Showing Spotlights 249 - 256 of 546 in category All (newest first):
 Researchers demonstrate a strategy for the fabrication of memristive nanodevices with stable and tunable performance by assembling ferritin monolayer inside a on-wire lithography-generated 12 nm gap. This work work uses the unique high iron loading capacity of Archaeoglobus fulgidus ferritin. The iron loading in the nanocages drastically impacts the performance of the memristive devices. The higher iron loading amount contributes to better memristive performance due to higher electrochemical activity of the ferric complex core.
Researchers demonstrate a strategy for the fabrication of memristive nanodevices with stable and tunable performance by assembling ferritin monolayer inside a on-wire lithography-generated 12 nm gap. This work work uses the unique high iron loading capacity of Archaeoglobus fulgidus ferritin. The iron loading in the nanocages drastically impacts the performance of the memristive devices. The higher iron loading amount contributes to better memristive performance due to higher electrochemical activity of the ferric complex core.
Dec 16th, 2013
 Advances in materials, fabrication strategies and device designs for flexible and stretchable electronics and sensors make it possible to envision a not-too-distant future where ultra-thin, flexible circuits based on inorganic semiconductors can be wrapped and attached to any imaginable surface, including body parts and even internal organs. Robotic technologies will also benefit as it becomes possible to fabricate electronic skin ('e-skin') that, for instance, could allow surgical robots to interact, in a soft contacting mode, with their surroundings through touch.
Advances in materials, fabrication strategies and device designs for flexible and stretchable electronics and sensors make it possible to envision a not-too-distant future where ultra-thin, flexible circuits based on inorganic semiconductors can be wrapped and attached to any imaginable surface, including body parts and even internal organs. Robotic technologies will also benefit as it becomes possible to fabricate electronic skin ('e-skin') that, for instance, could allow surgical robots to interact, in a soft contacting mode, with their surroundings through touch.
Nov 15th, 2013
 Advances in micro- and nanoscale engineering in the medical field have led to the development of various robotic designs that one day will allow a new level of minimally invasive medicine. These micro- and nanorobots will be able to reach a targeted area, provide treatments and therapies for a desired duration, measure the effects and, at the conclusion of the treatment, be removed or degrade without causing adverse effects. Ideally, all these tasks would be automated but they could also be performed under the direct supervision and control of an external user.
Advances in micro- and nanoscale engineering in the medical field have led to the development of various robotic designs that one day will allow a new level of minimally invasive medicine. These micro- and nanorobots will be able to reach a targeted area, provide treatments and therapies for a desired duration, measure the effects and, at the conclusion of the treatment, be removed or degrade without causing adverse effects. Ideally, all these tasks would be automated but they could also be performed under the direct supervision and control of an external user.
Nov 13th, 2013
 Theranostics - a combination of the words therapeutics and diagnostics - describes a treatment platform that combines a diagnostic test with targeted therapy based on the test results, i.e. a step towards personalized medicine. Theranostic nanomedicine has the potential for simultaneous and real time monitoring of drug delivery, trafficking of drug and therapeutic responses. Researchers have now demonstrated for the first time a MRI-visual order-disorder micellar nanostructures for smart cancer theranostics.
Theranostics - a combination of the words therapeutics and diagnostics - describes a treatment platform that combines a diagnostic test with targeted therapy based on the test results, i.e. a step towards personalized medicine. Theranostic nanomedicine has the potential for simultaneous and real time monitoring of drug delivery, trafficking of drug and therapeutic responses. Researchers have now demonstrated for the first time a MRI-visual order-disorder micellar nanostructures for smart cancer theranostics.
Nov 12th, 2013
 Material science is having a renewed influence on bioelectronics design beyond the incorporation of new functional nanomaterials. This newly established cooperation opens new windows for bioelectronics research, especially for fabricating flexible and smart devices. Recent advances in graphene research provide various possibilities to enhance performance characteristics and current approaches to design new bio-devices. Especially, smart and flexible bioelectronics on graphene has emerged as a new frontier in this area.
Material science is having a renewed influence on bioelectronics design beyond the incorporation of new functional nanomaterials. This newly established cooperation opens new windows for bioelectronics research, especially for fabricating flexible and smart devices. Recent advances in graphene research provide various possibilities to enhance performance characteristics and current approaches to design new bio-devices. Especially, smart and flexible bioelectronics on graphene has emerged as a new frontier in this area.
Oct 31st, 2013
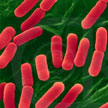 As the use of antibiotics increases for medical, veterinary and agricultural purposes, the increasing emergence of antibiotic-resistant strains of pathogenic bacteria is an unwelcome consequence. The incidence of the multidrug resistance (MDR) of bacteria which cause infections in hospitals/intensive care units is increasing, and finding microorganisms insensitive to more than 10 different antibiotics is not unusual. The emergence of superbugs has made it imperative to search for novel methods, which can combat the microbial resistance. Thus, application of nanotechnology in pharmaceuticals and microbiology is gaining importance to prevent the catastrophic consequences of antibiotic resistance.
As the use of antibiotics increases for medical, veterinary and agricultural purposes, the increasing emergence of antibiotic-resistant strains of pathogenic bacteria is an unwelcome consequence. The incidence of the multidrug resistance (MDR) of bacteria which cause infections in hospitals/intensive care units is increasing, and finding microorganisms insensitive to more than 10 different antibiotics is not unusual. The emergence of superbugs has made it imperative to search for novel methods, which can combat the microbial resistance. Thus, application of nanotechnology in pharmaceuticals and microbiology is gaining importance to prevent the catastrophic consequences of antibiotic resistance.
Sep 10th, 2013
 Conventional carbon-fiber electrodes have been the material of choice for identifying the chemical nature of neurotransmitters in the brain. Unfortunately, they have some limitations that leave some of the molecules that researchers are interested in just out of our reach. Further miniaturization of biologically compatible, carbon based electrode materials to the nanoscale promises to enhance the very characteristics that made microelectrodes so transformative in the first place, enabling high speed measurements in discrete spatial locations.
Conventional carbon-fiber electrodes have been the material of choice for identifying the chemical nature of neurotransmitters in the brain. Unfortunately, they have some limitations that leave some of the molecules that researchers are interested in just out of our reach. Further miniaturization of biologically compatible, carbon based electrode materials to the nanoscale promises to enhance the very characteristics that made microelectrodes so transformative in the first place, enabling high speed measurements in discrete spatial locations.
Sep 3rd, 2013
 Researchers report on a novel targeted drug-delivery vehicle for cancer therapy, which can selectively target the tumor niche while delivering an array of therapeutic agents. This targeting platform is based on unique vesicles ('nanoghosts') that are produced, for the first time, from intact cell membranes of stem cells with inherent homing abilities, and which may be loaded with different therapeutics. The team showed that such vesicles, encompassing the cell surface molecules and preserving the targeting mechanism of the cells from which they were made, can outperform conventional delivery systems based on liposomes or nanoparticles.
Researchers report on a novel targeted drug-delivery vehicle for cancer therapy, which can selectively target the tumor niche while delivering an array of therapeutic agents. This targeting platform is based on unique vesicles ('nanoghosts') that are produced, for the first time, from intact cell membranes of stem cells with inherent homing abilities, and which may be loaded with different therapeutics. The team showed that such vesicles, encompassing the cell surface molecules and preserving the targeting mechanism of the cells from which they were made, can outperform conventional delivery systems based on liposomes or nanoparticles.
Jul 26th, 2013
 Researchers demonstrate a strategy for the fabrication of memristive nanodevices with stable and tunable performance by assembling ferritin monolayer inside a on-wire lithography-generated 12 nm gap. This work work uses the unique high iron loading capacity of Archaeoglobus fulgidus ferritin. The iron loading in the nanocages drastically impacts the performance of the memristive devices. The higher iron loading amount contributes to better memristive performance due to higher electrochemical activity of the ferric complex core.
Researchers demonstrate a strategy for the fabrication of memristive nanodevices with stable and tunable performance by assembling ferritin monolayer inside a on-wire lithography-generated 12 nm gap. This work work uses the unique high iron loading capacity of Archaeoglobus fulgidus ferritin. The iron loading in the nanocages drastically impacts the performance of the memristive devices. The higher iron loading amount contributes to better memristive performance due to higher electrochemical activity of the ferric complex core.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





