Showing Spotlights 217 - 224 of 544 in category All (newest first):
 Advanced health monitoring systems and healthcare devices will become an integral part of the Internet of Things. As a harbinger of things to come, nanotechnology researchers have now demonstrated a smart thermal patch which can be used for thermotherapy for pain management in a user interactive way. To fabricate the device, the researchers used CMOS technology to devise a silicon based smart thermal patch which is flexible and stretchable.
Advanced health monitoring systems and healthcare devices will become an integral part of the Internet of Things. As a harbinger of things to come, nanotechnology researchers have now demonstrated a smart thermal patch which can be used for thermotherapy for pain management in a user interactive way. To fabricate the device, the researchers used CMOS technology to devise a silicon based smart thermal patch which is flexible and stretchable.
Dec 9th, 2014
 Studying the complex wiring of neural circuits and identifying the details of how individual neural circuits operate in epilepsy and other neurological disorders requires real-time observation of their locations, firing patterns, and other factors. These observations depend on high-resolution optical imaging and electrophysiological recording. Researchers have now developed a completely transparent graphene microelectrode that allows for simultaneous optical imaging and electrophysiological recordings of neural circuits.
Studying the complex wiring of neural circuits and identifying the details of how individual neural circuits operate in epilepsy and other neurological disorders requires real-time observation of their locations, firing patterns, and other factors. These observations depend on high-resolution optical imaging and electrophysiological recording. Researchers have now developed a completely transparent graphene microelectrode that allows for simultaneous optical imaging and electrophysiological recordings of neural circuits.
Nov 13th, 2014
 The majority of men who undergo radical prostatectomy for the treatment of prostate cancer will suffer from erectile dysfunction due to disruption of the cavernous nerve. This nerve has been identified as responsible for penile erection. The oral erectogenic PDE5 inhibitors like Viagra rely on the functioning of this nerve to provide the initial burst of nitric oxide necessary to initiate an erection. In this condition nanotechnology - in the form of a nanoparticle delivery system - may come to the rescue by targetting useful therapeutics for penile rehabilitation following radical prostatectomy.
The majority of men who undergo radical prostatectomy for the treatment of prostate cancer will suffer from erectile dysfunction due to disruption of the cavernous nerve. This nerve has been identified as responsible for penile erection. The oral erectogenic PDE5 inhibitors like Viagra rely on the functioning of this nerve to provide the initial burst of nitric oxide necessary to initiate an erection. In this condition nanotechnology - in the form of a nanoparticle delivery system - may come to the rescue by targetting useful therapeutics for penile rehabilitation following radical prostatectomy.
Oct 24th, 2014
 There is a significant need for new therapeutic approaches to combat diseases such as cancer and viral infections. Using RNA as a therapeutic modality brings to bear an entirely new approach, which not only allows for the construction of uniform scaffolds for attachment of functional entities, but also permits the use of all the different types of functionalities that are inherent in natural RNAs. New research demonstrates that multifunctional RNA nanoparticles with a nanoring design allow the use of different types of functionalities inherent in natural RNAs.
There is a significant need for new therapeutic approaches to combat diseases such as cancer and viral infections. Using RNA as a therapeutic modality brings to bear an entirely new approach, which not only allows for the construction of uniform scaffolds for attachment of functional entities, but also permits the use of all the different types of functionalities that are inherent in natural RNAs. New research demonstrates that multifunctional RNA nanoparticles with a nanoring design allow the use of different types of functionalities inherent in natural RNAs.
Oct 13th, 2014
 Researchers have explored the use of curcumin nanoparticles for the treatment of infected burn wounds, an application that resulted in reduced bacterial load and enhancing wound healing. Adding to the excitement regarding curcumin in multiple fields of medicine, most prominently in oncology, these new findings demonstrat that curcumin nanoparticles were more effective at both accelerating thermal burn wound closure and clearing infection with Methicillin Resistant S. aureus (MRSA) as compared to curcumin in its bulk size.
Researchers have explored the use of curcumin nanoparticles for the treatment of infected burn wounds, an application that resulted in reduced bacterial load and enhancing wound healing. Adding to the excitement regarding curcumin in multiple fields of medicine, most prominently in oncology, these new findings demonstrat that curcumin nanoparticles were more effective at both accelerating thermal burn wound closure and clearing infection with Methicillin Resistant S. aureus (MRSA) as compared to curcumin in its bulk size.
Sep 22nd, 2014
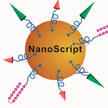 Gene transcription is tightly regulated by proteins called transcription factors. These transcription factor (TF) proteins are master regulators of transcriptional activity and gene expression. Transcription factors are responsible for transcribing the correct genes and therefore for producing the right quantity of proteins. TF-based gene regulation is a promising approach for many biological applications, however, several limitations hinder the full potential of TFs. To overcome these problems, an international team of researchers has developed an artificial, nanoparticle-based transcription factor, termed NanoScript, which is designed to mimic the structure and function of TFs.
Gene transcription is tightly regulated by proteins called transcription factors. These transcription factor (TF) proteins are master regulators of transcriptional activity and gene expression. Transcription factors are responsible for transcribing the correct genes and therefore for producing the right quantity of proteins. TF-based gene regulation is a promising approach for many biological applications, however, several limitations hinder the full potential of TFs. To overcome these problems, an international team of researchers has developed an artificial, nanoparticle-based transcription factor, termed NanoScript, which is designed to mimic the structure and function of TFs.
Aug 28th, 2014
 Researchers have demonstrated an active glucose-responsive self-powered fluidic pump based on transesterification reaction of acyclic diol boronate with glucose. The scientific principle of the project is to use well-known glucose/boronate chemistry to design a self-powered micropump device. Instead of synthesizing some new molecules with glucose/boronate reaction, a miniature pump utilizes the energy of this chemical reaction and pumps drugs when glucose levels are high.
Researchers have demonstrated an active glucose-responsive self-powered fluidic pump based on transesterification reaction of acyclic diol boronate with glucose. The scientific principle of the project is to use well-known glucose/boronate chemistry to design a self-powered micropump device. Instead of synthesizing some new molecules with glucose/boronate reaction, a miniature pump utilizes the energy of this chemical reaction and pumps drugs when glucose levels are high.
Aug 19th, 2014
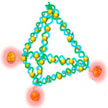 One way to eliminate the toxicity issue of synthetic nanomaterials used in nanomedicine is by working with truly biocompatible natural carriers for sensing and drug delivery applications. The emerging field of DNA nanotechnology may provide a solution. In new work, researchers have developed a novel theranostic platform which is made by utilizing a self-assembled DNA nanopyramid as scaffold for incorporation of both detection and therapeutic moieties to combat bacterial infection.
One way to eliminate the toxicity issue of synthetic nanomaterials used in nanomedicine is by working with truly biocompatible natural carriers for sensing and drug delivery applications. The emerging field of DNA nanotechnology may provide a solution. In new work, researchers have developed a novel theranostic platform which is made by utilizing a self-assembled DNA nanopyramid as scaffold for incorporation of both detection and therapeutic moieties to combat bacterial infection.
Jul 11th, 2014
 Advanced health monitoring systems and healthcare devices will become an integral part of the Internet of Things. As a harbinger of things to come, nanotechnology researchers have now demonstrated a smart thermal patch which can be used for thermotherapy for pain management in a user interactive way. To fabricate the device, the researchers used CMOS technology to devise a silicon based smart thermal patch which is flexible and stretchable.
Advanced health monitoring systems and healthcare devices will become an integral part of the Internet of Things. As a harbinger of things to come, nanotechnology researchers have now demonstrated a smart thermal patch which can be used for thermotherapy for pain management in a user interactive way. To fabricate the device, the researchers used CMOS technology to devise a silicon based smart thermal patch which is flexible and stretchable.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





