Showing Spotlights 345 - 352 of 624 in category All (newest first):
 It is not often that the prefix multipliers kilo and nano come together, and when they do, it usually is in the opening chapters of physical sciences textbooks where the point is made that the universe around us spans enormous space and time scales while operating in unimaginably small ones. We are truly awestruck and inspired by the tension. Kilometer-long nanowires do have a similar eponymous echo. Researchers have now reported the first successful fabrication of arrays of millions of ordered indefinitely long nanowires and nanotubes in a flexible polymer fiber. The results are kilometer-long nanowires - a novel approach to nanowire fabrication that might bring with it fresh solutions.
It is not often that the prefix multipliers kilo and nano come together, and when they do, it usually is in the opening chapters of physical sciences textbooks where the point is made that the universe around us spans enormous space and time scales while operating in unimaginably small ones. We are truly awestruck and inspired by the tension. Kilometer-long nanowires do have a similar eponymous echo. Researchers have now reported the first successful fabrication of arrays of millions of ordered indefinitely long nanowires and nanotubes in a flexible polymer fiber. The results are kilometer-long nanowires - a novel approach to nanowire fabrication that might bring with it fresh solutions.
Jun 23rd, 2011
 Radiation damage to materials is a major issue for builders of nuclear power plants as well as spacecraft engineers. The former have to worry about material failure due to the destructive radiation created within the reactor; the later are concerned about the exposure to space radiation of both materials and humans during long-term space missions. There is a consensus that interface engineering is the way to go to to improve resistance to extreme conditions in general and to reach radiation tolerance in particular - interfaces are places where defects created by energetic collision in solids can annihilate each other and thus render the material immune to irradiation. All kind of interfaces are being explored, in particular those formed by the contact between two different solid state phases.
Radiation damage to materials is a major issue for builders of nuclear power plants as well as spacecraft engineers. The former have to worry about material failure due to the destructive radiation created within the reactor; the later are concerned about the exposure to space radiation of both materials and humans during long-term space missions. There is a consensus that interface engineering is the way to go to to improve resistance to extreme conditions in general and to reach radiation tolerance in particular - interfaces are places where defects created by energetic collision in solids can annihilate each other and thus render the material immune to irradiation. All kind of interfaces are being explored, in particular those formed by the contact between two different solid state phases.
Jun 15th, 2011
 The future of your shirts, socks and gloves will be electronic. In years to come, wearable electronics will look nothing like even your smallest iPod or mobile phone today. Not only will such devices be embedded on textile substrates, but an electronics device or system could become the fabric itself. Here is some recent work that demonstrates the kind of issues scientists are working on today and that will help improve the performance of electronic textile structures. Using atomic layer deposition (ALD), researchers have grown coatings of inorganic materials on the surface of textiles like woven cotton and nonwoven polypropylene. By fabricating an all-fiber capacitor, they show that their coated materials are sufficiently conductive to perform in simple device architectures.
The future of your shirts, socks and gloves will be electronic. In years to come, wearable electronics will look nothing like even your smallest iPod or mobile phone today. Not only will such devices be embedded on textile substrates, but an electronics device or system could become the fabric itself. Here is some recent work that demonstrates the kind of issues scientists are working on today and that will help improve the performance of electronic textile structures. Using atomic layer deposition (ALD), researchers have grown coatings of inorganic materials on the surface of textiles like woven cotton and nonwoven polypropylene. By fabricating an all-fiber capacitor, they show that their coated materials are sufficiently conductive to perform in simple device architectures.
Jun 9th, 2011
 The small particles in volcanic ash can damage jet aircraft engines. During jet engine operation, large amounts of air are sucked in. If this air contains ash particles, especially glass-rich silicate ash, they will melt in the jet turbine's operating temperature of 1400-1500C and subsequently fuse to the turbine blades. This will drive the finely tuned blades out-of-balance and could stall the engine. In today's jet engines, a thermal barrier coating (TBC) insulates metallic engine parts from the heat generated by the combustion process. If ingested ash melts onto this coating it will penetrate it and, upon cooling, the molten ash forms a brittle glass that flakes off, taking the coating with it. A team of researchers have now examined a new class of ceramic TBC that could offer jet engines special protection against volcanic ash damage in the future.
The small particles in volcanic ash can damage jet aircraft engines. During jet engine operation, large amounts of air are sucked in. If this air contains ash particles, especially glass-rich silicate ash, they will melt in the jet turbine's operating temperature of 1400-1500C and subsequently fuse to the turbine blades. This will drive the finely tuned blades out-of-balance and could stall the engine. In today's jet engines, a thermal barrier coating (TBC) insulates metallic engine parts from the heat generated by the combustion process. If ingested ash melts onto this coating it will penetrate it and, upon cooling, the molten ash forms a brittle glass that flakes off, taking the coating with it. A team of researchers have now examined a new class of ceramic TBC that could offer jet engines special protection against volcanic ash damage in the future.
May 31st, 2011
 Microfluidic chips have proven to be a breakthrough analytical technique that has rendered analysis of proteins a medical routine. The sensitivity limits of immunoassays have been enhanced to picomolar concentrations using monoclonal antibodies, new labeling techniques, and devices for signal transduction and acquisition. It is now possible to routinely determine levels of hormones, cancer markers, response to infection with bacteria and viruses, monitor the evolution of a disease and test for medication levels. In a new review article, researchers argue that the most promising opportunities of microfluidics for diagnostics reside in point-of-care applications because a number of unmet needs can be fulfilled by microfluidic devices due to their portability, short sample processing time, and flexibility.
Microfluidic chips have proven to be a breakthrough analytical technique that has rendered analysis of proteins a medical routine. The sensitivity limits of immunoassays have been enhanced to picomolar concentrations using monoclonal antibodies, new labeling techniques, and devices for signal transduction and acquisition. It is now possible to routinely determine levels of hormones, cancer markers, response to infection with bacteria and viruses, monitor the evolution of a disease and test for medication levels. In a new review article, researchers argue that the most promising opportunities of microfluidics for diagnostics reside in point-of-care applications because a number of unmet needs can be fulfilled by microfluidic devices due to their portability, short sample processing time, and flexibility.
May 17th, 2011
 The potential use of antimicrobial surface coatings ranges from medicine, where medical device infection is associated with significant healthcare costs, to the construction industry and the food packaging industry. Thin films containing silver nanoparticles have been seen as promising candidate coatings. Silver is known as one of the oldest antimicrobial agents. Silver ions are thought to inhibit bacterial enzymes and bind to DNA. Silver nanomaterials have been used effectively against different bacteria, fungi and viruses. Using something like an advanced form of a rubber stamp, scientists have now developed a way to adhere an ultra-thin (just a few molecules thick) antibacterial coating to a wound. The "stamped" area shows bactericidal activity for at least 48 hours.
The potential use of antimicrobial surface coatings ranges from medicine, where medical device infection is associated with significant healthcare costs, to the construction industry and the food packaging industry. Thin films containing silver nanoparticles have been seen as promising candidate coatings. Silver is known as one of the oldest antimicrobial agents. Silver ions are thought to inhibit bacterial enzymes and bind to DNA. Silver nanomaterials have been used effectively against different bacteria, fungi and viruses. Using something like an advanced form of a rubber stamp, scientists have now developed a way to adhere an ultra-thin (just a few molecules thick) antibacterial coating to a wound. The "stamped" area shows bactericidal activity for at least 48 hours.
May 5th, 2011
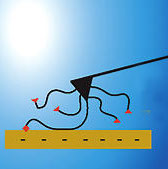 Glues adhere to solid materials via a multitude of fundamental physical or chemical interactions. Either chemical reaction times or solvent evaporation rates determine the point in time, when this interaction sets in and fixes the object to be glued. Electric potential has been used to attract polymers continuously to an electrode surface and to toggle molecules between states for a molecular switch. If you wanted to create electric glue, you would need to be able to control the interaction of a polymer and an electrode surface reversibly, thus creating a nanoscale system with electrochemically controlled adhesion. A research team now describes how Coulomb forces between polymers and surfaces may be measured, controlled, and manipulated.
Glues adhere to solid materials via a multitude of fundamental physical or chemical interactions. Either chemical reaction times or solvent evaporation rates determine the point in time, when this interaction sets in and fixes the object to be glued. Electric potential has been used to attract polymers continuously to an electrode surface and to toggle molecules between states for a molecular switch. If you wanted to create electric glue, you would need to be able to control the interaction of a polymer and an electrode surface reversibly, thus creating a nanoscale system with electrochemically controlled adhesion. A research team now describes how Coulomb forces between polymers and surfaces may be measured, controlled, and manipulated.
May 3rd, 2011
 Ink-jet printing of metal nanoparticles for conductive metal patterns has attracted great interest as an alternative to expensive fabrication techniques like vapor deposition. The bulk of the research in this area focuses on printing metal nanoparticle suspensions (metallic ink) for metallization. Printing conductive features by metallic nanoparticle inks must be followed by an additional step of sintering, usually achieved by heating to elevated temperatures. In this step, the nanoparticles composing the pattern will coalesce to form a continuous electrical contact. In new work, researchers have now demonstrated a new conductive ink that won't require a post printing sintering step. It is achieved by the addition of a latent sintering agent that gets into action after the printing step. Once the solvent evaporates, the sintering agent concentration increases, leading to the spontaneous sintering of the nanoparticles.
Ink-jet printing of metal nanoparticles for conductive metal patterns has attracted great interest as an alternative to expensive fabrication techniques like vapor deposition. The bulk of the research in this area focuses on printing metal nanoparticle suspensions (metallic ink) for metallization. Printing conductive features by metallic nanoparticle inks must be followed by an additional step of sintering, usually achieved by heating to elevated temperatures. In this step, the nanoparticles composing the pattern will coalesce to form a continuous electrical contact. In new work, researchers have now demonstrated a new conductive ink that won't require a post printing sintering step. It is achieved by the addition of a latent sintering agent that gets into action after the printing step. Once the solvent evaporates, the sintering agent concentration increases, leading to the spontaneous sintering of the nanoparticles.
Apr 18th, 2011
 It is not often that the prefix multipliers kilo and nano come together, and when they do, it usually is in the opening chapters of physical sciences textbooks where the point is made that the universe around us spans enormous space and time scales while operating in unimaginably small ones. We are truly awestruck and inspired by the tension. Kilometer-long nanowires do have a similar eponymous echo. Researchers have now reported the first successful fabrication of arrays of millions of ordered indefinitely long nanowires and nanotubes in a flexible polymer fiber. The results are kilometer-long nanowires - a novel approach to nanowire fabrication that might bring with it fresh solutions.
It is not often that the prefix multipliers kilo and nano come together, and when they do, it usually is in the opening chapters of physical sciences textbooks where the point is made that the universe around us spans enormous space and time scales while operating in unimaginably small ones. We are truly awestruck and inspired by the tension. Kilometer-long nanowires do have a similar eponymous echo. Researchers have now reported the first successful fabrication of arrays of millions of ordered indefinitely long nanowires and nanotubes in a flexible polymer fiber. The results are kilometer-long nanowires - a novel approach to nanowire fabrication that might bring with it fresh solutions.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





