Showing Spotlights 1225 - 1232 of 2787 in category All (newest first):
 Nanowire field-effect sensors show significant advantages of real-time, label-free and highly sensitive detection of a wide range of analytes in liquid phase, including proteins, nucleic acids, small molecules, and viruses in single-element or multiplexed formats. Motivated by the unique features of these sensors and the ease to integrate them in the currently available VLSI technology, researchers used molecularly modified silicon nanowire FETs to detect volatile organic compounds (VOCs) that are associated with environmental pollution, quality control, explosive materials, or various diseases.
Nanowire field-effect sensors show significant advantages of real-time, label-free and highly sensitive detection of a wide range of analytes in liquid phase, including proteins, nucleic acids, small molecules, and viruses in single-element or multiplexed formats. Motivated by the unique features of these sensors and the ease to integrate them in the currently available VLSI technology, researchers used molecularly modified silicon nanowire FETs to detect volatile organic compounds (VOCs) that are associated with environmental pollution, quality control, explosive materials, or various diseases.
Jan 30th, 2014
 Graphene is undoubtedly emerging as one of the most promising nanomaterials because of its unique combination of superb properties, which opens a way for its exploitation in a wide spectrum of applications ranging from electronics to optics, sensors, and biodevices. In this Nanowerk nanotechnology primer we summarize recent finding in graphene research and show the breadth of graphene applications in such areas as energy, electronics, sensors and many others.
Graphene is undoubtedly emerging as one of the most promising nanomaterials because of its unique combination of superb properties, which opens a way for its exploitation in a wide spectrum of applications ranging from electronics to optics, sensors, and biodevices. In this Nanowerk nanotechnology primer we summarize recent finding in graphene research and show the breadth of graphene applications in such areas as energy, electronics, sensors and many others.
Jan 29th, 2014
 Recently, nanotechnology researchers have begun to work with graphene foams - three-dimensional structures of interconnected graphene sheets with extremely high conductivity. Since graphene foam possesses a high porosity of close to 100%, this offers the opportunity to use it as a scaffold for other nanomaterials to generate synergistic effects. Now, researchers have fabricated vertically aligned ZnO nanowire arrays on 3D graphene foam and used this electrode to detect uric acid in a reliable statistical level from the serum of Parkinson's disease patients.
Recently, nanotechnology researchers have begun to work with graphene foams - three-dimensional structures of interconnected graphene sheets with extremely high conductivity. Since graphene foam possesses a high porosity of close to 100%, this offers the opportunity to use it as a scaffold for other nanomaterials to generate synergistic effects. Now, researchers have fabricated vertically aligned ZnO nanowire arrays on 3D graphene foam and used this electrode to detect uric acid in a reliable statistical level from the serum of Parkinson's disease patients.
Jan 27th, 2014
 Paper could lead to low-cost innovative devices and applications is lab-on-a-chip technology. In new work, researchers in Korea have, for the first time, used paper as a platform material for actively actuating an electronic microfluidic chip. This novel, powered fluidic chip - known as an active paper open chip (APOC) - allows the full range of fluidic operations by implementing an electric input on paper via an electrowetting technique.
Paper could lead to low-cost innovative devices and applications is lab-on-a-chip technology. In new work, researchers in Korea have, for the first time, used paper as a platform material for actively actuating an electronic microfluidic chip. This novel, powered fluidic chip - known as an active paper open chip (APOC) - allows the full range of fluidic operations by implementing an electric input on paper via an electrowetting technique.
Jan 23rd, 2014
 Transparent and flexible substrates are widely explored for flexible electronics and researchers have been working on techniques to develop thermally stable and biodegradable materials that are as easily printable as paper. Previously, we reported on a transparent and flexible nanopaper transistor. The same team has now reported a novel transparent paper substrate design optimized for solar cells. They introduced a novel transparent paper made of earth-abundant wood fibers that simultaneously achieves an ultrahigh transmittance and ultrahigh optical haze.
Transparent and flexible substrates are widely explored for flexible electronics and researchers have been working on techniques to develop thermally stable and biodegradable materials that are as easily printable as paper. Previously, we reported on a transparent and flexible nanopaper transistor. The same team has now reported a novel transparent paper substrate design optimized for solar cells. They introduced a novel transparent paper made of earth-abundant wood fibers that simultaneously achieves an ultrahigh transmittance and ultrahigh optical haze.
Jan 22nd, 2014
 In the field on controlling liquid movement on surfaces, super water-repellent surfaces have been well-documented. In contrast, comparatively fewer reports are available on the design of water pinning surfaces. In new work, scientists have achieved polymer films with exceptionally high water pinning forces through nanoimprinted surface structures, without the incorporation of any chemical treatment. This work contributes to the field on water pinning surfaces by providing a simple geometrical rule-of-thumb design of nanostructures to engineer polymeric surfaces with tunable water pinning ability.
In the field on controlling liquid movement on surfaces, super water-repellent surfaces have been well-documented. In contrast, comparatively fewer reports are available on the design of water pinning surfaces. In new work, scientists have achieved polymer films with exceptionally high water pinning forces through nanoimprinted surface structures, without the incorporation of any chemical treatment. This work contributes to the field on water pinning surfaces by providing a simple geometrical rule-of-thumb design of nanostructures to engineer polymeric surfaces with tunable water pinning ability.
Jan 14th, 2014
 Numerous previous research results have shown that gold nanorods (Au NRs) are able to effectively kill tumors upon irradiation with high laser doses, leading to hyperthermia-induced destruction of cancer cells (photodynamic therapy, PTT). In new work, researchers have demonstrated for the first time that bare gold nanorods alone can exert unprecedented photodynamic therapeutic (PDT) effects to result in cancer cellular deaths at low laser doses. Moreover, it appears that Au NRs-mediated PDT effects are far more effective in destruction of tumors in mice than the Au NRs-mediated PTT effects.
Numerous previous research results have shown that gold nanorods (Au NRs) are able to effectively kill tumors upon irradiation with high laser doses, leading to hyperthermia-induced destruction of cancer cells (photodynamic therapy, PTT). In new work, researchers have demonstrated for the first time that bare gold nanorods alone can exert unprecedented photodynamic therapeutic (PDT) effects to result in cancer cellular deaths at low laser doses. Moreover, it appears that Au NRs-mediated PDT effects are far more effective in destruction of tumors in mice than the Au NRs-mediated PTT effects.
Jan 13th, 2014
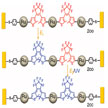 External stimuli, such as light, mechanical force, magnetic field, electrical field and electrochemical potential, are all driving forces that can be utilized to modulate the structure or conformation of molecules, and therefore to affect the performance of functional molecular devices. In new work, researchers take advantage of synergetic modulation by multiple external controls to explore multi-modulable molecular devices with the help of chemical tailoring, which have not been addressed so far.
External stimuli, such as light, mechanical force, magnetic field, electrical field and electrochemical potential, are all driving forces that can be utilized to modulate the structure or conformation of molecules, and therefore to affect the performance of functional molecular devices. In new work, researchers take advantage of synergetic modulation by multiple external controls to explore multi-modulable molecular devices with the help of chemical tailoring, which have not been addressed so far.
Jan 8th, 2014
 Nanowire field-effect sensors show significant advantages of real-time, label-free and highly sensitive detection of a wide range of analytes in liquid phase, including proteins, nucleic acids, small molecules, and viruses in single-element or multiplexed formats. Motivated by the unique features of these sensors and the ease to integrate them in the currently available VLSI technology, researchers used molecularly modified silicon nanowire FETs to detect volatile organic compounds (VOCs) that are associated with environmental pollution, quality control, explosive materials, or various diseases.
Nanowire field-effect sensors show significant advantages of real-time, label-free and highly sensitive detection of a wide range of analytes in liquid phase, including proteins, nucleic acids, small molecules, and viruses in single-element or multiplexed formats. Motivated by the unique features of these sensors and the ease to integrate them in the currently available VLSI technology, researchers used molecularly modified silicon nanowire FETs to detect volatile organic compounds (VOCs) that are associated with environmental pollution, quality control, explosive materials, or various diseases.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





