| Posted: Jan 05, 2016 | |
Microcannons that fire drug-loaded nanobullets |
|
| (Nanowerk Spotlight) The goal of a vast amount of nanomedicine research is the perfect drug carrier: it is injected into the body and transports itself to the correct target, such as a tumor, and delivers the required therapeutic drug dose at this target. This idealized concept was first proposed by German physician Paul Ehrlich at the beginning of the 20th century and was nicknamed the 'magic bullet' concept. | |
| Taking this 'bullet' concept literally, Joseph Wang and Sadik Esener, professors of nanoengineering at UC San Diego, and their teams have developed acoustically triggered microcannons, capable of versatile loading and effective firing of nanobullets, as novel tools toward advancing microscale tissue penetration of therapeutic payloads. | |
| "Designing the right tool for efficient tissue penetration at small scales is important for diverse biomedical applications in drug delivery and microsurgery applications," Wang tells Nanowerk. "Our new nanobullets could eventually be used to drive drugs directly deep into diseased tissues." | |
| The team reported their findings in the December 21, 2015 online edition of ACS Nano ("Acoustic Microcannons: Toward Advanced Microballistics"). | |
 |
|
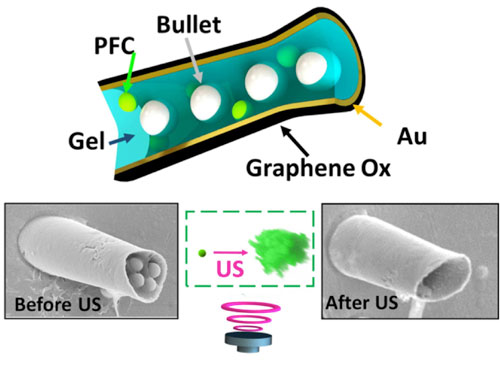
| Ultrasound (US)-triggered nanobullets firing based on vaporization of PFC droplets as a propulsion system. (Image: Department of Nanoengineering, UC San Diego) | |
| "Ultrasound-triggered vaporization of perfluorocarbon micro-emulsion is an extremely attractive candidate for externally triggering the actuation of micro/nanoscale ballistic tools," says Wang. "This technique is biocompatible and has been previously used to enhance the permeation and delivery of therapeutics into blood vessels and tissue. We expect that our new ultrasound-based ballistic approach could lead to efficient delivery devices capable of delivering a wide range of payloads deep into an identified target." | |
| To construct their device, the team first fabricated the hollowed cone structure of the microcannon using template electrodeposition of electrochemically reduced graphene oxide and gold on the walls of micropores in a polycarbonate membrane. | |
| They then infiltrated a liquid gel matrix by gravitational and capillarity forces into the hollow microcannons anchored in the template membrane. Entrapped in this gel were 1 µm silica nanobullets; it also contained a perfluorocarbon (PFC) emulsion. | |
| "Upon application of a focused ultrasound pulse by a piezoelectric transducer, the nanobullets are ejected rapidly from the microcannon," explains Wang. "The mechanism responsible for the propulsion thrust of the nanobullets relies on the momentum associated with the ultrasound-induced spontaneous vaporization of the PFC emulsion droplet into a rapidly expanding microbubble." | |
| The firing of a large amount nanobullets has been modeled by the team theoretically and demonstrated experimentally. The experimental data supports the theory that the ejection of nanobullets from the microcannon occurs due to vaporization of perfluorocarbon fluid, presenting remarkably high power, fast displacement speed and a large tissue penetration depth. | |
| The creation of a functioning microscale cannon will allow the efficient loading and firing of nanoscale therapeutic and imaging cargoes as nanoprojectiles deep into biological tissues. | |
| Future work by the team will aim at developing this microbullet platform technology for delivering a wide range of therapeutic payloads – including co-delivery of multiple drugs – and expanding the practical utility of the acoustic microcannons, for their use as single or array devices for transporting drug cocktails and vaccines, respectively. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
