Showing Spotlights 1497 - 1504 of 2785 in category All (newest first):
 Graphene-based materials are emerging as highly attractive materials for real applications, especially in the area of energy conversion and storage. There are four major energy-related areas where graphene will have an impact: solar cells, supercapacitors, lithium-ion batteries, and catalysis for fuel cells. A recent review gives a brief overview of the recent research concerning chemical and thermal approaches toward the production of well-defined graphene-based nanomaterials and their applications in energy-related areas. But before graphene-based nanomaterials and devices find widespread commercial use, two important problems have to be solved: one is the preparation of graphene-based nanomaterials with well-defined structures, and the other is the controllable fabrication of these materials into functional devices.
Graphene-based materials are emerging as highly attractive materials for real applications, especially in the area of energy conversion and storage. There are four major energy-related areas where graphene will have an impact: solar cells, supercapacitors, lithium-ion batteries, and catalysis for fuel cells. A recent review gives a brief overview of the recent research concerning chemical and thermal approaches toward the production of well-defined graphene-based nanomaterials and their applications in energy-related areas. But before graphene-based nanomaterials and devices find widespread commercial use, two important problems have to be solved: one is the preparation of graphene-based nanomaterials with well-defined structures, and the other is the controllable fabrication of these materials into functional devices.
Apr 18th, 2012
 In recent years billions of dollars have been invested in nanotechnology research. Nanotechnology is said to have such pervasive consequences that the 21st century is already proclaimed to become the 'nano-century'. Nanotechnology may also contribute to development in the global South. The new characteristics of nanotechnologies are also said to offer solutions for problems faced by people in the global South, especially in the fields of water, energy, and health. For instance water filters, energy storage systems, solar powered electricity and portable diagnostic tests may be developed and improved using nanotechnology. But the very same features that give rise to new opportunities may also generate new risks. There may be risks to the human body and environment. But for instance also investing in nanotechnology itself can be seen as a risk.
In recent years billions of dollars have been invested in nanotechnology research. Nanotechnology is said to have such pervasive consequences that the 21st century is already proclaimed to become the 'nano-century'. Nanotechnology may also contribute to development in the global South. The new characteristics of nanotechnologies are also said to offer solutions for problems faced by people in the global South, especially in the fields of water, energy, and health. For instance water filters, energy storage systems, solar powered electricity and portable diagnostic tests may be developed and improved using nanotechnology. But the very same features that give rise to new opportunities may also generate new risks. There may be risks to the human body and environment. But for instance also investing in nanotechnology itself can be seen as a risk.
Apr 17th, 2012
 Ultra- or supercapacitors are emerging as a key enabling storage technology for use in fuel-efficient transport as well as in renewable energy. These devices combine the advantages of conventional capacitors - they can rapidly deliver high current densities on demand - and batteries - they can store a large amount of electrical energy. Today's commercial supercapacitors store energy in two closely spaced layers with opposing charges and offer fast charge/discharge rates and the ability to sustain millions of cycles. Researchers have come up with various electrode materials to improve the performance of supercapacitors, focussing mostly on porous carbon due to its high surface areas, tunable structures, good conductivities, and low cost. In recent years, this has increasingly included research on various carbon nanomaterials such as carbon nanotubes, carbon nano-onions, or graphene. In new work, researchers have demonstrated that a common daily waste - the eggshell membrane - can be converted into a high-performance carbon material for supercapacitors.
Ultra- or supercapacitors are emerging as a key enabling storage technology for use in fuel-efficient transport as well as in renewable energy. These devices combine the advantages of conventional capacitors - they can rapidly deliver high current densities on demand - and batteries - they can store a large amount of electrical energy. Today's commercial supercapacitors store energy in two closely spaced layers with opposing charges and offer fast charge/discharge rates and the ability to sustain millions of cycles. Researchers have come up with various electrode materials to improve the performance of supercapacitors, focussing mostly on porous carbon due to its high surface areas, tunable structures, good conductivities, and low cost. In recent years, this has increasingly included research on various carbon nanomaterials such as carbon nanotubes, carbon nano-onions, or graphene. In new work, researchers have demonstrated that a common daily waste - the eggshell membrane - can be converted into a high-performance carbon material for supercapacitors.
Apr 12th, 2012
 The global cement industry is currently one of the largest single emitter of carbon dioxide, generating on average about 830 kg of this greenhouse gas for each 1000 kg of cement produced. Considering that the worldwide annual production of cement is a whopping 3.8 trillion kg, the cement industry alone accounts for approximately 5-6% of man-made CO2 emissions. Researchers have now presented a solar-powered process to produce cement without any carbon dioxide. In their STEP (Solar Thermal Electrochemical Production) process, cement limestone undergoes low energy electrolysis to produce lime, O2 and reduced carbonate without carbon dioxide emission. In this new technique, the kiln limestone-to-lime process is replaced by an electrolysis process which changes the product of the reaction of the limestone as it is converted to lime.
The global cement industry is currently one of the largest single emitter of carbon dioxide, generating on average about 830 kg of this greenhouse gas for each 1000 kg of cement produced. Considering that the worldwide annual production of cement is a whopping 3.8 trillion kg, the cement industry alone accounts for approximately 5-6% of man-made CO2 emissions. Researchers have now presented a solar-powered process to produce cement without any carbon dioxide. In their STEP (Solar Thermal Electrochemical Production) process, cement limestone undergoes low energy electrolysis to produce lime, O2 and reduced carbonate without carbon dioxide emission. In this new technique, the kiln limestone-to-lime process is replaced by an electrolysis process which changes the product of the reaction of the limestone as it is converted to lime.
Apr 11th, 2012
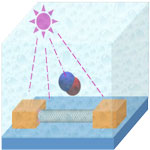 Single-walled carbon nanotubes arguably are the ultimate biosensor among nanoscale semiconducting materials due to their high surface-to-volume ratio and unique electronic structure. After more than a decade of excitement though, more and more researchers in the nanotube field believe that pristine SWCNTs are very limited as a sensing material. Ironically the ultrahigh sensitivity of SWCNTs is easily compromised by various unintentional contaminants from the device fabrication process as well as the ambient environment. As a result, significant efforts have been focused on all kinds of ways to functionalize or decorate nanotubes with other species in order to improve their sensitivity. Researchers have now shown that applying continuous in situ ultraviolet light illumination during gas detection can enhance a SWCNT-sensor's performance by orders of magnitude under otherwise identical sensing conditions.
Single-walled carbon nanotubes arguably are the ultimate biosensor among nanoscale semiconducting materials due to their high surface-to-volume ratio and unique electronic structure. After more than a decade of excitement though, more and more researchers in the nanotube field believe that pristine SWCNTs are very limited as a sensing material. Ironically the ultrahigh sensitivity of SWCNTs is easily compromised by various unintentional contaminants from the device fabrication process as well as the ambient environment. As a result, significant efforts have been focused on all kinds of ways to functionalize or decorate nanotubes with other species in order to improve their sensitivity. Researchers have now shown that applying continuous in situ ultraviolet light illumination during gas detection can enhance a SWCNT-sensor's performance by orders of magnitude under otherwise identical sensing conditions.
Apr 10th, 2012
 On-wire lithography is a recently developed nanotechnology fabrication technique that allows researchers to synthesize billions of gapped nanowires with nanometer control of gap length, within a single experiment. These gaps can then be used to integrate different material segments into a single nanowire in order to fabricate functional devices. In recent work, researchers have reported a simple but efficient method to use OWL to mass produce nanotube-bridged nanowires, including carbon nanotube (CNT) channels with channel lengths as small as 5 nm. Since the CNT-bridged nanowires are comprised of CNT junctions with gold electrodes, each of the nanowires could for instance work as a CNT-based sensing device, ballistic transistors, or resonators.
On-wire lithography is a recently developed nanotechnology fabrication technique that allows researchers to synthesize billions of gapped nanowires with nanometer control of gap length, within a single experiment. These gaps can then be used to integrate different material segments into a single nanowire in order to fabricate functional devices. In recent work, researchers have reported a simple but efficient method to use OWL to mass produce nanotube-bridged nanowires, including carbon nanotube (CNT) channels with channel lengths as small as 5 nm. Since the CNT-bridged nanowires are comprised of CNT junctions with gold electrodes, each of the nanowires could for instance work as a CNT-based sensing device, ballistic transistors, or resonators.
Apr 5th, 2012
 All-optical processes could allow dramatic speed increase in photonics by eliminating the need to convert photonic signals to electronic signals and back for switching. The many opportunities that all-optical processes could bring to photonics have been hampered by the lack of materials that combined photosensitivity with fast, large, and reversible changes in their optical properties at the influence of light. By exploited the universal capabilities of an 'active polymeric template' for confining, orienting, and stabilizing a wide range of self-organized materials, researchers recently have exploited a wide range of optical, electro-optical and all-optical effects which confirm the extraordinary capability of their 'active polymeric template' to induce self-organization, without using any kind of surface chemistry or functionalization.
All-optical processes could allow dramatic speed increase in photonics by eliminating the need to convert photonic signals to electronic signals and back for switching. The many opportunities that all-optical processes could bring to photonics have been hampered by the lack of materials that combined photosensitivity with fast, large, and reversible changes in their optical properties at the influence of light. By exploited the universal capabilities of an 'active polymeric template' for confining, orienting, and stabilizing a wide range of self-organized materials, researchers recently have exploited a wide range of optical, electro-optical and all-optical effects which confirm the extraordinary capability of their 'active polymeric template' to induce self-organization, without using any kind of surface chemistry or functionalization.
Apr 4th, 2012
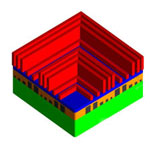 The concept of a 'superlens' has attracted significant research interest in the imaging and photolithography fields since the concept was proposed back in 2000. A superlens would allow you to view objects much smaller than the roughly 200 nanometers that a regular optical lens with visible light would permit. Since superlenses have demonstrated the capability of subdiffraction-limit imaging, they have been envisioned as a promising technology for potential nanophotolithography. Unfortunately, all the experimentally demonstrated photoresist patterns exhibited very low profile depths, leading to poor contrasts, which are far below industrial requirements. Researchers have now experimentally demonstrated sub-50 nm resolution nanophotolithography by using a smooth silver superlens under 365 nm UV light in a conventional photolithography setup.
The concept of a 'superlens' has attracted significant research interest in the imaging and photolithography fields since the concept was proposed back in 2000. A superlens would allow you to view objects much smaller than the roughly 200 nanometers that a regular optical lens with visible light would permit. Since superlenses have demonstrated the capability of subdiffraction-limit imaging, they have been envisioned as a promising technology for potential nanophotolithography. Unfortunately, all the experimentally demonstrated photoresist patterns exhibited very low profile depths, leading to poor contrasts, which are far below industrial requirements. Researchers have now experimentally demonstrated sub-50 nm resolution nanophotolithography by using a smooth silver superlens under 365 nm UV light in a conventional photolithography setup.
Apr 3rd, 2012
 Graphene-based materials are emerging as highly attractive materials for real applications, especially in the area of energy conversion and storage. There are four major energy-related areas where graphene will have an impact: solar cells, supercapacitors, lithium-ion batteries, and catalysis for fuel cells. A recent review gives a brief overview of the recent research concerning chemical and thermal approaches toward the production of well-defined graphene-based nanomaterials and their applications in energy-related areas. But before graphene-based nanomaterials and devices find widespread commercial use, two important problems have to be solved: one is the preparation of graphene-based nanomaterials with well-defined structures, and the other is the controllable fabrication of these materials into functional devices.
Graphene-based materials are emerging as highly attractive materials for real applications, especially in the area of energy conversion and storage. There are four major energy-related areas where graphene will have an impact: solar cells, supercapacitors, lithium-ion batteries, and catalysis for fuel cells. A recent review gives a brief overview of the recent research concerning chemical and thermal approaches toward the production of well-defined graphene-based nanomaterials and their applications in energy-related areas. But before graphene-based nanomaterials and devices find widespread commercial use, two important problems have to be solved: one is the preparation of graphene-based nanomaterials with well-defined structures, and the other is the controllable fabrication of these materials into functional devices.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





