Showing Spotlights 1801 - 1808 of 2777 in category All (newest first):
 Here is a perfect example of how someone, who apparently doesn't understand or care much about the science, writes a sensational press release hyping nanotechnology by cherry-picking information and distorting issues. And all that to sell a product that doesn't even have to do with nanotechnology. Two days ago we ran a press release from Thomson Reuters about a brief report they compiled on patent data relating to nanotechnology in the cosmetics industry. Now, Thomson Reuters is in the business of selling information and information services products and applications. Their press release basically is advertising for their IP Market Reports. There is nothing wrong with that. What is very wrong, though, is the nonsense and unbalanced take on certain aspects on nanotechnologies. Let's take a closer look.
Here is a perfect example of how someone, who apparently doesn't understand or care much about the science, writes a sensational press release hyping nanotechnology by cherry-picking information and distorting issues. And all that to sell a product that doesn't even have to do with nanotechnology. Two days ago we ran a press release from Thomson Reuters about a brief report they compiled on patent data relating to nanotechnology in the cosmetics industry. Now, Thomson Reuters is in the business of selling information and information services products and applications. Their press release basically is advertising for their IP Market Reports. There is nothing wrong with that. What is very wrong, though, is the nonsense and unbalanced take on certain aspects on nanotechnologies. Let's take a closer look.
Jul 15th, 2010
 Researchers in the UK have now conducted experiments that explored the elementary question of what it is that makes some bacteria pathogenic, and some not? Based on their findings, they have demonstrated that a simple vesicle (nanocapsule) system can be used as a 'nano-Trojan horse' for controlling bacterial growth and infection. Integrated into wound dressings, this novel material can automatically detect infection by pathogenic bacteria and respond to this by releasing an antibiotic into the wound, and changing color to alert medical staff. The researchers show that pathogenic bacteria can be used to be the agents of their own destruction by releasing toxins that rupture nanocapsules containing an antimicrobial agent.
Researchers in the UK have now conducted experiments that explored the elementary question of what it is that makes some bacteria pathogenic, and some not? Based on their findings, they have demonstrated that a simple vesicle (nanocapsule) system can be used as a 'nano-Trojan horse' for controlling bacterial growth and infection. Integrated into wound dressings, this novel material can automatically detect infection by pathogenic bacteria and respond to this by releasing an antibiotic into the wound, and changing color to alert medical staff. The researchers show that pathogenic bacteria can be used to be the agents of their own destruction by releasing toxins that rupture nanocapsules containing an antimicrobial agent.
Jul 14th, 2010
 Tailing after emerging nanotechnology applications in biomedical and electronic industries, the construction industry recently started seeking out a way to advance conventional construction materials using a variety of manufactured nanomaterials. The use of nanotechnology materials and applications in the construction industry should be considered not only for enhancing material properties and functions but also in the context of energy conservation. This is a particularly important prospect since a high percentage of all energy used (e.g., 41% in the United States) is consumed by commercial buildings and residential houses by applications such as heating, lighting, and air conditioning. A recent review by scientists at Rice University has looked at the benefits of using nanomaterials in construction materials but also highlights the potentially harmful aspects of releasing nanomaterials into the environment.
Tailing after emerging nanotechnology applications in biomedical and electronic industries, the construction industry recently started seeking out a way to advance conventional construction materials using a variety of manufactured nanomaterials. The use of nanotechnology materials and applications in the construction industry should be considered not only for enhancing material properties and functions but also in the context of energy conservation. This is a particularly important prospect since a high percentage of all energy used (e.g., 41% in the United States) is consumed by commercial buildings and residential houses by applications such as heating, lighting, and air conditioning. A recent review by scientists at Rice University has looked at the benefits of using nanomaterials in construction materials but also highlights the potentially harmful aspects of releasing nanomaterials into the environment.
Jul 13th, 2010
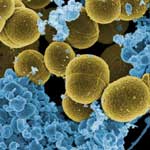 Life-threatening infectious diseases caused by antibiotic-resistant pathogens have been of great concern in both community and hospital settings. This increasing emergence of antibiotic-resistant strains of pathogens has necessitated the development of new antimicrobial surfaces and coatings. As antimicrobial surfaces have become popular in such areas as consumer products, public spaces such as schools and offices, and public transportation, the market for these coatings has quickly grown into a market worth hundreds of million of dollars. New work, by a team from Rensselaer Polytechnic Institute (RPI) has now combined the antimicrobial property of a cell lytic enzyme (lysostaphin) and the excellent properties of carbon nanotubes as an immobilization support in preparing nanocomposite paints that are highly effective against antibiotic-resistant strains of Staphylococcus aureus - methicillin-resistant Staphylococcus aureus (MRSA).
Life-threatening infectious diseases caused by antibiotic-resistant pathogens have been of great concern in both community and hospital settings. This increasing emergence of antibiotic-resistant strains of pathogens has necessitated the development of new antimicrobial surfaces and coatings. As antimicrobial surfaces have become popular in such areas as consumer products, public spaces such as schools and offices, and public transportation, the market for these coatings has quickly grown into a market worth hundreds of million of dollars. New work, by a team from Rensselaer Polytechnic Institute (RPI) has now combined the antimicrobial property of a cell lytic enzyme (lysostaphin) and the excellent properties of carbon nanotubes as an immobilization support in preparing nanocomposite paints that are highly effective against antibiotic-resistant strains of Staphylococcus aureus - methicillin-resistant Staphylococcus aureus (MRSA).
Jul 12th, 2010
 Biomechanical energy is one of the main energy components in biological systems. Developing an effective technique that can convert biomechanical energy into electricity is important for the future of in vivo implantable biosensors and other nanomedical devices. Researchers have already shown the conversion of biomechanical energy into electricity by a muscle-movement-driven nanogenerator to harvest mechanical energy from body movement under in vitro conditions. In a first demonstration of using nanotechnology to convert tiny physical motion into electricity in an in vivo environment, the same team has now reported the implanting of a nanogenerator in a live rat to harvest energy generated by its breath and heartbeat.
Biomechanical energy is one of the main energy components in biological systems. Developing an effective technique that can convert biomechanical energy into electricity is important for the future of in vivo implantable biosensors and other nanomedical devices. Researchers have already shown the conversion of biomechanical energy into electricity by a muscle-movement-driven nanogenerator to harvest mechanical energy from body movement under in vitro conditions. In a first demonstration of using nanotechnology to convert tiny physical motion into electricity in an in vivo environment, the same team has now reported the implanting of a nanogenerator in a live rat to harvest energy generated by its breath and heartbeat.
Jul 9th, 2010
 After achieving the 45-nm process, today's semiconductor industry is nearing the 20-nm process and looking for techniques that would enable sub-22-nm-half-pitch line patterns. Following the continuous increase in exposure tool numerical aperture, researchers are pursuing reductions in exposure wavelengths. This effort had them look at extreme ultraviolet (EUV: 13.4 nm in wavelength) as an exposure light source. Unlike the numerical aperture engineering, change of a light source to EUV demands development of its related components, such as photoresist and optics. Until a reliable solution for EUV lithography is developed, EUV interference lithography (EUVIL) would not solely advance the lithographic technology but would also help to optimize photoresist materials for EUV.
After achieving the 45-nm process, today's semiconductor industry is nearing the 20-nm process and looking for techniques that would enable sub-22-nm-half-pitch line patterns. Following the continuous increase in exposure tool numerical aperture, researchers are pursuing reductions in exposure wavelengths. This effort had them look at extreme ultraviolet (EUV: 13.4 nm in wavelength) as an exposure light source. Unlike the numerical aperture engineering, change of a light source to EUV demands development of its related components, such as photoresist and optics. Until a reliable solution for EUV lithography is developed, EUV interference lithography (EUVIL) would not solely advance the lithographic technology but would also help to optimize photoresist materials for EUV.
Jul 8th, 2010
 Given the massive interest and rapid developments in graphene research, scientists are now convinced that the controlled preparation of graphene-based materials with hierarchical and well-defined structures will pave the way for achieving high-performance applications of graphene in various technological fields such as optoelectronics, energy storage, polymer composites and catalysis. Self-assembly techniques have become some of the most effective strategies for this purpose. Although 2D self-assembly of graphene has been studied extensively from the perspectives of fundamental research and commercial applications, 3D self-assembly of 2D nanoscale graphene into functional macrostructures with well-defined networks remains as a great challenge and represents an important hurdle towards practical applications. Researchers in China have now provided a solution to this problem by demonstrating the successful preparation of self-assembled graphene hydrogel via a one-step hydrothermal process.
Given the massive interest and rapid developments in graphene research, scientists are now convinced that the controlled preparation of graphene-based materials with hierarchical and well-defined structures will pave the way for achieving high-performance applications of graphene in various technological fields such as optoelectronics, energy storage, polymer composites and catalysis. Self-assembly techniques have become some of the most effective strategies for this purpose. Although 2D self-assembly of graphene has been studied extensively from the perspectives of fundamental research and commercial applications, 3D self-assembly of 2D nanoscale graphene into functional macrostructures with well-defined networks remains as a great challenge and represents an important hurdle towards practical applications. Researchers in China have now provided a solution to this problem by demonstrating the successful preparation of self-assembled graphene hydrogel via a one-step hydrothermal process.
Jul 7th, 2010
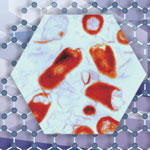 Researchers have made the surprising finding that graphene-based nanomaterials possess excellent antibacterial properties. Although antibacterial materials are widely used in daily life, and the antibacterial properties of nanomaterials are increasingly being explored and developed as commercial products, their cytotoxicity and biocompatibility has raised questions and concerns. Chinese researchers now found that graphene derivatives - graphene oxide, graphene oxide and reduced graphene oxide - can effectively inhibit bacterial growth. This is a significant finding as previous have proven that graphene, particularly graphene oxide, is biocompatible and cells can grow well on graphene substrates. Furthermore, while silver and silver nanoparticles have been well know to be antibacterial, they and other nanomaterials are often cytotoxic.
Researchers have made the surprising finding that graphene-based nanomaterials possess excellent antibacterial properties. Although antibacterial materials are widely used in daily life, and the antibacterial properties of nanomaterials are increasingly being explored and developed as commercial products, their cytotoxicity and biocompatibility has raised questions and concerns. Chinese researchers now found that graphene derivatives - graphene oxide, graphene oxide and reduced graphene oxide - can effectively inhibit bacterial growth. This is a significant finding as previous have proven that graphene, particularly graphene oxide, is biocompatible and cells can grow well on graphene substrates. Furthermore, while silver and silver nanoparticles have been well know to be antibacterial, they and other nanomaterials are often cytotoxic.
Jul 5th, 2010
 Here is a perfect example of how someone, who apparently doesn't understand or care much about the science, writes a sensational press release hyping nanotechnology by cherry-picking information and distorting issues. And all that to sell a product that doesn't even have to do with nanotechnology. Two days ago we ran a press release from Thomson Reuters about a brief report they compiled on patent data relating to nanotechnology in the cosmetics industry. Now, Thomson Reuters is in the business of selling information and information services products and applications. Their press release basically is advertising for their IP Market Reports. There is nothing wrong with that. What is very wrong, though, is the nonsense and unbalanced take on certain aspects on nanotechnologies. Let's take a closer look.
Here is a perfect example of how someone, who apparently doesn't understand or care much about the science, writes a sensational press release hyping nanotechnology by cherry-picking information and distorting issues. And all that to sell a product that doesn't even have to do with nanotechnology. Two days ago we ran a press release from Thomson Reuters about a brief report they compiled on patent data relating to nanotechnology in the cosmetics industry. Now, Thomson Reuters is in the business of selling information and information services products and applications. Their press release basically is advertising for their IP Market Reports. There is nothing wrong with that. What is very wrong, though, is the nonsense and unbalanced take on certain aspects on nanotechnologies. Let's take a closer look. 
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





