Showing Spotlights 193 - 200 of 544 in category All (newest first):
 The manufacture of nanoparticles has reached a very high level of control of their shape, size and chemical nature. However, assembling nanoparticles in a controlled manner and with clearly defined functionalities in three-dimensional space remains quite a challenge. Researchers have now taken a first step towards the goal of protein-driven assembly of nanoparticles. In this ground-breaking work, they show that gold nanoparticles with a diameter of 10nm can be assembled using two different protein pairs.
The manufacture of nanoparticles has reached a very high level of control of their shape, size and chemical nature. However, assembling nanoparticles in a controlled manner and with clearly defined functionalities in three-dimensional space remains quite a challenge. Researchers have now taken a first step towards the goal of protein-driven assembly of nanoparticles. In this ground-breaking work, they show that gold nanoparticles with a diameter of 10nm can be assembled using two different protein pairs.
Mar 8th, 2016
 Many virus detection platforms, including conventional fluorescent label-based ones, have limitations because they are time-intensive and not easily compatible with point-of-care use without the existence of significant infrastructure and expert staff. Researchers have now developed a technique capable of specifically visualizing label-free single viruses in complex solutions in real-time. This approach eliminates virtually all sample preparation.
Many virus detection platforms, including conventional fluorescent label-based ones, have limitations because they are time-intensive and not easily compatible with point-of-care use without the existence of significant infrastructure and expert staff. Researchers have now developed a technique capable of specifically visualizing label-free single viruses in complex solutions in real-time. This approach eliminates virtually all sample preparation.
Feb 5th, 2016
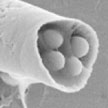 The goal of a vast amount of nanomedicine research is the perfect drug carrier: it is injected into the body and transports itself to the correct target, such as a tumor, and delivers the required therapeutic drug dose at this target. This idealized concept was first proposed at the beginning of the 20th century and was nicknamed the 'magic bullet' concept. Taking this 'bullet' concept literally, researchers have developed acoustically triggered microcannons, capable of versatile loading and effective firing of nanobullets, as novel tools toward advancing microscale tissue penetration of therapeutic payloads.
The goal of a vast amount of nanomedicine research is the perfect drug carrier: it is injected into the body and transports itself to the correct target, such as a tumor, and delivers the required therapeutic drug dose at this target. This idealized concept was first proposed at the beginning of the 20th century and was nicknamed the 'magic bullet' concept. Taking this 'bullet' concept literally, researchers have developed acoustically triggered microcannons, capable of versatile loading and effective firing of nanobullets, as novel tools toward advancing microscale tissue penetration of therapeutic payloads.
Jan 5th, 2016
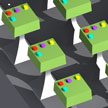 Researchers have developed a suspended planar-array chip whose in situ capabilities with a spatial molecular-probe arrangement combine the advantages of both suspended arrays and planar arrays. This opens the way towards the multiplexed detection of intracellular biological parameters using a single device in dramatically reduced volumes, such as inside a living HeLa cell. The chip's volume represents only about 0.35% of the total volume of a typical HeLa cell.
Researchers have developed a suspended planar-array chip whose in situ capabilities with a spatial molecular-probe arrangement combine the advantages of both suspended arrays and planar arrays. This opens the way towards the multiplexed detection of intracellular biological parameters using a single device in dramatically reduced volumes, such as inside a living HeLa cell. The chip's volume represents only about 0.35% of the total volume of a typical HeLa cell.
Dec 29th, 2015
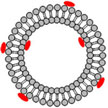 Researchers have demonstrated a system that provides photo-triggered release of local anesthetics in a manner that could be adjusted by varying the irradiance and the duration of irradiation. From the clinical point of view, this is important in that it demonstrates a method by which patients would be able to take control of relatively local pain, being able to deliver local analgesia on demand, for the duration and with the intensity desired.
Researchers have demonstrated a system that provides photo-triggered release of local anesthetics in a manner that could be adjusted by varying the irradiance and the duration of irradiation. From the clinical point of view, this is important in that it demonstrates a method by which patients would be able to take control of relatively local pain, being able to deliver local analgesia on demand, for the duration and with the intensity desired.
Dec 17th, 2015
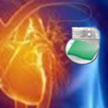 Adding to the options for wirelessly powering implants from outside the body, researchers are proposing a light-driven powering device using near infrared rays (nIR). Flashing light impulses, which are absorbed by the device, induce temperature fluctuation, thus generating voltage/current pulses which can be used for charging a battery or biological stimulations. This flexible and compact device can generate electrical pulses with controllable amplitude and width when remotely irradiated by nIR. Not only can it supply power to implantable bioelectronics, but it also provides adjustable electrical pulses for nerve stimulation.
Adding to the options for wirelessly powering implants from outside the body, researchers are proposing a light-driven powering device using near infrared rays (nIR). Flashing light impulses, which are absorbed by the device, induce temperature fluctuation, thus generating voltage/current pulses which can be used for charging a battery or biological stimulations. This flexible and compact device can generate electrical pulses with controllable amplitude and width when remotely irradiated by nIR. Not only can it supply power to implantable bioelectronics, but it also provides adjustable electrical pulses for nerve stimulation.
Nov 6th, 2015
 Previous reports have shown that when nanoparticles enter the blood stream, the proteins that adsorb onto nanoparticles when they enter the body form a protein corona that hinders interactions between the targeting ligands on the nanoparticles and their binding partners on the cells' surface. To address this issue, scientists have developed a strategy that enables directing the formation of protein coronas on nanoparticles that are enriched in plasma proteins with natural targeting capabilities.
Previous reports have shown that when nanoparticles enter the blood stream, the proteins that adsorb onto nanoparticles when they enter the body form a protein corona that hinders interactions between the targeting ligands on the nanoparticles and their binding partners on the cells' surface. To address this issue, scientists have developed a strategy that enables directing the formation of protein coronas on nanoparticles that are enriched in plasma proteins with natural targeting capabilities.
Oct 28th, 2015
 The complexity of the microenvironment of a biological cell is influenced by many factors, including surface topography and chemistry; matrix stiffness; mechanical stress; molecular liquid composition and other physiochemical parameters. However, most artificial biointerfaces are developed based on just a single chemical or physical factor to direct cell behaviors. The functions performed by these artificial biointerfaces are far simpler than those performed in the natural cell microenvironment. In an effort to more closely mimic a cell's natural environment, researchers have fabricated an antibody modified reduced graphene oxide platform and used it to significantly improve the efficiency for capturing circulating tumor cells.
The complexity of the microenvironment of a biological cell is influenced by many factors, including surface topography and chemistry; matrix stiffness; mechanical stress; molecular liquid composition and other physiochemical parameters. However, most artificial biointerfaces are developed based on just a single chemical or physical factor to direct cell behaviors. The functions performed by these artificial biointerfaces are far simpler than those performed in the natural cell microenvironment. In an effort to more closely mimic a cell's natural environment, researchers have fabricated an antibody modified reduced graphene oxide platform and used it to significantly improve the efficiency for capturing circulating tumor cells.
Oct 14th, 2015
 The manufacture of nanoparticles has reached a very high level of control of their shape, size and chemical nature. However, assembling nanoparticles in a controlled manner and with clearly defined functionalities in three-dimensional space remains quite a challenge. Researchers have now taken a first step towards the goal of protein-driven assembly of nanoparticles. In this ground-breaking work, they show that gold nanoparticles with a diameter of 10nm can be assembled using two different protein pairs.
The manufacture of nanoparticles has reached a very high level of control of their shape, size and chemical nature. However, assembling nanoparticles in a controlled manner and with clearly defined functionalities in three-dimensional space remains quite a challenge. Researchers have now taken a first step towards the goal of protein-driven assembly of nanoparticles. In this ground-breaking work, they show that gold nanoparticles with a diameter of 10nm can be assembled using two different protein pairs.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





