Showing Spotlights 1201 - 1208 of 2798 in category All (newest first):
 While nanotechnology researchers have made great progress over the past few years in developing self-propelled nano objects, these tiny devices still fall far short of what their natural counterparts' performance. Today, artificial nanomotors lack the sophisticated functionality of biomotors and are limited to a very narrow range of environments and fuels. In another step towards realizing the vision of tiny vessels roaming around in human blood vessels working as surgical nanorobots, researchers have now demonstrated, for the first time, externally driven nanomotors that move in undiluted human blood.
While nanotechnology researchers have made great progress over the past few years in developing self-propelled nano objects, these tiny devices still fall far short of what their natural counterparts' performance. Today, artificial nanomotors lack the sophisticated functionality of biomotors and are limited to a very narrow range of environments and fuels. In another step towards realizing the vision of tiny vessels roaming around in human blood vessels working as surgical nanorobots, researchers have now demonstrated, for the first time, externally driven nanomotors that move in undiluted human blood.
Apr 17th, 2014
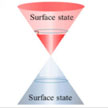 Recently, a new Dirac material - a lattice system where the excitations are described by relativistic Dirac or Weyl equations - namely a topological insulator (TI), entered researchers' sight. TIs possess a small band gap in their bulk state and a gapless metallic state at their edge/surface. A research group working on two-dimensional materials photonicshas now experimentally demonstrated for the first time that TI may be a novel microwave-absorbing material.
Recently, a new Dirac material - a lattice system where the excitations are described by relativistic Dirac or Weyl equations - namely a topological insulator (TI), entered researchers' sight. TIs possess a small band gap in their bulk state and a gapless metallic state at their edge/surface. A research group working on two-dimensional materials photonicshas now experimentally demonstrated for the first time that TI may be a novel microwave-absorbing material.
Apr 15th, 2014
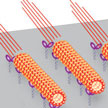 Over the past few years, researchers have demonstrated that microtubules driven by kinesin make flexible, responsive and effective molecular shuttles for nanotransport applications. In order to fully control microtubules driven by kinesin it has to be possible to switch them on, switch them off, and regulate the speed and direction of their movements - achievements that until now researchers have't fully attained yet. Now, though, it has become possible, for the first time, to achieve complete control over on/off switching of the movement of a nanomachine.
Over the past few years, researchers have demonstrated that microtubules driven by kinesin make flexible, responsive and effective molecular shuttles for nanotransport applications. In order to fully control microtubules driven by kinesin it has to be possible to switch them on, switch them off, and regulate the speed and direction of their movements - achievements that until now researchers have't fully attained yet. Now, though, it has become possible, for the first time, to achieve complete control over on/off switching of the movement of a nanomachine.
Apr 14th, 2014
 Nanocellulose from wood is a promising nanomaterial with potential applications as a substrate for printing electronics, filtration, or biomedicine. Researchers have now reported on a method to control the surface chemistry of nanocellulose. They fabricated nanocellulose gels that have a significantly higher swelling degree in neutral and alkaline conditions, compared to an acid environment. This material could be of great interest for critical wound healing applications.
Nanocellulose from wood is a promising nanomaterial with potential applications as a substrate for printing electronics, filtration, or biomedicine. Researchers have now reported on a method to control the surface chemistry of nanocellulose. They fabricated nanocellulose gels that have a significantly higher swelling degree in neutral and alkaline conditions, compared to an acid environment. This material could be of great interest for critical wound healing applications.
Apr 10th, 2014
 Nanomaterials for nanomedicine and biological applications are often two-component structures - referred to as 'nanoconstructs' -consisting of a 'hard' nanoparticle core and a 'soft' shell of biomolecular ligands. Researchers have now demonstrated a nanoconstruct with enhanced in vitro efficacy. This highly loaded nanoconstruct was taken up by pancreatic cancer cells and fibrosarcoma cells at fast rates. The team found that the increased loading of Apt on AuNS also resulted in an enhanced in vitro response.
Nanomaterials for nanomedicine and biological applications are often two-component structures - referred to as 'nanoconstructs' -consisting of a 'hard' nanoparticle core and a 'soft' shell of biomolecular ligands. Researchers have now demonstrated a nanoconstruct with enhanced in vitro efficacy. This highly loaded nanoconstruct was taken up by pancreatic cancer cells and fibrosarcoma cells at fast rates. The team found that the increased loading of Apt on AuNS also resulted in an enhanced in vitro response.
Apr 9th, 2014
 Researchers have proposed an alternative way of making graphene from rice husk. This research, using an ordinary synthetic apparatus and abundant agricultural waste, suggest that low cost graphene materials could now be easily and cheaply synthesized on an industrial scale. Due to its abundance, risk husk has already received much attention as a starting material in generating high-value-added materials such as silica and porous carbon.
Researchers have proposed an alternative way of making graphene from rice husk. This research, using an ordinary synthetic apparatus and abundant agricultural waste, suggest that low cost graphene materials could now be easily and cheaply synthesized on an industrial scale. Due to its abundance, risk husk has already received much attention as a starting material in generating high-value-added materials such as silica and porous carbon.
Apr 7th, 2014
 Scientists have great expectations that nanotechnologies will bring them closer to the goal of creating computer systems that can simulate and emulate the brain's abilities for sensation, perception, action, interaction and cognition while rivaling its low power consumption and compact size - basically a brain-on-a-chip. Already, scientists are working hard on laying the foundations for what is called neuromorphic engineering - a new interdisciplinary discipline that includes nanotechnologies and whose goal is to design artificial neural systems with physical architectures similar to biological nervous systems.
Scientists have great expectations that nanotechnologies will bring them closer to the goal of creating computer systems that can simulate and emulate the brain's abilities for sensation, perception, action, interaction and cognition while rivaling its low power consumption and compact size - basically a brain-on-a-chip. Already, scientists are working hard on laying the foundations for what is called neuromorphic engineering - a new interdisciplinary discipline that includes nanotechnologies and whose goal is to design artificial neural systems with physical architectures similar to biological nervous systems.
Apr 4th, 2014
 New solar cell technology allows your T-shirt to generate power from its interwoven solar cell wires. Researchers have developed a novel efficient wire-shaped polymer solar cell by incorporating a thin layer of titania nanoparticles between the photoactive material and electrode. An aligned carbon nanotube fiber enabled high flexibility and stability of the resulting polymer solar cell. These miniature polymer solar cell wires, when woven into textiles, can serve as a power source.
New solar cell technology allows your T-shirt to generate power from its interwoven solar cell wires. Researchers have developed a novel efficient wire-shaped polymer solar cell by incorporating a thin layer of titania nanoparticles between the photoactive material and electrode. An aligned carbon nanotube fiber enabled high flexibility and stability of the resulting polymer solar cell. These miniature polymer solar cell wires, when woven into textiles, can serve as a power source.
Apr 3rd, 2014
 While nanotechnology researchers have made great progress over the past few years in developing self-propelled nano objects, these tiny devices still fall far short of what their natural counterparts' performance. Today, artificial nanomotors lack the sophisticated functionality of biomotors and are limited to a very narrow range of environments and fuels. In another step towards realizing the vision of tiny vessels roaming around in human blood vessels working as surgical nanorobots, researchers have now demonstrated, for the first time, externally driven nanomotors that move in undiluted human blood.
While nanotechnology researchers have made great progress over the past few years in developing self-propelled nano objects, these tiny devices still fall far short of what their natural counterparts' performance. Today, artificial nanomotors lack the sophisticated functionality of biomotors and are limited to a very narrow range of environments and fuels. In another step towards realizing the vision of tiny vessels roaming around in human blood vessels working as surgical nanorobots, researchers have now demonstrated, for the first time, externally driven nanomotors that move in undiluted human blood.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





