Showing Spotlights 233 - 240 of 544 in category All (newest first):
 For years, scientists and engineers have worked to design electronics which can interface with the body. However, typical silicon wafer-based electronics, which are planar and stiff, are not suited to interface with the soft, curvilinear, and dynamic environment that biology presents. By exploiting the features of shape-memory polymer (SMP) substrates, an international team of researchers has now demonstrated a unique form of adaptive electronics which softly conform or deploy into 3D shapes after exposure to a stimulus. The resulting organic thin-film transistors (OTFTs) can change their mechanical properties from rigid and planar, to soft and compliant, in order to enable soft and conformal wrapping around 3D objects, including biological tissue.
For years, scientists and engineers have worked to design electronics which can interface with the body. However, typical silicon wafer-based electronics, which are planar and stiff, are not suited to interface with the soft, curvilinear, and dynamic environment that biology presents. By exploiting the features of shape-memory polymer (SMP) substrates, an international team of researchers has now demonstrated a unique form of adaptive electronics which softly conform or deploy into 3D shapes after exposure to a stimulus. The resulting organic thin-film transistors (OTFTs) can change their mechanical properties from rigid and planar, to soft and compliant, in order to enable soft and conformal wrapping around 3D objects, including biological tissue.
May 7th, 2014
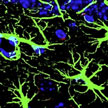 Prevention and treatment of neurological disorders in humans necessitate delivery of therapeutic or neuroprotective agents across the so-called blood-brain barrier (BBB) into the brain. The scarcity of techniques for brain-specific delivery of therapeutic molecules using non-invasive approaches has led researchers to increasingly explore the promising potential of nanotechnology toward the diagnosis and treatment of diseases/disorders incurable with present techniques. A recent example of these efforts is the research to analyze the intra- and intercellular transport and fate of novel nanoparticles for drug delivery to the central nervous system.
Prevention and treatment of neurological disorders in humans necessitate delivery of therapeutic or neuroprotective agents across the so-called blood-brain barrier (BBB) into the brain. The scarcity of techniques for brain-specific delivery of therapeutic molecules using non-invasive approaches has led researchers to increasingly explore the promising potential of nanotechnology toward the diagnosis and treatment of diseases/disorders incurable with present techniques. A recent example of these efforts is the research to analyze the intra- and intercellular transport and fate of novel nanoparticles for drug delivery to the central nervous system.
May 1st, 2014
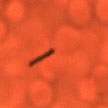 While nanotechnology researchers have made great progress over the past few years in developing self-propelled nano objects, these tiny devices still fall far short of what their natural counterparts' performance. Today, artificial nanomotors lack the sophisticated functionality of biomotors and are limited to a very narrow range of environments and fuels. In another step towards realizing the vision of tiny vessels roaming around in human blood vessels working as surgical nanorobots, researchers have now demonstrated, for the first time, externally driven nanomotors that move in undiluted human blood.
While nanotechnology researchers have made great progress over the past few years in developing self-propelled nano objects, these tiny devices still fall far short of what their natural counterparts' performance. Today, artificial nanomotors lack the sophisticated functionality of biomotors and are limited to a very narrow range of environments and fuels. In another step towards realizing the vision of tiny vessels roaming around in human blood vessels working as surgical nanorobots, researchers have now demonstrated, for the first time, externally driven nanomotors that move in undiluted human blood.
Apr 17th, 2014
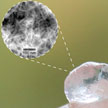 Nanocellulose from wood is a promising nanomaterial with potential applications as a substrate for printing electronics, filtration, or biomedicine. Researchers have now reported on a method to control the surface chemistry of nanocellulose. They fabricated nanocellulose gels that have a significantly higher swelling degree in neutral and alkaline conditions, compared to an acid environment. This material could be of great interest for critical wound healing applications.
Nanocellulose from wood is a promising nanomaterial with potential applications as a substrate for printing electronics, filtration, or biomedicine. Researchers have now reported on a method to control the surface chemistry of nanocellulose. They fabricated nanocellulose gels that have a significantly higher swelling degree in neutral and alkaline conditions, compared to an acid environment. This material could be of great interest for critical wound healing applications.
Apr 10th, 2014
 Nanomaterials for nanomedicine and biological applications are often two-component structures - referred to as 'nanoconstructs' -consisting of a 'hard' nanoparticle core and a 'soft' shell of biomolecular ligands. Researchers have now demonstrated a nanoconstruct with enhanced in vitro efficacy. This highly loaded nanoconstruct was taken up by pancreatic cancer cells and fibrosarcoma cells at fast rates. The team found that the increased loading of Apt on AuNS also resulted in an enhanced in vitro response.
Nanomaterials for nanomedicine and biological applications are often two-component structures - referred to as 'nanoconstructs' -consisting of a 'hard' nanoparticle core and a 'soft' shell of biomolecular ligands. Researchers have now demonstrated a nanoconstruct with enhanced in vitro efficacy. This highly loaded nanoconstruct was taken up by pancreatic cancer cells and fibrosarcoma cells at fast rates. The team found that the increased loading of Apt on AuNS also resulted in an enhanced in vitro response.
Apr 9th, 2014
 Scientists have great expectations that nanotechnologies will bring them closer to the goal of creating computer systems that can simulate and emulate the brain's abilities for sensation, perception, action, interaction and cognition while rivaling its low power consumption and compact size - basically a brain-on-a-chip. Already, scientists are working hard on laying the foundations for what is called neuromorphic engineering - a new interdisciplinary discipline that includes nanotechnologies and whose goal is to design artificial neural systems with physical architectures similar to biological nervous systems.
Scientists have great expectations that nanotechnologies will bring them closer to the goal of creating computer systems that can simulate and emulate the brain's abilities for sensation, perception, action, interaction and cognition while rivaling its low power consumption and compact size - basically a brain-on-a-chip. Already, scientists are working hard on laying the foundations for what is called neuromorphic engineering - a new interdisciplinary discipline that includes nanotechnologies and whose goal is to design artificial neural systems with physical architectures similar to biological nervous systems.
Apr 4th, 2014
 Ice accumulation is not just a cost and safety problem for the airline industry and other transportation areas, together with undesired environmental impacts. Here are some examples: Transmission lines and power network towers may deform or even collapse with the burden of excess amount of ice; ice accretion on wind turbine blades can cause a production loss as much as 50% of the annual production; frost and ice accumulation in refrigerators and heat exchangers results in a decrease of heat transfer efficiency. Therefore, great efforts have been made to understand the mechanism of icing and investigations on anti-icing and deicing have been extensively carried out. Various anti-icing and deicing methods have been developed such as for instance nanocoatings and other nanostructured surfaces.
Ice accumulation is not just a cost and safety problem for the airline industry and other transportation areas, together with undesired environmental impacts. Here are some examples: Transmission lines and power network towers may deform or even collapse with the burden of excess amount of ice; ice accretion on wind turbine blades can cause a production loss as much as 50% of the annual production; frost and ice accumulation in refrigerators and heat exchangers results in a decrease of heat transfer efficiency. Therefore, great efforts have been made to understand the mechanism of icing and investigations on anti-icing and deicing have been extensively carried out. Various anti-icing and deicing methods have been developed such as for instance nanocoatings and other nanostructured surfaces.
Mar 18th, 2014
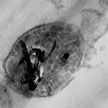 The investigation of effects of engineered nanomaterials on endothelial cells - which form the inner lining of blood vessels - is a critical safety issue. Already, various engineered nanomaterials are being designed for biomedical applications for intravascular use and other nanomaterials may reach the vasculature as a result of occupational, environmental, or other types of exposure. Researchers have now elucidated the mechanism of cytotoxicity of carboxylated MWCNTs on cultured endothelial cells and they show a new potential way of pharmacological cytoprotection against cytotoxic effect of carboxylated MWCNTs.
The investigation of effects of engineered nanomaterials on endothelial cells - which form the inner lining of blood vessels - is a critical safety issue. Already, various engineered nanomaterials are being designed for biomedical applications for intravascular use and other nanomaterials may reach the vasculature as a result of occupational, environmental, or other types of exposure. Researchers have now elucidated the mechanism of cytotoxicity of carboxylated MWCNTs on cultured endothelial cells and they show a new potential way of pharmacological cytoprotection against cytotoxic effect of carboxylated MWCNTs.
Mar 12th, 2014
 For years, scientists and engineers have worked to design electronics which can interface with the body. However, typical silicon wafer-based electronics, which are planar and stiff, are not suited to interface with the soft, curvilinear, and dynamic environment that biology presents. By exploiting the features of shape-memory polymer (SMP) substrates, an international team of researchers has now demonstrated a unique form of adaptive electronics which softly conform or deploy into 3D shapes after exposure to a stimulus. The resulting organic thin-film transistors (OTFTs) can change their mechanical properties from rigid and planar, to soft and compliant, in order to enable soft and conformal wrapping around 3D objects, including biological tissue.
For years, scientists and engineers have worked to design electronics which can interface with the body. However, typical silicon wafer-based electronics, which are planar and stiff, are not suited to interface with the soft, curvilinear, and dynamic environment that biology presents. By exploiting the features of shape-memory polymer (SMP) substrates, an international team of researchers has now demonstrated a unique form of adaptive electronics which softly conform or deploy into 3D shapes after exposure to a stimulus. The resulting organic thin-film transistors (OTFTs) can change their mechanical properties from rigid and planar, to soft and compliant, in order to enable soft and conformal wrapping around 3D objects, including biological tissue.

 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





