, rope-like agglomerates induced more pronounced cytotoxic effects than asbestos fibers at the same concentrations. The study underlines the need for thorough materials characterization prior to toxicological studies and corroborates the role of agglomeration in the cytotoxic effect of nanomaterials.
Mar 12th, 2007
 The question if certain engineered nanoparticles are toxic, and if yes to what degree, is still one of the major issues that hasn't been properly answered yet. Most studies in the literature thus far have focused on the environmental aspects of nanoparticle toxicity, and these studies have been conducted primarily on industrial or natural/incidental nanoparticles. However, engineered nanoparticles are at the forefront of the rapidly developing field of nanomedicine; and here they are deliberately injected into the body to perform a specific medical application: fluorescent agents for imaging; drug delivery vehicles; or therapeutic agents for the destruction of cancer cells (for instance in thermolysis); just to name a few. A brand new review article provides the first comprehensive summary of the properties of engineered nanoparticles which determine their interaction with components of the immune system. It concludes that nanoparticle-based therapeutics are no more intrinsically immunotoxic than traditional pharmaceuticals, such as biotechnology-derived or small molecules. Moreover, incorporation of traditional drugs into nanotechnology formulations frequently results in a decrease in immunotoxicity compared to the native drug. Although many questions still require thorough investigation, the available data suggest that nanoparticles can be engineered to become the next generation of biocompatible drug delivery platforms.
The question if certain engineered nanoparticles are toxic, and if yes to what degree, is still one of the major issues that hasn't been properly answered yet. Most studies in the literature thus far have focused on the environmental aspects of nanoparticle toxicity, and these studies have been conducted primarily on industrial or natural/incidental nanoparticles. However, engineered nanoparticles are at the forefront of the rapidly developing field of nanomedicine; and here they are deliberately injected into the body to perform a specific medical application: fluorescent agents for imaging; drug delivery vehicles; or therapeutic agents for the destruction of cancer cells (for instance in thermolysis); just to name a few. A brand new review article provides the first comprehensive summary of the properties of engineered nanoparticles which determine their interaction with components of the immune system. It concludes that nanoparticle-based therapeutics are no more intrinsically immunotoxic than traditional pharmaceuticals, such as biotechnology-derived or small molecules. Moreover, incorporation of traditional drugs into nanotechnology formulations frequently results in a decrease in immunotoxicity compared to the native drug. Although many questions still require thorough investigation, the available data suggest that nanoparticles can be engineered to become the next generation of biocompatible drug delivery platforms.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





